 Back
BackChapter 2: Measurements in General Chemistry
Study Guide - Smart Notes

Measurements in Chemistry
Introduction to Units of Measurement
In chemistry, precise measurement is essential for experiments and calculations. The metric system and the International System of Units (SI) are universally used for scientific measurements, ensuring consistency and accuracy across the globe. Common measurements include length, volume, mass, temperature, and time.

SI and Metric Units
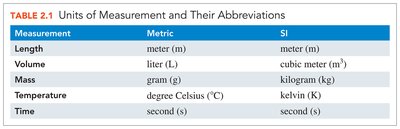
The following table summarizes the primary units used in the metric and SI systems for various types of measurements:
Measurement | Metric | SI |
|---|---|---|
Length | meter (m) | meter (m) |
Volume | liter (L) | cubic meter (m3) |
Mass | gram (g) | kilogram (kg) |
Temperature | degree Celsius (°C) | kelvin (K) |
Time | second (s) | second (s) |
Length
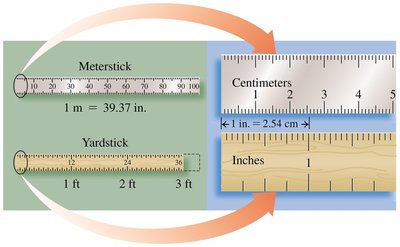
Length is measured in meters (m) in both the metric and SI systems. Chemists often use centimeters (cm) for smaller lengths. Useful relationships include:
1 m = 1.094 yd
1 m = 39.37 in.
1 m = 100 cm
2.54 cm = 1 in.
Volume
Volume is the space occupied by a substance. In the SI system, it is measured in cubic meters (m3), but chemists commonly use liters (L) and milliliters (mL). Key relationships include:
1 m3 = 1000 L
1 L = 1000 mL
1 mL = 1 cm3
1 L = 1.057 qt
946.3 mL = 1 qt

Mass
Mass measures the quantity of material in an object. The SI unit is the kilogram (kg), but grams (g) are often used in the laboratory. Important conversions include:
1 kg = 1000 g
1 kg = 2.205 lb
453.6 g = 1 lb


Temperature
Temperature indicates how hot or cold an object is. It is measured in degrees Celsius (°C) and kelvin (K) in the SI system. The Celsius scale is commonly used in chemistry, while kelvin is used for scientific calculations.

Time
Time is measured in seconds (s) in both the metric and SI systems. Accurate time measurement is essential for experiments and calculations.

Measured Numbers and Significant Figures
Measured vs. Exact Numbers
Measured numbers are obtained using measuring tools and have a degree of uncertainty. Exact numbers are obtained by counting or by definition and have no uncertainty.

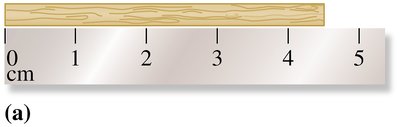
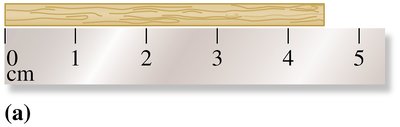
Reporting Measurements
When reporting measurements, the last digit is estimated. For example, if a ruler is marked in centimeters, you can estimate to the nearest tenth of a centimeter.

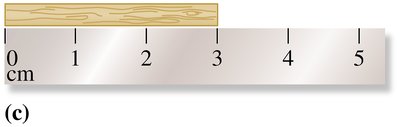
Significant Figures
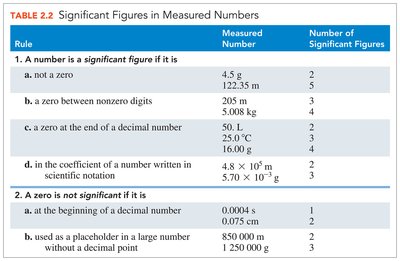
Significant figures (SFs) include all certain digits plus one estimated digit. The rules for determining significant figures are:
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Zeros at the end of a decimal number are significant.
Zeros at the beginning of a decimal number are not significant.
Zeros at the end of nondecimal numbers are not significant.
Rule | Measured Number | Number of Significant Figures |
|---|---|---|
Not a zero | 4.5 g | 2 |
Zero between nonzero digits | 205 m | 3 |
Zero at the end of a decimal | 50. L | 2 |
Zero at the beginning of a decimal | 0.0004 s | 1 |
Placeholder zero in large number | 850 000 m | 2 |
Exact Numbers
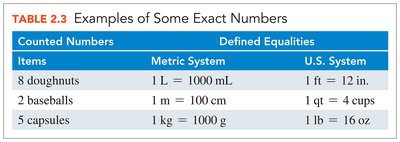
Exact numbers are obtained by counting or by definition (e.g., 1 m = 100 cm). They do not affect the number of significant figures in calculations.

