 Back
BackChapter 4: Chemical Reactions and Chemical Quantities – Study Guide
Study Guide - Smart Notes

Chapter 4: Chemical Reactions and Chemical Quantities
The Greenhouse Effect and Global Warming
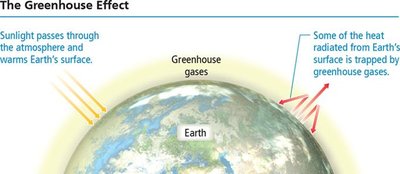
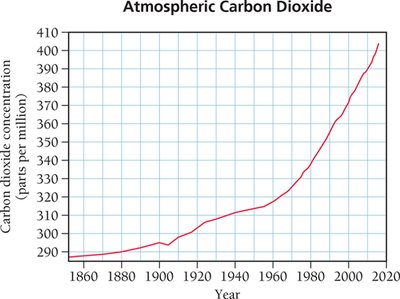
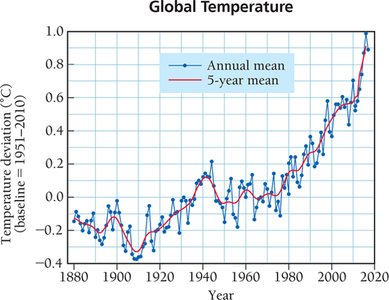
The greenhouse effect is a natural process that warms Earth's surface. Greenhouse gases allow sunlight to enter the atmosphere, warming the surface, and trap some of the heat radiated from the surface, preventing it from escaping into space. The balance between incoming and outgoing energy determines Earth's average temperature. Human activities, such as the combustion of fossil fuels, contribute to increased atmospheric CO2 levels, which are linked to global warming.
Greenhouse gases: Include CO2, methane, and water vapor.
Global warming: Refers to the observed rise in Earth's average temperature, correlated with increased CO2 levels.
Combustion reactions: Major source of anthropogenic CO2.

Chemical Reactions and Equations
A chemical reaction is a process in which substances are converted into new substances. Chemical equations are shorthand representations of these reactions, showing reactants, products, their states, and the relative quantities involved.
Reactants: Starting substances in a reaction.
Products: Substances formed as a result of the reaction.
States: Indicated by (g) for gas, (l) for liquid, (s) for solid, and (aq) for aqueous.
Abbreviation | State |
|---|---|
(g) | Gas |
(l) | Liquid |
(s) | Solid |
(aq) | Aqueous (water solution) |
Balancing Chemical Equations
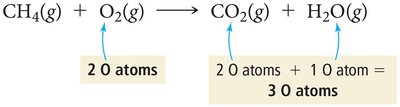
Balancing chemical equations ensures the Law of Conservation of Mass is obeyed. The number of atoms of each element must be the same on both sides of the equation. Coefficients are adjusted to achieve balance, not subscripts.
Law of Conservation of Mass: Matter is neither created nor destroyed in a chemical reaction.
Balanced equation: Equal numbers of each atom on both sides.
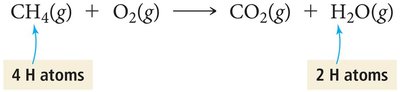
Stoichiometry: Quantitative Relationships in Reactions
Stoichiometry is the study of the numerical relationships between reactants and products in a chemical reaction. The coefficients in a balanced equation specify the relative amounts in moles of each substance.
Mole-to-mole conversions: Use the ratio from the balanced equation as a conversion factor.
Example: For the combustion of octane:
2 moles of octane react with 25 moles of oxygen to produce 16 moles of carbon dioxide and 18 moles of water.
Mass-to-Mass Conversions
To relate masses of reactants and products, use molar masses and stoichiometric ratios.
Conversion steps: Mass A → Moles A → Moles B → Mass B
Example: If 4.0 × 1015 g C8H18 is burned, calculate grams of CO2 formed using molar masses and stoichiometric ratios.
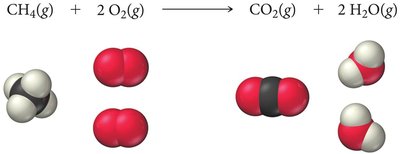
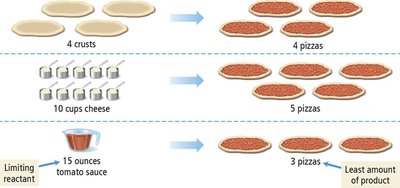
Limiting Reactant, Theoretical Yield, and Percent Yield
In reactions with multiple reactants, the limiting reactant is the one that is completely consumed first, thus limiting the amount of product formed. The theoretical yield is the maximum amount of product that can be made from the limiting reactant. The actual yield is the amount actually produced, and percent yield measures reaction efficiency.
Limiting reactant: Determines the maximum product.
Excess reactant: Not completely consumed.
Theoretical yield: Calculated from limiting reactant.
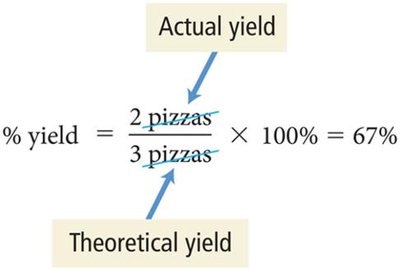
Percent yield:

Limiting Reactant in Chemical Reactions
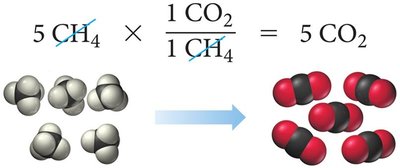
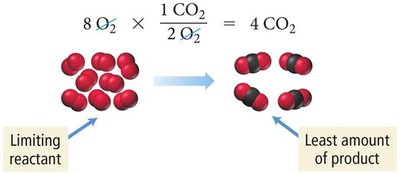
To determine the limiting reactant, calculate the amount of product each reactant can produce. The reactant that produces the least amount of product is the limiting reactant.
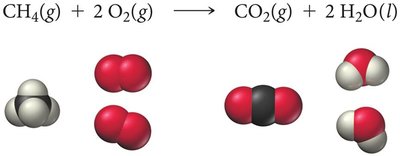
Example: Combustion of methane:
If 5 molecules of CH4 and 8 molecules of O2 are available, O2 is the limiting reactant.
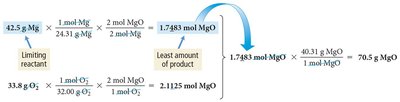
Calculating Limiting Reactant, Theoretical Yield, and Percent Yield from Masses
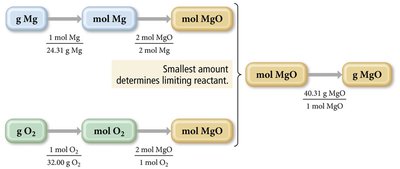
In laboratory settings, reactant quantities are often measured in grams. To find the limiting reactant and theoretical yield, convert grams to moles, use stoichiometric ratios, and convert back to grams if needed.
Example: For Mg and O2 reaction:
Convert grams of each reactant to moles, determine which produces less MgO, and calculate percent yield.
Combustion Reactions
Combustion reactions involve a substance reacting with oxygen to form oxygen-containing compounds, often including water, and releasing heat. Common examples include the combustion of methane and ethanol.
Methane combustion:
Ethanol combustion:
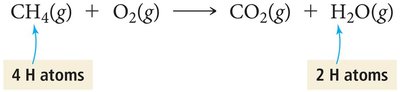
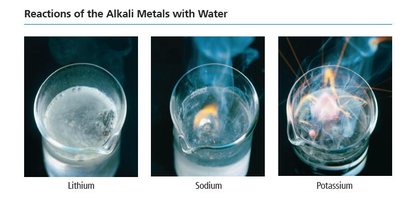
Alkali Metal and Halogen Reactions
Alkali metals react vigorously with nonmetals and water, forming ionic compounds and hydrogen gas. Halogens react with metals to form metal halides, with hydrogen to form hydrogen halides, and with each other to form interhalogen compounds.
Alkali metal + water:
Halogen + metal:
Halogen + hydrogen:
Summary Table: Key Concepts in Chemical Reactions
Concept | Definition | Example |
|---|---|---|
Chemical Reaction | Process converting substances into new ones | Combustion of methane |
Stoichiometry | Quantitative relationships in reactions | Mole-to-mole conversions |
Limiting Reactant | Reactant that determines maximum product | O2 in methane combustion |
Theoretical Yield | Maximum product from limiting reactant | Calculated from stoichiometry |
Percent Yield | Actual yield divided by theoretical yield | Efficiency calculation |
