 Back
BackChapter 3: Molecules, Compounds, and Chemical Equations – Structured Study Notes
Study Guide - Smart Notes

Chapter 3: Molecules, Compounds, and Chemical Equations
Introduction to Molecules and Compounds
Molecules and compounds are fundamental units in chemistry, formed by atoms held together through chemical bonds. Understanding their properties, formation, and classification is essential for mastering general chemistry.
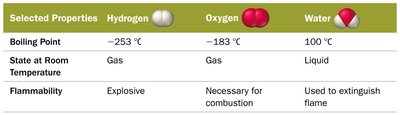
Selected Properties of Hydrogen, Oxygen, and Water
Hydrogen: Gas at room temperature, explosive, boiling point −253°C.
Oxygen: Gas at room temperature, necessary for combustion, boiling point −183°C.
Water: Liquid at room temperature, used to extinguish flames, boiling point 100°C.

Mixtures vs. Compounds
Mixtures: Can have any ratio of components (e.g., hydrogen and oxygen mixture).
Compounds: Have a fixed ratio of elements (e.g., water always has two hydrogen atoms to one oxygen atom).
Chemical Bonds
Chemical bonds are the forces that hold atoms together in compounds. They arise from attractions between charged particles (electrons and protons).
Ionic Bonds: Occur between metals and nonmetals; involve transfer of electrons, forming cations and anions.
Covalent Bonds: Occur between nonmetals; involve sharing of electrons, forming molecules.
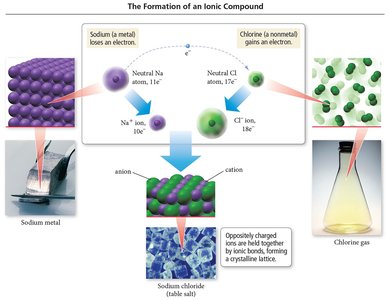
The Formation of Ionic Compounds
Metal atoms lose electrons to become cations.
Nonmetal atoms gain electrons to become anions.
Electrostatic attraction between oppositely charged ions forms ionic bonds.
Ionic vs. Molecular Compounds
Ionic Compounds: Composed of cations (usually metals) and anions (usually nonmetals), bound by ionic bonds. Basic unit is the formula unit (e.g., NaCl).
Molecular Compounds: Composed of molecules formed by covalent bonds between nonmetals.

