 Back
BackChapter 19: Spontaneity, Entropy, and Free Energy – Thermodynamics Study Guide
Study Guide - Smart Notes

Spontaneous Processes and Entropy
Definitions and Concepts
Thermodynamics examines the energy changes and spontaneity of chemical processes. A spontaneous process occurs without external intervention, while a nonspontaneous process requires continuous external action. The key factor governing spontaneity is the entropy of the universe, not just the enthalpy (heat change) of the system.
Spontaneous process: Occurs naturally (e.g., melting ice above 0°C, water flowing downhill).
Nonspontaneous process: Requires continuous input (e.g., pumping water uphill).
Entropy (S): A measure of molecular randomness or disorder. Greater disorder means higher entropy.
Spontaneity is driven by an increase in the entropy of the universe (Second Law of Thermodynamics).
Examples of Entropy Changes
Melting of ice: Solid to liquid increases disorder, so entropy increases.
Evaporation of a liquid: Liquid to gas greatly increases entropy due to more molecular freedom.
Dissolving a salt: Crystalline solid and pure liquid become a mixture, generally increasing entropy.
Entropy and the Second Law of Thermodynamics
Fundamental Laws
First Law: Energy is conserved.
Second Law: In any spontaneous process, the entropy of the universe increases.
Mathematically:
If , the process is spontaneous.

General Trends in Entropy Changes
Formation of liquids or solutions from solids increases entropy.
Formation of gases from solids or liquids increases entropy.
Increase in the number of gas molecules in a reaction increases entropy.
Raising temperature increases entropy due to increased molecular motion.
Predicting Entropy Changes in Reactions
Decomposition of ammonium nitrate: Produces gases, so entropy increases.
Conversion of SO2 to SO3: Fewer gas molecules, so entropy decreases.
Extraction of sucrose from solution: Crystallization decreases entropy.
Water gas shift reaction: Number of gas molecules unchanged, so entropy change is uncertain.
The Effect of Temperature on Spontaneity
Temperature and Entropy
Spontaneity depends on both the system and the surroundings. If the system's entropy decreases, the surroundings' entropy must increase by a greater amount for the process to be spontaneous. The entropy change of the surroundings is primarily determined by heat flow and temperature.
Exothermic processes () increase .
Endothermic processes () decrease .
The magnitude of is inversely proportional to temperature:
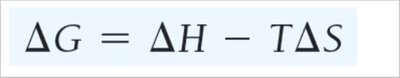
Gibbs Free Energy
Definition and Relationship to Spontaneity
Gibbs Free Energy (G) is the energy available to do work at constant temperature and pressure. The change in Gibbs free energy () determines spontaneity:
If , the process is spontaneous.
If , the process is nonspontaneous.
If , the system is at equilibrium.

Connection to Entropy of the Universe
Thus, a negative corresponds to a positive (spontaneous process).
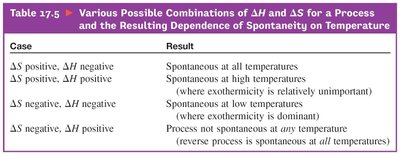
Temperature Dependence of Spontaneity
When both and are positive, the process is spontaneous at high temperatures.
When both are negative, the process is spontaneous at low temperatures.
When and , the process is spontaneous at all temperatures.
When and , the process is nonspontaneous at all temperatures.
Entropy Changes in Chemical Reactions
Standard Entropy Change
The standard entropy change for a reaction () is calculated as:
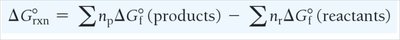
Free Energy and Chemical Reactions
Standard Free Energy Change
is the change in free energy when reactants in their standard states are converted to products in their standard states.
It can be calculated by:
The Dependence of Free Energy on Pressure
Non-Standard Conditions
For gases, free energy depends on pressure:
For reactions: , where Q is the reaction quotient.
At equilibrium: , so , where K is the equilibrium constant.
Free Energy and Equilibrium
Relationship Between ΔG and K
If , (products favored at equilibrium).
If , (reactants favored at equilibrium).
If , (system at equilibrium).
Free Energy and Work
Maximum Work
The maximum work obtainable from a process at constant T and P is .
In real systems, not all free energy is converted to work due to inefficiencies and energy loss as heat.
Summary Tables and Visuals
Key Tables
Interplay of and : Determines if a process is spontaneous based on the sign and magnitude of entropy changes.
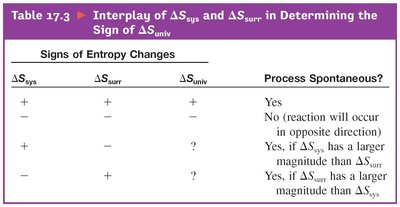
Combinations of and : Shows how spontaneity depends on temperature and the signs of enthalpy and entropy changes.

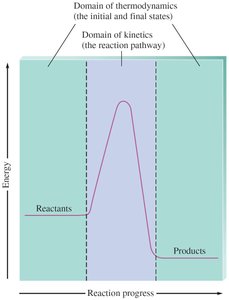
Visualizing Thermodynamics and Kinetics
The energy profile of a reaction distinguishes between thermodynamics (initial and final states) and kinetics (the reaction pathway and activation energy).
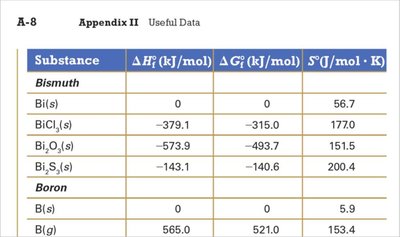
Sample Data Table
Standard enthalpy, free energy, and entropy values for selected substances are used in calculations.
Key Equations
*Additional info: Some tables and equations have been expanded for clarity and completeness. The study notes are structured to provide a comprehensive overview of thermodynamics as covered in a general chemistry course, focusing on spontaneity, entropy, and free energy.*
