 Back
BackChapter 3: Matter and Energy – Classification, Properties, Temperature, and Energy Calculations
Study Guide - Smart Notes

Chapter 3: Matter and Energy
Classification of Matter
Matter is anything that has mass and occupies space. It can be classified based on its composition into pure substances and mixtures.
Pure Substances: Have a fixed or definite composition. They are further divided into:
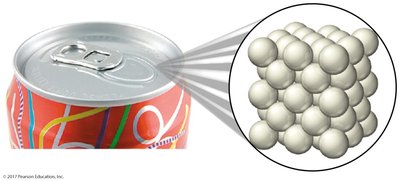
Elements: Substances composed of only one type of atom (e.g., copper, aluminum).
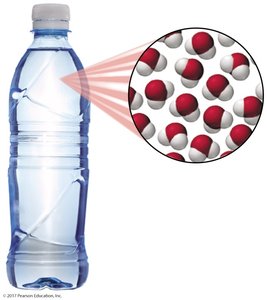
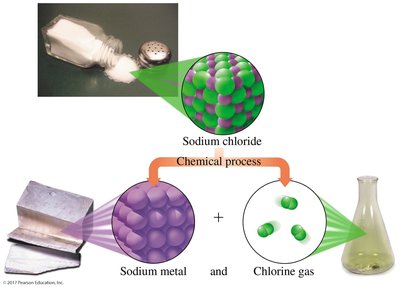
Compounds: Substances composed of two or more elements chemically combined in a fixed ratio (e.g., water, NaCl).
Mixtures: Consist of two or more substances physically combined. They can be separated by physical methods and are classified as:
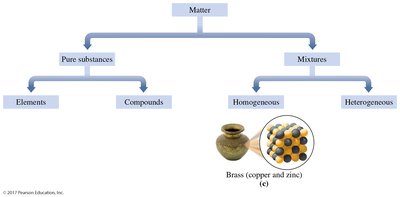
Homogeneous Mixtures (Solutions): Uniform composition throughout; components are not visibly distinguishable (e.g., brass, air).
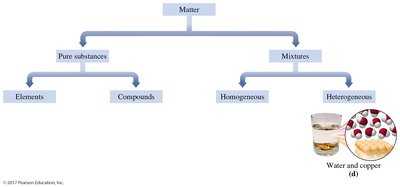
Heterogeneous Mixtures: Composition varies; different parts are visible (e.g., water and copper, salad).
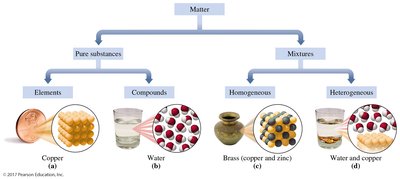
Pure Substances: Elements and Compounds
Elements: Cannot be broken down into simpler substances by chemical means. Examples include copper (Cu), lead (Pb), and aluminum (Al).
Compounds: Can be decomposed into simpler substances (elements) by chemical processes. For example, water (H2O) can be broken down into hydrogen and oxygen.
Mixtures and Their Separation
Mixtures: Can be separated by physical methods such as filtration, distillation, or chromatography.
Examples: Spaghetti and water can be separated using a strainer (filtration).


Homogeneous and Heterogeneous Mixtures
Homogeneous Mixtures: Uniform throughout; examples include brass (copper and zinc alloy).
Heterogeneous Mixtures: Non-uniform; examples include water and copper.
States and Properties of Matter
States of Matter
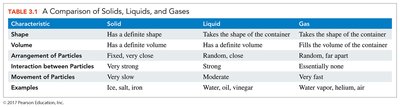
Matter exists in three physical states: solid, liquid, and gas. Each state has distinct properties based on particle arrangement and movement.
Solids: Definite shape and volume; particles are closely packed and vibrate in place.
Liquids: Definite volume but no definite shape; particles move more freely but remain close together.
Gases: No definite shape or volume; particles are far apart and move rapidly.
Physical and Chemical Properties
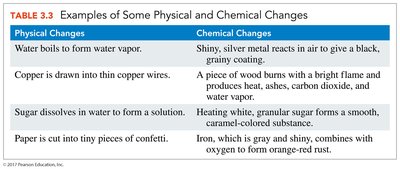
Physical Properties: Observed or measured without changing the substance's identity (e.g., color, melting point, density).
Chemical Properties: Describe the ability of a substance to undergo chemical changes (e.g., flammability, reactivity).
Physical and Chemical Changes
Physical Changes: Do not alter the chemical composition (e.g., melting, boiling, dissolving).
Chemical Changes: Result in the formation of new substances (e.g., rusting, burning).

