 Back
BackChapter 17
Study Guide - Smart Notes

Chapter 17: Lipids
17.1 Introduction to Lipids
Lipids are a diverse group of biomolecules characterized by their solubility in organic solvents and insolubility in water. They play essential roles in cell membranes, energy storage, and signaling. Lipids are either based on fatty acids or a steroid nucleus.
Definition: Lipids are biomolecules containing fatty acids or a steroid nucleus.
Solubility: Soluble in nonpolar organic solvents, not in water.
Biological Importance: Components of cell membranes, fat-soluble vitamins, and steroid hormones.
Classification: Fatty acid-based lipids and steroid-based lipids.
17.2 Fatty Acids
Fatty acids are long-chain hydrocarbons with a terminal carboxylic acid group. They are classified as saturated or unsaturated based on the presence of double bonds.
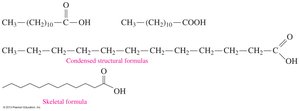
Structure: Typically 12–18 carbon atoms, with a carboxyl group (–COOH).
Solubility: Insoluble in water due to long hydrocarbon chains.
Saturated Fatty Acids: Only single C–C bonds; molecules pack closely, resulting in higher melting points and solid state at room temperature.
Unsaturated Fatty Acids: One or more C=C double bonds; cis configuration causes kinks, preventing close packing and resulting in lower melting points and liquid state at room temperature.
Properties of Saturated Fatty Acids
Fit closely together in a regular pattern.
Significant dispersion forces between chains.
Higher melting points; solid at room temperature.

Properties of Unsaturated Fatty Acids
Chains have "curls" due to cis double bonds.
Do not pack closely; fewer attractions between chains.
Lower melting points; liquid at room temperature.
Types of Unsaturated Fatty Acids
Monounsaturated: One double bond (e.g., oleic acid).
Polyunsaturated: More than one double bond (e.g., linoleic acid, linolenic acid).
Essential Fatty Acids: Cannot be synthesized by humans; must be obtained from diet.
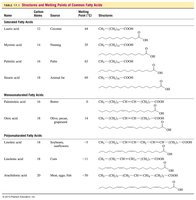
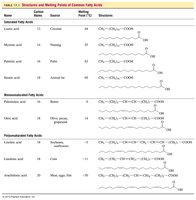
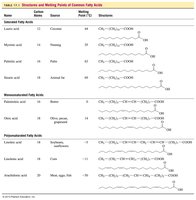
Table: Structures and Melting Points of Common Fatty Acids
Name | Carbon Atoms | Source | Melting Point (°C) | Structure |
|---|---|---|---|---|
Lauric acid | 12 | Coconut | 44 | CH3(CH2)10COOH |
Myristic acid | 14 | Nutmeg | 54 | CH3(CH2)12COOH |
Palmitic acid | 16 | Palm oil | 63 | CH3(CH2)14COOH |
Stearic acid | 18 | Animal fat | 70 | CH3(CH2)16COOH |
Oleic acid | 18 | Olive oil | 14 | CH3(CH2)7CH=CH(CH2)7COOH |
Linoleic acid | 18 | Sunflower oil | -5 | CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOH |
Linolenic acid | 18 | Flaxseed | -11 | CH3CH2(CH=CHCH2)3(CH2)6COOH |

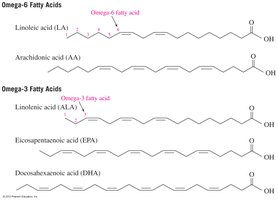
Omega-3 and Omega-6 Fatty Acids
Omega-3 and omega-6 fatty acids are types of polyunsaturated fatty acids distinguished by the position of the first double bond from the methyl end of the chain. They are important for cardiovascular health and must be obtained from the diet.
Omega-3: First double bond at C3 (e.g., linolenic acid).
Omega-6: First double bond at C6 (e.g., linoleic acid).
Health Benefits: Omega-3 fatty acids reduce blood clot formation and risk of heart disease.
17.3 Waxes and Triacylglycerols
Waxes
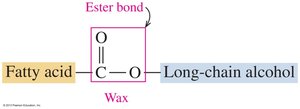
Waxes are esters formed from saturated fatty acids and long-chain alcohols. They serve as protective coatings in plants and animals.
Structure: Ester bond between fatty acid and long-chain alcohol.
Function: Prevent water loss from leaves; used in candles, polishes, and cosmetics.

