 Back
BackChapter 4: Molecules and Compounds – Structure, Bonding, and Composition
Study Guide - Smart Notes

Molecules and Compounds
Introduction to Molecules and Compounds
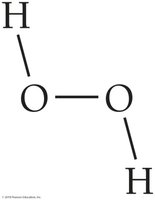
Molecules are formed when two or more elements combine chemically. These elements may be the same or different, resulting in either elemental molecules (e.g., O2) or molecular compounds (e.g., H2O). The diversity of substances in nature arises from the ability of elements to form a wide variety of compounds.
Molecule: A group of two or more atoms held together by chemical bonds.
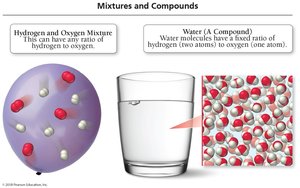
Compound: A substance composed of two or more different elements combined in a fixed ratio.
Law of Definite Proportion
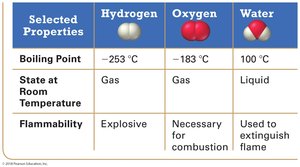
The Law of Definite Proportion states that a chemical compound always contains the same proportion of elements by mass. For example, water (H2O) always consists of two hydrogen atoms for every one oxygen atom, regardless of the sample size or source.
Chemical Bonds
Types of Chemical Bonds
Chemical bonds are the forces that hold atoms together in compounds. They result from the attractions between charged particles (electrons and protons) in atoms. There are three main types of chemical bonds:
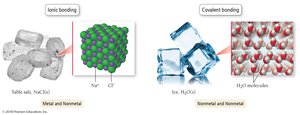
Ionic Bonds: Formed by the transfer of electrons from a metal to a nonmetal.
Covalent Bonds: Formed by the sharing of electrons between nonmetals.
Metallic Bonds: Involve the pooling of electrons among a lattice of metal atoms (not covered in detail here).
Ionic Bonds and Ionic Compounds
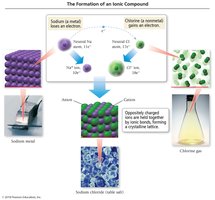
Ionic bonds occur between metals and nonmetals, involving the transfer of electrons. The resulting oppositely charged ions (cations and anions) are held together by electrostatic forces, forming an ionic compound. In the solid phase, these compounds form a crystal lattice structure.
Cation: A positively charged ion (typically a metal that has lost electrons).
Anion: A negatively charged ion (typically a nonmetal that has gained electrons).
Covalent Bonds and Molecular Compounds
Covalent bonds occur between nonmetal atoms and involve the sharing of electrons. The atoms held together by covalent bonds form molecules, which are the basic units of molecular compounds.
Representing Compounds
Chemical Formulas
Chemical formulas indicate the types and numbers of atoms in a compound. There are three main types:
Empirical Formula: Shows the simplest whole-number ratio of elements in a compound.
Molecular Formula: Shows the actual number of each type of atom in a molecule.
Structural Formula: Shows how atoms are bonded together in a molecule.
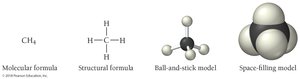
Molecular Models
Molecular models provide three-dimensional representations of molecules. Common types include:
Ball-and-stick model: Atoms are balls, bonds are sticks.
Space-filling model: Atoms are shown as spheres that fill the space between them.

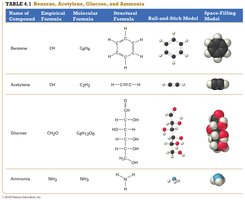
Lewis Structures and the Octet Rule
Lewis Structures
The Lewis model represents valence electrons as dots around the chemical symbol. Lewis structures are used to illustrate the arrangement of valence electrons in molecules and ions, focusing on the transfer or sharing of electrons during bonding.

The Octet Rule
Atoms tend to gain, lose, or share electrons to achieve a noble gas configuration (eight valence electrons, or an octet). There are exceptions, such as hydrogen (duet rule), and elements in period 3 or below, which can have expanded octets.

Lewis Structures for Ionic Compounds
Lewis symbols can be used to represent the transfer of electrons in ionic compounds. For example, calcium loses two electrons to form Ca2+, and each chlorine atom gains one electron to form Cl-, resulting in CaCl2.
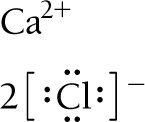
Ionic Bonding: Properties and Energetics
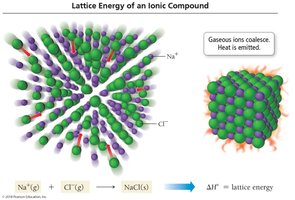
Lattice Energy
Lattice energy is the energy released when gaseous ions form an ionic solid. It is always exothermic and depends on the charges and sizes of the ions. The crystal lattice maximizes the attraction between cations and anions, resulting in a stable structure.
Electrical Conductivity of Ionic Compounds
Ionic solids do not conduct electricity because their ions are fixed in place. However, when melted or dissolved in water, the ions are free to move, allowing the compound to conduct electricity.


