Back
BackChapter 20: Nuclear Chemistry – Structure, Reactions, and Applications
Study Guide - Smart Notes

Chapter 20: Nuclear Chemistry
Nuclear Structure and Stability
Nuclear chemistry focuses on the structure, stability, and transformations of atomic nuclei. Unlike chemical reactions, which involve electrons, nuclear reactions involve changes in the nucleus, often resulting in the emission of particles and energy.
Nucleus Composition: The nucleus contains protons (positively charged) and neutrons (neutral), collectively called nucleons.
Isotopes: Atoms of the same element with different numbers of neutrons. Identified by their mass number (A) = protons + neutrons.
Nuclide: A specific isotope of an element.
Stability: Determined by the neutron-to-proton (N/Z) ratio and the presence of certain 'magic numbers' of nucleons (2, 8, 20, 28, 50, 82, 126).
Band of Stability: Stable nuclei fall within a narrow band on a plot of neutrons vs. protons. Nuclei outside this band are radioactive.
Number of Stable Isotopes | Proton Number | Neutron Number |
|---|---|---|
157 | even | even |
53 | even | odd |
50 | odd | even |
5 | odd | odd |
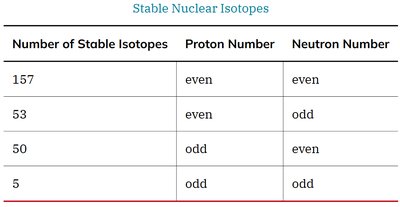
Magic Numbers: Nuclei with certain numbers of protons or neutrons (2, 8, 20, 28, 50, 82, 126) are especially stable, similar to noble gas electron configurations.
Radioactivity and Types of Radiation
Radioactivity is the spontaneous emission of particles or energy from unstable nuclei. This process can change one element into another (transmutation).
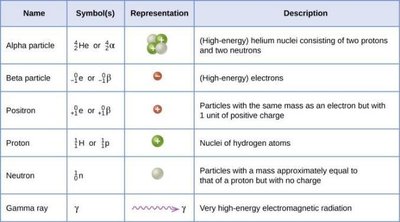
Alpha (α) Decay: Emission of a helium nucleus (2 protons, 2 neutrons).
Beta (β) Decay: Emission of an electron (β-) or positron (β+).
Gamma (γ) Emission: Emission of high-energy photons; no change in atomic or mass number.
Electron Capture: An inner electron is captured by the nucleus, converting a proton to a neutron.
Name | Symbol(s) | Representation | Description |
|---|---|---|---|
Alpha particle | or |
| Helium nuclei (2 protons, 2 neutrons) |
Beta particle | or | High-energy electrons | |
Positron | or | Electron with positive charge | |
Proton | or | Hydrogen nucleus | |
Neutron | Neutral nucleon | ||
Gamma ray | High-energy electromagnetic radiation |
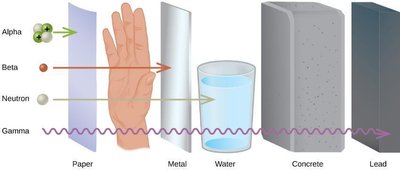
Penetrating Power of Radiation
The ability of radiation to penetrate matter varies:
Alpha: Stopped by paper or skin.
Beta: Stopped by metal or thick plastic.
Gamma: Requires thick lead or concrete for shielding.
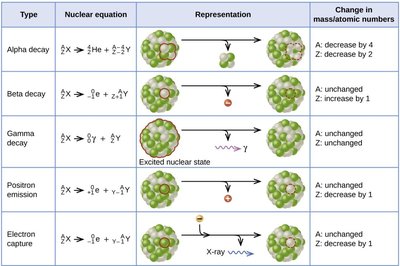
Nuclear Equations and Decay Types
Nuclear equations represent the transformation of one nuclide into another, conserving both atomic and mass numbers.
Type | Nuclear equation | Representation | Change in mass/atomic numbers |
|---|---|---|---|
Alpha decay |
| A: decrease by 4 Z: decrease by 2 | |
Beta decay | A: unchanged Z: increase by 1 | ||
Gamma decay | A: unchanged Z: unchanged | ||
Positron emission | A: unchanged Z: decrease by 1 | ||
Electron capture | A: unchanged Z: decrease by 1 |
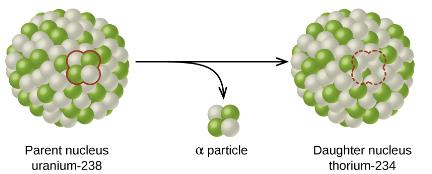
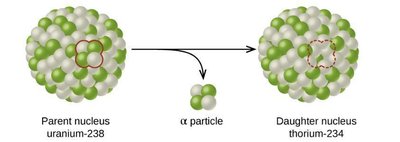
Example: Alpha Decay of Uranium-238
When uranium-238 undergoes alpha decay, it emits an alpha particle and forms thorium-234:
Equation:
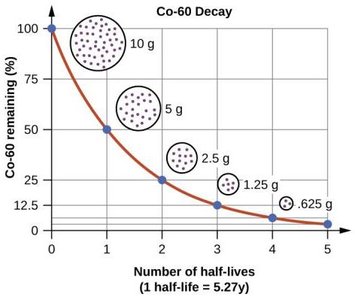
Radioactive Decay Kinetics and Half-Life
Radioactive decay follows first-order kinetics, meaning the rate depends on the number of radioactive nuclei present. The half-life () is the time required for half of the radioactive nuclei to decay.