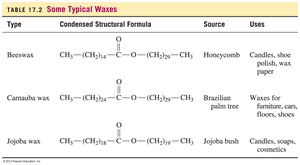
Type | Condensed Structural Formula | Source | Uses |
|---|---|---|---|
Beeswax | CH3(CH2)24COO(CH2)29CH3 | Honeycomb | Candles, shoe polish, wax paper |
Carnauba wax | CH3(CH2)24COO(CH2)33CH3 | Brazilian palm tree | Waxes for furniture, cars, shoes |
Jojoba wax | CH3(CH2)19COO(CH2)21CH3 | Jojoba bush | Candles, soaps, cosmetics |
Triacylglycerols (Fats and Oils)
Triacylglycerols are esters formed from glycerol and three fatty acids. They are the main form of energy storage in animals and plants.
Structure: Glycerol forms ester bonds with three fatty acids.
Function: Energy storage; fats are solid at room temperature, oils are liquid.
Formation: Esterification reaction between glycerol and fatty acids.

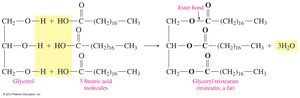
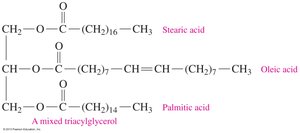
17.4 Chemical Properties of Triacylglycerols
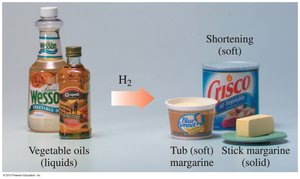
Triacylglycerols undergo reactions similar to alkenes and esters, including hydrogenation and hydrolysis.
Hydrogenation: Addition of H2 across double bonds, converting unsaturated fats to saturated fats.
Hydrolysis: Splitting ester bonds by water in the presence of acid, base, or enzyme.
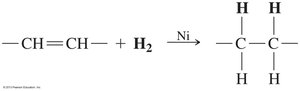
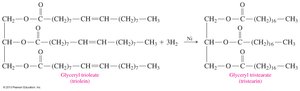
17.5 Phospholipids
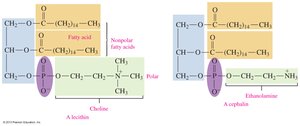
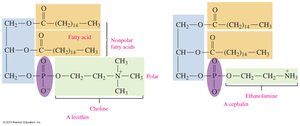
Phospholipids are major components of cell membranes. Glycerophospholipids contain two fatty acids, a phosphate group, and an amino alcohol.
Structure: Glycerol backbone, two fatty acids, phosphate group, and amino alcohol (choline, serine, or ethanolamine).
Function: Structural component of cell membranes; amphipathic nature allows interaction with both polar and nonpolar substances.
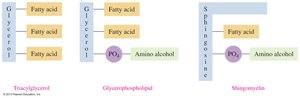
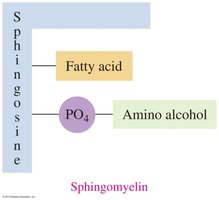
Sphingolipids
Sphingolipids are phospholipids containing sphingosine instead of glycerol. Sphingomyelins are abundant in the myelin sheath of nerve cells.
Sphingosine: 18-carbon unsaturated amino alcohol.
Sphingomyelins: Amide bond to fatty acid, ester bond to phosphate and amino alcohol.
Function: Insulate and protect nerve cells; loss leads to diseases like multiple sclerosis.
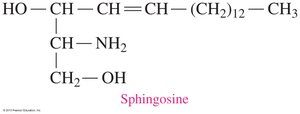
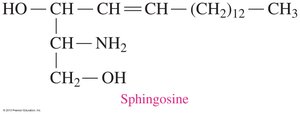
17.6 Steroids
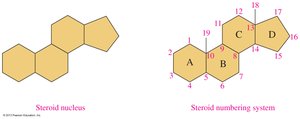