Counted Numbers | Metric System | U.S. System |
|---|---|---|
8 doughnuts | 1 L = 1000 mL | 1 ft = 12 in. |
2 baseballs | 1 m = 100 cm | 1 qt = 4 cups |
5 capsules | 1 kg = 1000 g | 1 lb = 16 oz |
Significant Figures in Calculations
Rounding Off
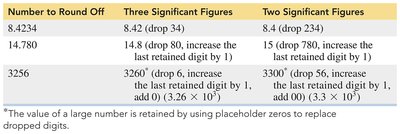
When performing calculations, the answer must be rounded to the correct number of significant figures:
If the first digit to be dropped is 4 or less, drop it and all following digits.
If the first digit to be dropped is 5 or greater, increase the last retained digit by 1.

Number to Round Off | Three Significant Figures | Two Significant Figures |
|---|---|---|
8.4234 | 8.42 | 8.4 |
14.780 | 14.8 | 15 |
3256 | 3260 | 3300 |
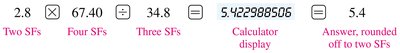
Multiplication and Division
For multiplication and division, the final answer should have the same number of significant figures as the measurement with the fewest significant figures.

Addition and Subtraction
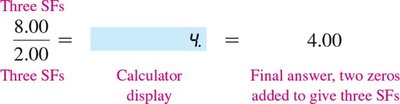
For addition and subtraction, the final answer should have the same number of decimal places as the measurement with the fewest decimal places.

Metric Prefixes and Equalities
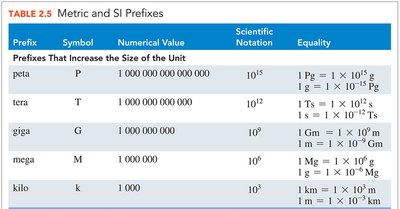
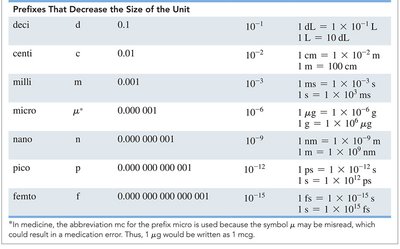
Metric and SI Prefixes
Prefixes are used to indicate multiples or fractions of units. For example, kilo- means 1000 times the unit, centi- means one-hundredth, and milli- means one-thousandth.
Prefix | Symbol | Numerical Value | Scientific Notation |
|---|---|---|---|
kilo | k | 1,000 | 103 |
centi | c | 0.01 | 10-2 |
milli | m | 0.001 | 10-3 |
micro | μ | 0.000001 | 10-6 |
Equalities and Conversion Factors
Equalities show the relationship between two units that measure the same quantity. For example:
1 m = 100 cm = 1 × 102 cm
1 m = 1000 mm = 1 × 103 mm
1 cm = 10 mm = 1 × 101 mm
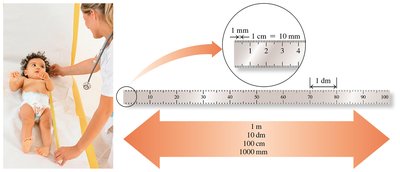
Writing and Using Conversion Factors
Writing Conversion Factors
Conversion factors are ratios derived from equalities that allow conversion from one unit to another. For example, from 1 m = 100 cm, the conversion factors are:
1 m / 100 cm
100 cm / 1 m
Conversion Factors with Powers
When converting area or volume, both the number and the unit must be squared or cubed. For example:
Equality: 1 m = 100 cm
Area: (1 m)2 = (100 cm)2 or 1 m2 = 10,000 cm2
Problem Solving Using Unit Conversion
Problem-Solving Process
To solve unit conversion problems:
Identify the given and needed units.
Write a plan to convert the given unit to the needed unit.
Identify conversion factors that cancel units and provide the needed unit.
Set up and solve the calculation.
Density and Specific Gravity
Density
Density compares the mass of an object to its volume and is defined as:
Units for density:
Solids/Liquids: grams per cubic centimeter (g/cm3) or grams per milliliter (g/mL)
Gases: grams per liter (g/L)
Specific Gravity
Specific gravity is the ratio of the density of a substance to the density of water (1.00 g/mL at 4°C):
It is a unitless quantity and is commonly used in clinical and laboratory settings.
*Additional info: This summary covers all major topics from Chapter 2: Measurements, including units, significant figures, conversion factors, and density, with relevant images and tables to reinforce key concepts.*