First-order decay law:
or
Half-life relationship:
Shorter half-lives mean more rapid decay ("hotter" samples).
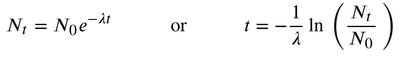
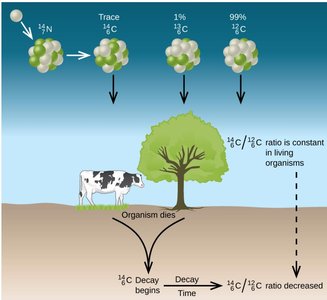
Applications: Radiometric and Radiocarbon Dating
Radioactive decay is used to date geological and archaeological samples by comparing the ratio of parent to daughter nuclides and using known half-lives.
Radiocarbon Dating: Measures the ratio of to in formerly living material. Useful for dating objects up to about 50,000 years old.
Nuclear Transmutation, Fission, and Fusion
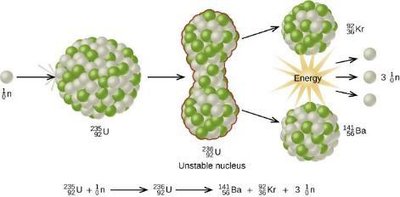
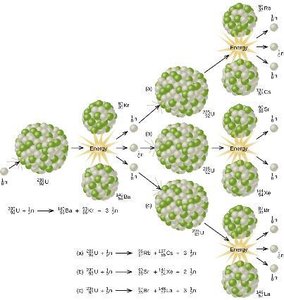
Transmutation is the conversion of one element into another via nuclear reactions. Two major types of energy-releasing nuclear reactions are fission and fusion.
Fission: Splitting a heavy nucleus (e.g., ) into smaller nuclei, releasing energy and neutrons. Used in nuclear reactors and atomic bombs.
Fusion: Combining light nuclei (e.g., hydrogen isotopes) to form a heavier nucleus, releasing even more energy per gram than fission. Powers the sun and hydrogen bombs.
Chain Reaction: In fission, released neutrons can induce further fission events, leading to a self-sustaining chain reaction if a critical mass is present.
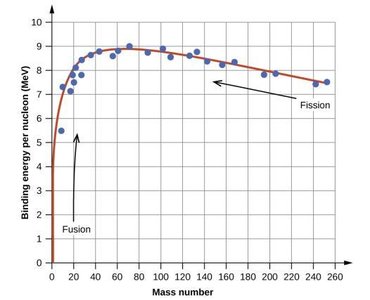
Mass Defect and Nuclear Binding Energy
When nucleons combine to form a nucleus, some mass is lost and converted to energy (binding energy), which holds the nucleus together.
Mass defect (): Difference between the mass of separated nucleons and the nucleus.
Binding energy:
Binding energy per nucleon: Indicates nuclear stability; highest for nuclei with mass number around 56.
Biological Effects of Radiation
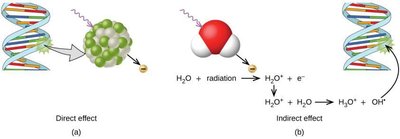
Ionizing radiation can damage biological molecules directly or indirectly (via the formation of reactive species such as hydroxyl radicals from water). The extent of damage depends on the type and dose of radiation.
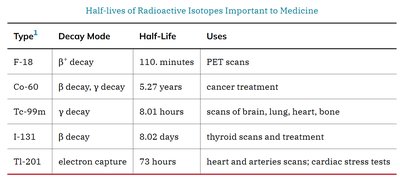
Summary Table: Half-Lives of Radioactive Isotopes Important to Medicine
Type | Decay Mode | Half-Life | Uses |
|---|---|---|---|
F-18 | β− decay | 110 minutes | PET scans |
Co-60 | β− decay, γ decay | 5.27 years | Cancer treatment |
Tc-99m | γ decay | 8.01 hours | Scans of brain, lung, heart, bone |
I-131 | β− decay | 8.02 days | Thyroid scans and treatment |
Tl-201 | Electron capture | 73 hours | Heart and arteries scans; cardiac stress tests |
Additional info: Nuclear chemistry is foundational for understanding radioactive dating, nuclear energy, and the biological effects of radiation. Mastery of nuclear equations, decay kinetics, and the properties of nuclear particles is essential for further study in chemistry and related fields.