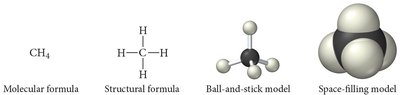
Types of Chemical Formulas
Chemical formulas represent the composition of compounds in different ways:
Empirical Formula: Shows the simplest whole-number ratio of atoms.
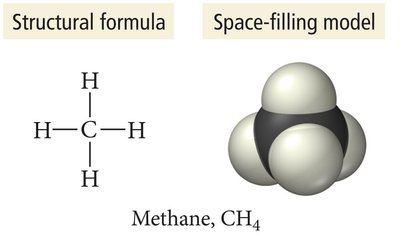
Molecular Formula: Shows the actual number of atoms of each element in a molecule.
Structural Formula: Shows how atoms are connected, including geometry.
Molecular Models
Ball-and-Stick Model: Atoms as balls, bonds as sticks; color-coded for elements.
Space-Filling Model: Atoms fill space between each other, representing molecular shape more realistically.
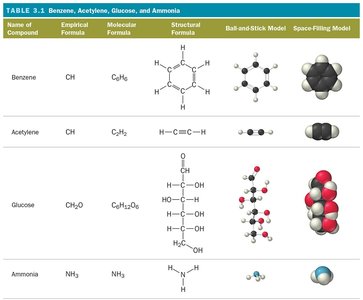
Comparison of Formula Types
Name of Compound | Empirical Formula | Molecular Formula | Structural Formula | Ball-and-Stick Model | Space-Filling Model |
|---|---|---|---|---|---|
Benzene | CH | C6H6 | Ring structure | Ball-and-stick | Space-filling |
Acetylene | CH | C2H2 | Linear structure | Ball-and-stick | Space-filling |
Glucose | CH2O | C6H12O6 | Ring structure | Ball-and-stick | Space-filling |
Ammonia | NH3 | NH3 | Trigonal pyramidal | Ball-and-stick | Space-filling |
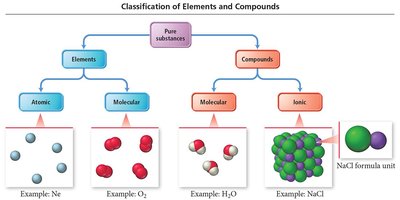
Classification of Elements and Compounds
Pure substances are classified as elements or compounds. Elements can be atomic or molecular, while compounds can be molecular or ionic.
Atomic Elements: Exist as single atoms (e.g., Ne, Na).
Molecular Elements: Exist as molecules (e.g., O2, N2, P4, S8).
Molecular Compounds: Molecules formed by covalent bonds (e.g., H2O).
Ionic Compounds: Formula units formed by ionic bonds (e.g., NaCl).
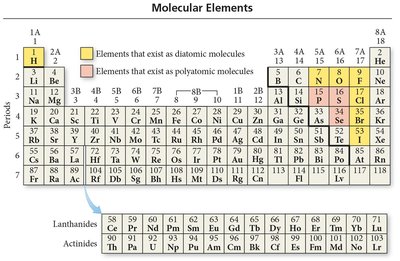
Molecular Elements in the Periodic Table
Seven diatomic elements: H2, N2, O2, F2, Cl2, Br2, I2.
Polyatomic elements: P4, S8.
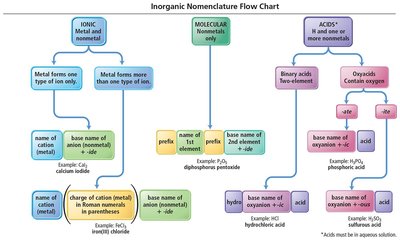
Naming Ionic Compounds
Ionic compounds are named based on the type of metal present:
Type I: Metal forms only one type of ion (charge is invariant).
Type II: Metal forms more than one type of ion (charge varies).
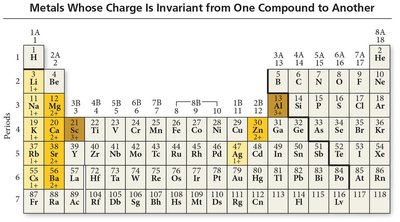
Type I Ionic Compounds
Metal ion always has the same charge.
Binary compounds contain only two different elements.
Name: cation name + base name of anion (nonmetal) + -ide.

Examples of Type I Binary Ionic Compounds
KCl: Potassium chloride
CaO: Calcium oxide
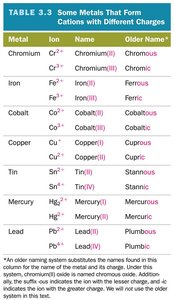
Type II Ionic Compounds
Metal can form more than one kind of cation (often transition metals).
Name: cation name + charge in Roman numerals (in parentheses) + base name of anion + -ide.

Polyatomic Ions
Groups of covalently bonded atoms with an overall charge.
Common polyatomic ions include nitrate (NO3−), carbonate (CO32−), sulfate (SO42−), etc.
Name ionic compounds containing polyatomic ions using the ion's name.
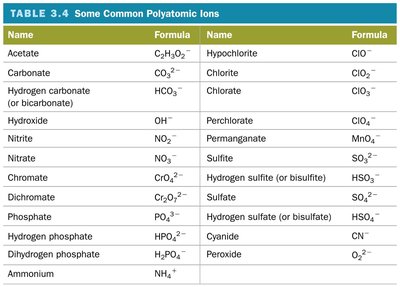
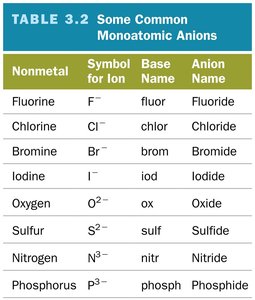
Oxyanions
Anions containing oxygen and another element.
Named based on the number of oxygen atoms: -ate (more O), -ite (less O).
Prefixes hypo- (less than) and per- (more than) are used for series with more than two ions.
Hydrated Ionic Compounds
Contain a specific number of water molecules per formula unit.
Named using prefixes: mono-, di-, tri-, tetra-, penta-, hexa-, hepta-, octa-.
Example: MgSO4·7H2O is magnesium sulfate heptahydrate.
Naming Molecular Compounds
Molecular compounds are named using prefixes to indicate the number of each element present.
Prefixes: mono-, di-, tri-, tetra-, penta-, hexa-, hepta-, octa-, nona-, deca-.
If only one atom of the first element, the prefix mono- is omitted.
Name: prefix + name of first element + prefix + base name of second element + -ide.

