 Back
BackChapter 3: Molecules, Compounds, and Chemical Equations - Structured Study Notes
Study Guide - Smart Notes

Molecules, Compounds, and Chemical Equations
Chemical Bonds and Types of Compounds
Chemical bonds are the forces that hold atoms together in compounds. These bonds arise from the attractions between charged particles (electrons and protons) within atoms. Compounds are classified based on the nature of their bonds: ionic or covalent.
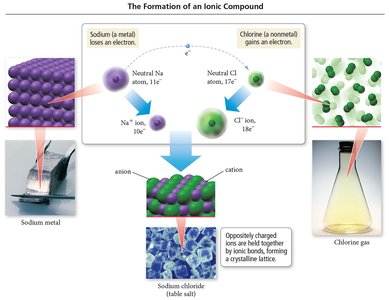
Ionic Bonds: Occur between metals and nonmetals, involving the transfer of electrons from the metal to the nonmetal. The metal becomes a cation, and the nonmetal becomes an anion. The resulting oppositely charged ions attract each other, forming an ionic bond.
Covalent Bonds: Occur between two or more nonmetals, where atoms share electrons, forming molecules. These compounds are also called molecular compounds.
Properties and Classification of Elements and Compounds
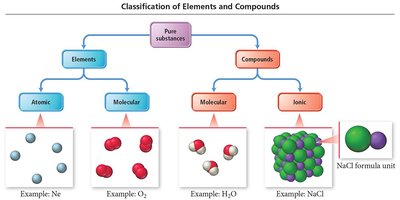
Elements and compounds can be classified based on their atomic or molecular nature and the type of bonding present.
Atomic Elements: Exist as single atoms (e.g., Na, Ne).
Molecular Elements: Exist as molecules, typically diatomic (H2, N2, O2, F2, Cl2, Br2, I2) or polyatomic (P4, S8).
Molecular Compounds: Composed of molecules formed by covalent bonds.
Ionic Compounds: Composed of cations and anions held together by ionic bonds.
Types of Chemical Formulas
Chemical formulas represent the composition of compounds in different ways:
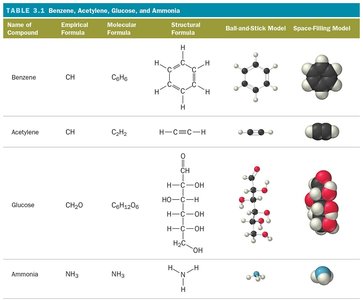
Empirical Formula: Shows the simplest whole-number ratio of atoms in a compound.
Molecular Formula: Shows the actual number of atoms of each element in a molecule.
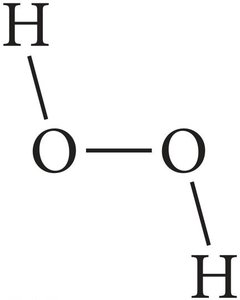
Structural Formula: Uses lines to represent covalent bonds and shows how atoms are connected.
Example: For H2O2, the empirical formula is HO, the molecular formula is H2O2, and the structural formula shows the arrangement of atoms.
Molecular Models
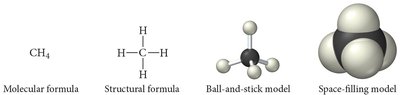
Molecular models help visualize the structure and geometry of molecules:
Ball-and-Stick Model: Atoms are balls, bonds are sticks, and colors represent different elements.
Space-Filling Model: Atoms fill the space between each other, closely representing the molecule's actual shape.
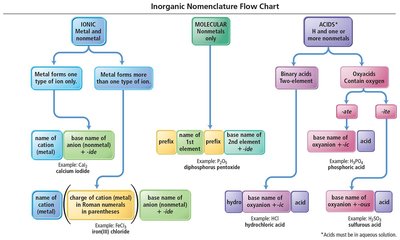
Naming Ionic Compounds
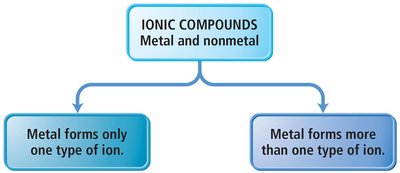
Ionic compounds are named based on the type of metal present:
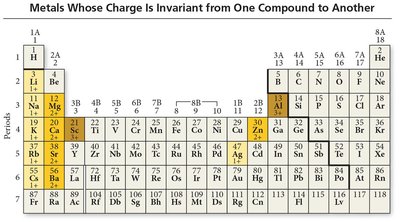
Type I Ionic Compounds: Contain metals with invariant charge (e.g., Na+, Ca2+). The name is the metal followed by the base name of the anion with the suffix '-ide'.
Type II Ionic Compounds: Contain metals that can form more than one type of cation (often transition metals). The charge is specified using Roman numerals in parentheses.