Temperature and Its Measurement
Temperature Scales
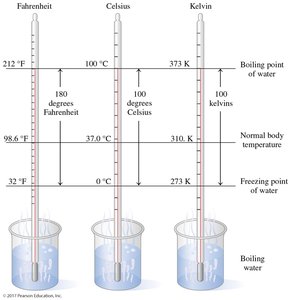
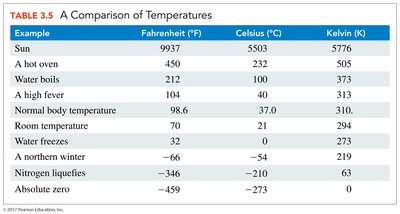
Temperature is a measure of the average kinetic energy of particles. The three main temperature scales are Fahrenheit (°F), Celsius (°C), and Kelvin (K).
Celsius: Used in science; freezing point of water is 0°C, boiling point is 100°C.
Fahrenheit: Used in the United States; freezing point of water is 32°F, boiling point is 212°F.
Kelvin: The SI unit; absolute zero (0 K) is the lowest possible temperature.
Temperature Conversions
To convert from Celsius to Fahrenheit:
To convert from Fahrenheit to Celsius:
To convert from Celsius to Kelvin:
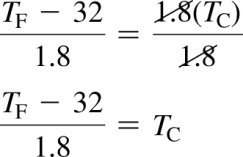

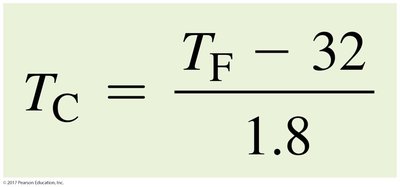
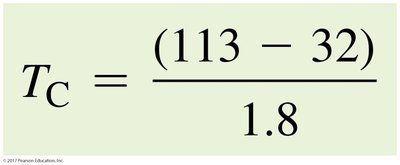
Energy: Forms and Calculations
Kinetic and Potential Energy
Kinetic Energy: Energy of motion (e.g., moving water, a rolling ball).
Potential Energy: Stored energy due to position or composition (e.g., water behind a dam, chemical bonds).

Heat and Units of Energy
Joule (J): SI unit of energy. 1 kJ = 1000 J.
Calorie (cal): Amount of energy needed to raise 1 g of water by 1°C. 1 cal = 4.184 J (exact).
kilocalorie (kcal): 1 kcal = 1000 cal.
Specific Heat
Specific Heat (SH): The amount of heat required to raise the temperature of 1 g of a substance by 1°C.
Equation:
Where is heat (J), is mass (g), is specific heat (J/g°C), and is the temperature change (°C).
Energy and Nutrition
Energy in food is measured in Calories (1 Cal = 1 kcal = 4184 J).
Carbohydrates, fats, and proteins provide different amounts of energy per gram.
Energy values are determined using a calorimeter and are listed on food labels.
Summary Table: Physical and Chemical Properties and Changes
Physical | Chemical |
|---|---|
Property: Color, shape, odor, luster, size, melting point, density | Property: Ability to form another substance (e.g., paper can burn, iron can rust) |
Change: Change of state, size, or shape (identity retained) | Change: Original substance converted to new substances (e.g., paper burns, iron rusts) |

Additional info: These notes cover the classification of matter, physical and chemical properties and changes, temperature scales and conversions, energy forms, specific heat, and energy in nutrition, all of which are foundational topics in general chemistry.