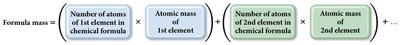
Formula Mass and Molar Mass
The mass of a molecule or formula unit is calculated by summing the atomic masses of its constituent atoms.
Formula Mass: Sum of atomic masses in a chemical formula.
Molar Mass: Mass of one mole of a compound, expressed in grams per mole.
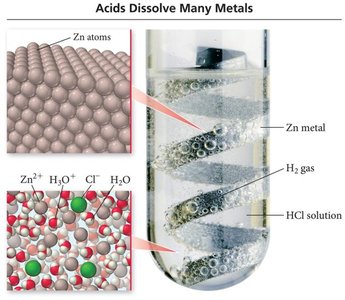
Acids
Acids are molecular compounds that release hydrogen ions (H+) when dissolved in water. They are characterized by sour taste and ability to dissolve many metals.
Binary Acids: Contain H+ and a nonmetal anion.
Oxyacids: Contain H+ and a polyatomic anion with oxygen.
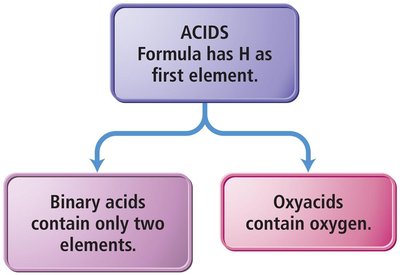

Naming Binary Acids
Prefix hydro- + base name of nonmetal + -ic + "acid".
Example: HCl(aq) is hydrochloric acid.

Naming Oxyacids
If polyatomic ion ends in -ate, change to -ic + "acid".
If polyatomic ion ends in -ite, change to -ous + "acid".

Inorganic Nomenclature Flow Chart
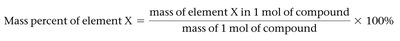
Composition of Compounds
The percentage by mass of each element in a compound can be determined from its formula or experimental analysis.
Mass Percent Formula:

Determining Empirical and Molecular Formulas
The empirical formula gives the simplest ratio of atoms, while the molecular formula gives the actual number of atoms. Experimental data and molar mass are used to determine these formulas.
Empirical Formula: Simplest whole-number ratio.
Molecular Formula: Whole-number multiple of empirical formula.
Formula:
n calculation:
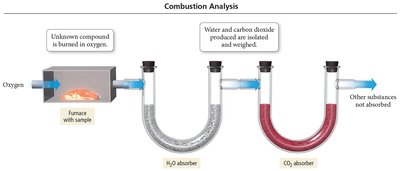
Combustion Analysis
Used to determine empirical formulas by burning a compound in oxygen and measuring the products.
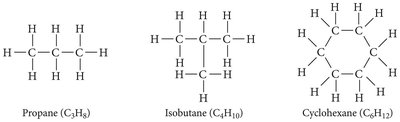
Organic Compounds and Hydrocarbons
Organic compounds are primarily composed of carbon and hydrogen, sometimes with other elements. Hydrocarbons are the simplest organic compounds, containing only carbon and hydrogen.
Alkanes: Only single bonds (suffix -ane).
Alkenes: At least one double bond (suffix -ene).
Alkynes: At least one triple bond (suffix -yne).
Base names: meth-, eth-, prop-, but-, pent-, hex-, hept-, oct-, non-, dec-.
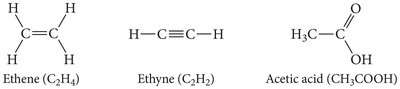
Functionalized Hydrocarbons
Functional groups impart specific chemical properties to organic compounds.
Families of organic compounds are defined by their functional groups.
Example: Alcohols (–OH group), carboxylic acids (–COOH group), amines (–NH2 group).
