 Back
BackChapter 21: Nuclear Chemistry – Study Notes
Study Guide - Smart Notes

Chapter 21: Nuclear Chemistry
21.1 Radioactivity and Nuclear Equations
Nuclear chemistry studies the changes that occur in atomic nuclei. Unlike chemical reactions, which involve electrons, nuclear reactions involve changes in the nucleus, often resulting in the emission of radiation and the transformation of elements.
Isotopes: Atoms of the same element with different numbers of neutrons. Example: Uranium-234, Uranium-235, Uranium-238.
Radioactivity: The spontaneous emission of radiation by certain unstable nuclei (radionuclides).
Nuclear Equations: Must balance both atomic number (Z) and mass number (A).
Types of Radioactive Decay
Alpha (α) Decay: Emission of a helium-4 nucleus (2 protons, 2 neutrons). Decreases atomic number by 2 and mass number by 4.
Beta (β) Decay: Emission of a high-speed electron. Increases atomic number by 1; mass number unchanged.
Gamma (γ) Emission: Emission of high-energy photons. No change in atomic or mass number; often accompanies other decays.
Positron Emission: Emission of a positron (same mass as electron, charge +1). Decreases atomic number by 1; mass number unchanged.
Electron Capture: An inner electron is captured by the nucleus, converting a proton to a neutron. Decreases atomic number by 1; mass number unchanged.
Type | Nuclear Equation | Δ Atomic Number | Δ Mass Number |
|---|---|---|---|
Alpha emission | -2 | -4 | |
Beta emission | +1 | 0 | |
Positron emission | -1 | 0 | |
Electron capture | -1 | 0 |
Summary of Nuclear Particles
Particle | Symbol |
|---|---|
Neutron | |
Proton | |
Electron (Beta) | |
Alpha particle | |
Positron |
21.2 Patterns of Nuclear Stability
Nuclear stability depends on the ratio of neutrons to protons (n/p) and the total number of nucleons. The strong nuclear force holds the nucleus together, but too many or too few neutrons can cause instability.
Belt of Stability: Stable nuclei are found in a region where the n/p ratio is optimal (about 1:1 for small nuclei, higher for larger nuclei).
Unstable Nuclei: Nuclei above the belt (too many neutrons) decay by beta emission; nuclei below the belt (too many protons) decay by positron emission or electron capture. Nuclei with Z > 83 are unstable and decay by alpha emission.
Magic Numbers: Nuclei with 2, 8, 20, 28, 50, 82 protons or 2, 8, 20, 28, 50, 82, 126 neutrons are especially stable.
Even Numbers: Nuclei with even numbers of protons and neutrons are more stable than those with odd numbers.


Proton Number | Neutron Number | Number of Stable Isotopes |
|---|---|---|
Even | Even | 157 |
Even | Odd | 53 |
Odd | Even | 50 |
Odd | Odd | 5 |
21.3 Nuclear Transmutations
Nuclear transmutation occurs when a nucleus is struck by another particle, causing a change in its composition. This process is used to create synthetic isotopes and new elements.
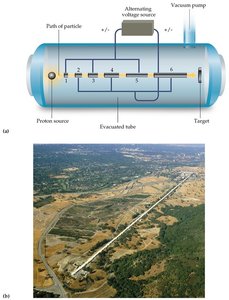
Particle Accelerators: Devices such as linear accelerators, cyclotrons, and synchrotrons accelerate particles to high speeds for nuclear collisions.
Transuranium Elements: Elements with atomic numbers greater than 92, produced by neutron bombardment or by colliding large nuclei with light nuclei at high energy.

21.4 Kinetics of Radioactive Decay
Radioactive decay follows first-order kinetics, meaning the rate depends on the amount of radioactive substance present. The half-life is the time required for half of a sample to decay.
Decay Law:
Half-life:
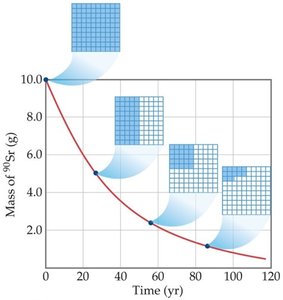
Radiometric Dating
By measuring the ratio of parent to daughter isotopes and knowing the half-life, the age of objects can be determined (e.g., carbon dating, uranium-lead dating).

21.5 Detection of Radioactivity
Several methods are used to detect and measure radioactivity:
Film Badges: Measure cumulative exposure to radiation.
Geiger Counters: Detect and measure ionizing radiation by counting electrical pulses generated by ionized gas.
Scintillation Counters: Use phosphors that emit light when struck by radiation; the light is converted to an electronic signal.
Radiotracers: Radioisotopes used to trace chemical and biological processes.


21.6 Energy Changes in Nuclear Reactions
Nuclear reactions involve large energy changes due to the conversion of mass to energy, as described by Einstein's equation . The mass defect is the difference between the mass of a nucleus and the sum of its nucleons, representing the nuclear binding energy.
Binding Energy: The energy required to break a nucleus into its component protons and neutrons.
Fission: Splitting heavy nuclei into smaller nuclei, releasing energy.
Fusion: Combining small nuclei to form larger ones, releasing even more energy per nucleon than fission.
21.7 Nuclear Power: Fission and Fusion
Fission: Used in nuclear power plants. A chain reaction is sustained if enough fissile material (critical mass) is present. Control rods absorb neutrons to regulate the reaction.
Fusion: Powers stars and hydrogen bombs. Fusion of light nuclei (e.g., hydrogen isotopes) releases enormous energy but requires extremely high temperatures and pressures.
21.9 Radiation in the Environment and Living Systems
We are constantly exposed to natural and artificial sources of radiation. Ionizing radiation can damage biological molecules, especially water, leading to the formation of reactive radicals that can harm DNA and other cellular components.
Radiation Dose Units: Gray (Gy), rad, rem (relative biological effectiveness included).
Biological Effects: Effects depend on type, amount, and location of exposure. Alpha particles are most dangerous if ingested or inhaled; gamma rays are most dangerous externally.
Average Exposure: About 360 mrem/year from natural sources.
Dose (rem) | Effect |
|---|---|
0-25 | No detectable clinical effects |
25-50 | Slight, temporary decrease in white blood cell counts |
100-200 | Nausea; marked decrease in white blood cell counts |
500 | Death of half the exposed population within 30 days |
Additional info: Nuclear chemistry is essential for understanding energy production, medical diagnostics, dating of ancient objects, and the risks and benefits of radiation in our environment.
