 Back
BackChapter 3: Periodic Properties of the Elements – Structured Study Notes
Study Guide - Smart Notes

Periodic Properties of the Elements
Elemental Patterns and the Periodic Law
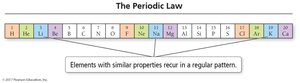
The periodic law describes the recurring patterns in the physical and chemical properties of elements, which are fundamental for predicting elemental behavior and discovering new elements. These patterns are observed when elements are arranged in order of increasing atomic number.
Periodic Property: A property that exhibits a repeating pattern across the periodic table.
Example: Density increases as you move down a column in the periodic table, as seen in the group containing boron, aluminum, gallium, and indium.
Mass-to-volume ratio: Also increases down a column, reflecting atomic structure changes.
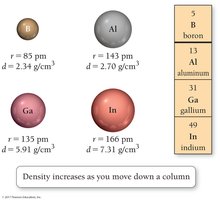
Periodic Law: When elements are arranged in order of increasing atomic number, sets of properties recur periodically.
Mendeleev’s Periodic Table and Predictions
Mendeleev organized elements by increasing mass and grouped elements with similar properties in columns. He left gaps for undiscovered elements and predicted their properties, such as eka-silicon (later discovered as germanium).
Prediction Example: Mendeleev predicted properties for gallium and germanium before their discovery, which closely matched their actual properties.

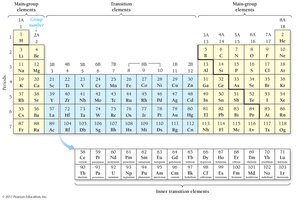
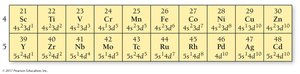
The Modern Periodic Table
The modern periodic table arranges elements by increasing atomic number. Rows are called periods, and columns are called groups or families. Elements in the same group have similar properties.
Main-group elements: Properties are predictable based on position.
Transition and inner transition elements: Properties are less predictable.
Electron Configuration and Quantum Theory
Electron Configuration: How Electrons Occupy Orbitals
Quantum-mechanical theory explains the arrangement of electrons in atoms. Electrons occupy orbitals, and their arrangement is described by the electron configuration.
Orbitals: Regions in space where electrons are likely to be found.
Electron configuration: Notation showing the distribution of electrons among orbitals.