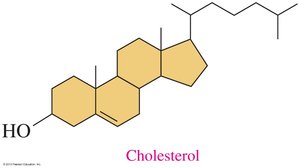
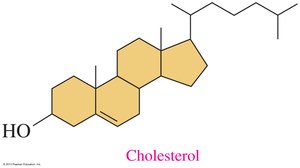
Steroids are lipids with a characteristic four-ring structure. Cholesterol is the most important steroid in the body, serving as a precursor for steroid hormones and bile salts.
Structure: Four fused rings (three cyclohexane, one cyclopentane).
Cholesterol: Component of cell membranes, precursor for hormones and bile salts.
Bile Salts: Synthesized from cholesterol; aid in digestion and absorption of lipids.
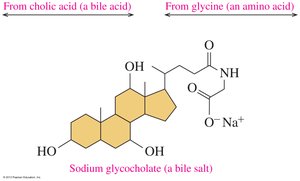
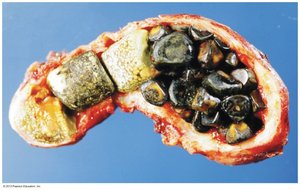
Lipoproteins
Lipoproteins are complexes that transport lipids in the blood. They differ in density, composition, and function.
Types: Chylomicrons, VLDL, LDL, HDL.
LDL: "Bad" cholesterol; deposits cholesterol in arteries.
HDL: "Good" cholesterol; removes cholesterol from tissues.
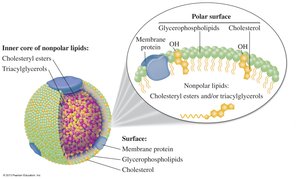
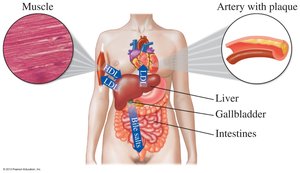
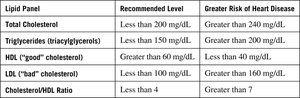
Table: Lipid Panel and Heart Disease Risk
Lipid Panel | Recommended Level | Greater Risk of Heart Disease |
|---|---|---|
Total Cholesterol | Less than 200 mg/dL | Greater than 240 mg/dL |
Triglycerides (triacylglycerols) | Less than 150 mg/dL | Greater than 200 mg/dL |
HDL ("good" cholesterol) | Greater than 60 mg/dL | Less than 40 mg/dL |
LDL ("bad" cholesterol) | Less than 100 mg/dL | Greater than 160 mg/dL |
Cholesterol/HDL Ratio | Less than 4 | Greater than 7 |
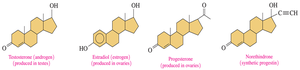
Steroid Hormones and Adrenal Corticosteroids
Steroid hormones are chemical messengers derived from cholesterol. Adrenal corticosteroids regulate metabolism and immune response.
Sex Hormones: Androgens (testosterone), estrogens (estradiol).
Adrenal Corticosteroids: Aldosterone (electrolyte balance), cortisol (stress response), prednisone (anti-inflammatory).
17.7 Cell Membranes
Cell membranes are composed of a lipid bilayer, primarily glycerophospholipids, with embedded proteins and cholesterol. The fluid mosaic model describes the dynamic nature of membranes.
Lipid Bilayer: Hydrophilic heads face outward; hydrophobic tails face inward.
Fluid Mosaic Model: Membrane is flexible, with proteins and carbohydrates for communication.
Transport Pathways: Diffusion (passive), facilitated transport (protein channels), active transport (against gradient).