Polyatomic Ions and Oxyanions
Many ionic compounds contain polyatomic ions, which are groups of covalently bonded atoms with an overall charge. Oxyanions are polyatomic ions containing oxygen and another element.
Oxyanions: Named based on the number of oxygen atoms. '-ate' for more oxygen, '-ite' for fewer. Prefixes 'hypo-' and 'per-' are used for series with more than two ions.
Hydrated Ionic Compounds
Hydrates are ionic compounds with a specific number of water molecules associated with each formula unit. Prefixes such as mono-, di-, tri-, etc., indicate the number of water molecules.
Naming Molecular Compounds
Molecular compounds are named using prefixes to indicate the number of each type of atom present. The prefix 'mono-' is usually omitted for the first element.
Prefixes: mono-, di-, tri-, tetra-, penta-, hexa-, hepta-, octa-, nona-, deca-
Example: CO2 is carbon dioxide, N2O4 is dinitrogen tetroxide.

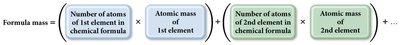
Formula Mass and Molar Mass
The formula mass (or molecular mass) is the sum of the atomic masses of all atoms in a molecule or formula unit. The molar mass is the mass of one mole of a compound.
Formula Mass Equation:
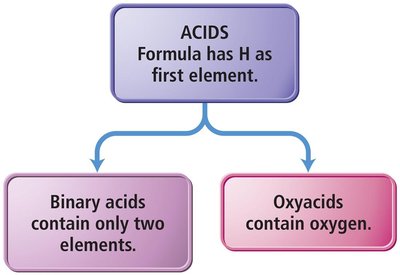
Acids: Properties and Naming
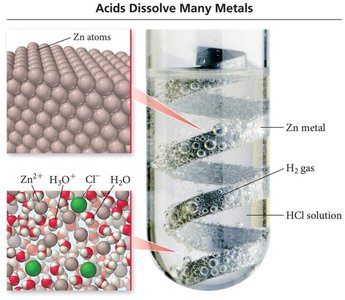
Acids are molecular compounds that release hydrogen ions (H+) when dissolved in water. They are characterized by sour taste and their ability to dissolve many metals.
Binary Acids: Contain H+ and a nonmetal anion. Named with 'hydro-' prefix, base name of nonmetal, '-ic' suffix, and 'acid'.
Oxyacids: Contain H+ and a polyatomic oxyanion. If the oxyanion ends in '-ate', the acid name ends in '-ic'; if '-ite', it ends in '-ous'.



Composition of Compounds and Empirical Formulas
The composition of compounds can be described by the percentage by mass of each element. The empirical formula is the simplest whole-number ratio of atoms in a compound.
Mass Percent Equation:
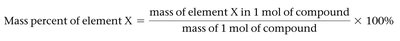
Empirical Formula: Determined from percent composition or mass analysis.
Molecular Formula: Is a whole-number multiple of the empirical formula.
Molecular Formula Equation:

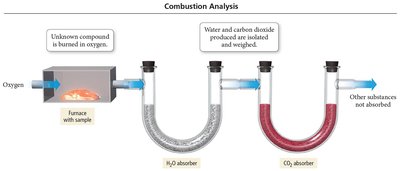
Combustion Analysis
Combustion analysis is a method used to determine the empirical formula of organic compounds by burning the sample in oxygen and measuring the amounts of water and carbon dioxide produced.
Organic Compounds and Hydrocarbons
Organic compounds are primarily composed of carbon and hydrogen, sometimes with other elements. Hydrocarbons are organic compounds containing only carbon and hydrogen, and are classified based on the types of bonds present.
Alkanes: Only single bonds (-ane suffix).
Alkenes: At least one double bond (-ene suffix).
Alkynes: At least one triple bond (-yne suffix).
Base Names: meth-, eth-, prop-, but-, pent-, hex-, hept-, oct-, non-, dec-
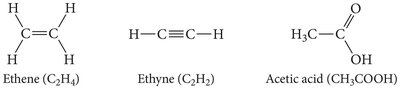
Functionalized Hydrocarbons
Functional groups are specific atoms or groups of atoms that impart characteristic chemical properties to organic compounds. Families of organic compounds are defined by their functional groups.
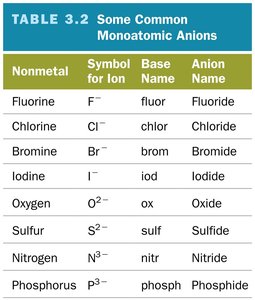
Summary Table: Common Monoatomic Anions
This table lists common monoatomic anions, their symbols, base names, and anion names.
Nonmetal | Symbol for Ion | Base Name | Anion Name |
|---|---|---|---|
Fluorine | F- | fluor | Fluoride |
Chlorine | Cl- | chlor | Chloride |
Bromine | Br- | brom | Bromide |
Iodine | I- | iod | Iodide |
Oxygen | O2- | ox | Oxide |
Sulfur | S2- | sulf | Sulfide |
Nitrogen | N3- | nitr | Nitride |
Phosphorus | P3- | phosph | Phosphide |
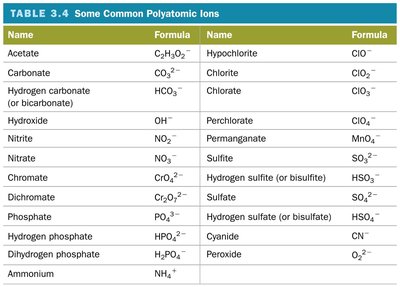
Summary Table: Common Polyatomic Ions
This table lists common polyatomic ions, their names, and formulas.
Name | Formula | Name | Formula |
|---|---|---|---|
Acetate | C2H3O2- | Hypochlorite | ClO- |
Carbonate | CO32- | Chlorite | ClO2- |
Hydrogen carbonate | HCO3- | Chlorate | ClO3- |
Hydroxide | OH- | Perchlorate | ClO4- |
Nitrite | NO2- | Permanganate | MnO4- |
Nitrate | NO3- | Sulfite | SO32- |
Chromate | CrO42- | Hydrogen sulfite | HSO3- |
Dichromate | Cr2O72- | Sulfate | SO42- |
Phosphate | PO43- | Hydrogen sulfate | HSO4- |
Hydrogen phosphate | HPO42- | Cyanide | CN- |
Dihydrogen phosphate | H2PO4- | Peroxide | O22- |
Ammonium | NH4+ |

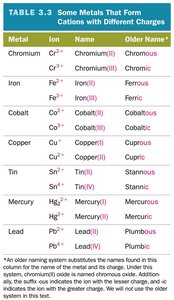
Summary Table: Metals with Variable Charges
This table lists metals that form cations with different charges, their names, and older naming conventions.
Metal | Ion | Name | Older Name |
|---|---|---|---|
Chromium | Cr2+, Cr3+ | Chromium(II), Chromium(III) | Chromous, Chromic |
Iron | Fe2+, Fe3+ | Iron(II), Iron(III) | Ferrous, Ferric |
Cobalt | Co2+, Co3+ | Cobalt(II), Cobalt(III) | Cobaltous, Cobaltic |
Copper | Cu+, Cu2+ | Copper(I), Copper(II) | Cuprous, Cupric |
Tin | Sn2+, Sn4+ | Tin(II), Tin(IV) | Stannous, Stannic |
Mercury | Hg22+, Hg2+ | Mercury(I), Mercury(II) | Mercurous, Mercuric |
Lead | Pb2+, Pb4+ | Lead(II), Lead(IV) | Plumbous, Plumbic |
 Additional info: All tables and images included are directly relevant to the explanation and reinforce the educational content. No tangential or incomplete images were used.
Additional info: All tables and images included are directly relevant to the explanation and reinforce the educational content. No tangential or incomplete images were used.