Quantum Numbers and Electron Spin
Each electron in an atom is described by four quantum numbers: principal (n), angular momentum (l), magnetic (ml), and spin (ms). The spin quantum number (ms) can be +½ or −½, representing two possible spin orientations.
Pauli Exclusion Principle: No two electrons in an atom can have the same set of four quantum numbers.
Orbital diagrams: Visual representations using boxes and arrows to show electron spins.
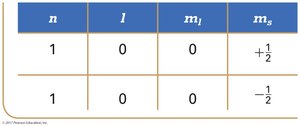
n | l | ml | ms |
|---|---|---|---|
1 | 0 | 0 | +½ |
1 | 0 | 0 | −½ |
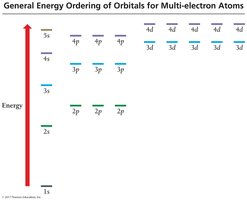
Sublevel Energy Splitting, Shielding, and Penetration
In multi-electron atoms, sublevels split in energy due to electron-electron interactions, shielding, and penetration. The order of energy is: s < p < d < f. Shielding reduces the attraction between electrons and the nucleus, while penetration describes how close an electron can get to the nucleus.
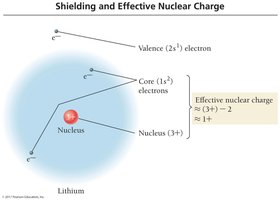
Effective Nuclear Charge (Zeff): The net positive charge experienced by an electron.
Penetration: s orbitals penetrate more than p, d, or f orbitals, leading to lower energy.
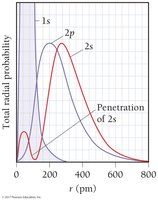
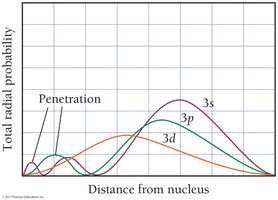
Electron Filling Principles
Electrons fill orbitals from lowest to highest energy (Aufbau principle), with no more than two electrons per orbital (Pauli exclusion principle). Hund’s rule states that electrons occupy degenerate orbitals singly before pairing.
Order of filling: 1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p
Hund’s Rule: Maximize unpaired electrons in degenerate orbitals.
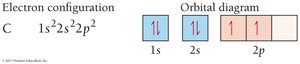
Valence Electrons and Periodic Table Organization
Core and Valence Electrons
Core electrons are in lower-energy shells, while valence electrons are in the outermost shell and participate in chemical bonding. The number of valence electrons determines an atom’s chemical behavior.
Example: For Si, 1s22s22p6 are core electrons, 3s23p2 are valence electrons.
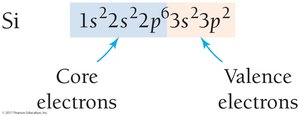
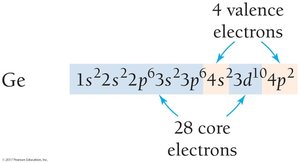
Orbital Blocks in the Periodic Table
The periodic table is divided into s, p, d, and f blocks, corresponding to the filling of quantum sublevels. The group number of a main-group element equals its number of valence electrons, and the row number equals its highest principal quantum number.
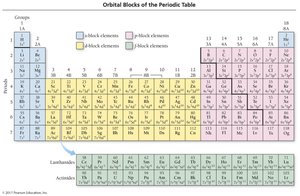
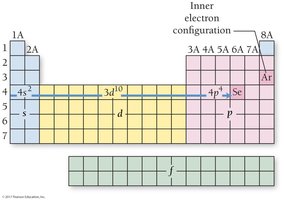
Transition Metals and Electron Configuration Anomalies
Transition and Inner Transition Metals
Transition metals (d block) and inner transition metals (f block) often have irregular electron configurations due to small energy differences between s and d orbitals. Some elements, like Cr and Cu, have experimentally determined configurations that differ from expected patterns.
Element | Expected Configuration | Experimental Configuration |
|---|---|---|
Cr | [Ar]4s23d4 | [Ar]4s13d5 |
Cu | [Ar]4s23d9 | [Ar]4s13d10 |
Mo | [Kr]5s24d4 | [Kr]5s14d5 |
Ru | [Kr]5s24d6 | [Kr]5s14d7 |
Pd | [Kr]5s24d8 | [Kr]5s04d10 |
Periodic Trends and Elemental Properties
Effective Nuclear Charge (Zeff) and Screening
Effective nuclear charge is the net positive charge attracting an electron, calculated as , where Z is the nuclear charge and S is the number of electrons in lower energy levels. Core electrons shield outer electrons efficiently, while valence electrons do not shield each other well.
Atomic Radii Trends
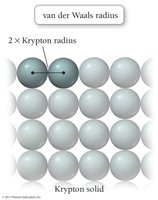
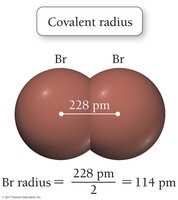
Atomic radius is the average distance from the nucleus to the outermost electrons. It decreases across a period due to increasing effective nuclear charge and increases down a group due to higher principal quantum numbers.
Van der Waals radius: Nonbonding atomic radius.
Covalent radius: Bonding atomic radius.
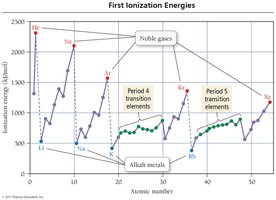
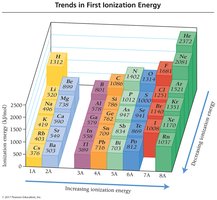
Ionization Energy Trends
Ionization energy is the minimum energy required to remove an electron from an atom in the gas phase. First ionization energy decreases down a group and increases across a period due to changes in effective nuclear charge and electron distance from the nucleus.
Equation:
Trend: Higher for elements with greater effective nuclear charge and smaller atomic radius.
Electron Affinity Trends
Electron affinity is the energy change when an electron is added to an atom in the gas phase. It generally becomes more negative across a period, with halogens having the highest electron affinities.
Equation:
Trend: More negative (exothermic) across a period; irregular down groups.
Metallic Character Trends
Metallic character describes how closely an element’s properties match those of metals. It increases down a group and decreases across a period.
Metals: Malleable, ductile, good conductors, low ionization energy.
Nonmetals: Brittle, dull, insulators, high electron affinity.
Summary Table: Key Periodic Trends
Property | Trend Across Period | Trend Down Group |
|---|---|---|
Atomic Radius | Decreases | Increases |
Ionization Energy | Increases | Decreases |
Electron Affinity | Becomes more negative | Irregular |
Metallic Character | Decreases | Increases |