Writing and Naming Ionic Compounds
Formulas of Ionic Compounds
The formula of an ionic compound reflects the smallest whole-number ratio of cations to anions that results in electrical neutrality. For example, Ca2+ and O2- combine to form CaO.
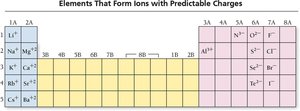
Naming Ionic Compounds
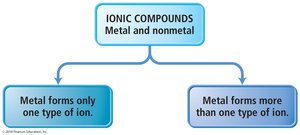
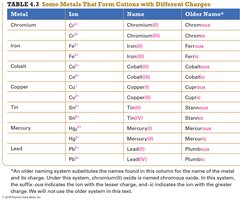
Ionic compounds are named based on the cation and anion present. If the metal forms only one type of ion (Type I), the name is simply the cation followed by the anion with an -ide ending. If the metal forms more than one type of ion (Type II), the charge is specified with a Roman numeral.


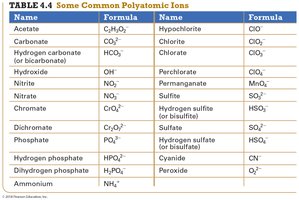
Polyatomic Ions
Many ionic compounds contain polyatomic ions, which are ions composed of two or more atoms covalently bonded. Common polyatomic ions include sulfate (SO42-), nitrate (NO3-), and ammonium (NH4+).
Covalent Bonding and Molecular Compounds
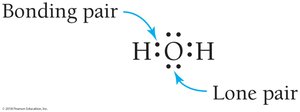
Bonding and Lone Pairs
In covalent bonds, electrons are shared between atoms. Shared electrons are called bonding pairs, while non-shared electrons are called lone pairs.
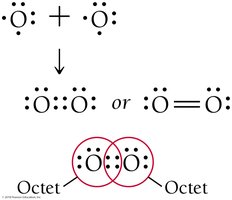
Single, Double, and Triple Bonds
Atoms can share one, two, or three pairs of electrons, resulting in single, double, or triple covalent bonds, respectively. For example, O2 has a double bond, and N2 has a triple bond.
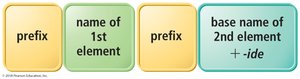
Naming Molecular Compounds
Rules for Naming Binary Molecular Compounds
Molecular compounds are named using prefixes to indicate the number of each atom. The first element is named first, followed by the second element with an -ide ending. The prefix mono- is omitted for the first element if only one atom is present.
Formula Mass and Percent Composition
Calculating Formula Mass
The formula mass (or molecular mass) of a compound is the sum of the atomic masses of all atoms in its chemical formula.
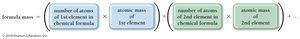
Percent Composition
The percent composition of an element in a compound is calculated as:
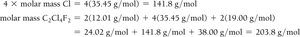
Empirical and Molecular Formulas
Determining Empirical Formulas
The empirical formula gives the simplest whole-number ratio of atoms in a compound. It can be determined from percent composition or combustion analysis data.
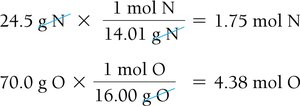
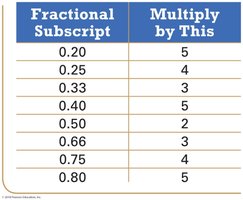
Determining Molecular Formulas
The molecular formula is a whole-number multiple of the empirical formula. It is determined by dividing the molar mass of the compound by the empirical formula mass:
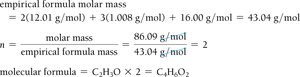
Combustion Analysis
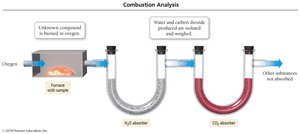
Combustion Analysis Technique
Combustion analysis is used to determine the empirical formula of compounds containing C, H, and O. The compound is burned in oxygen, and the masses of CO2 and H2O produced are measured to calculate the amounts of C and H in the original sample.
Organic Compounds
Introduction to Organic Compounds
Organic compounds are primarily composed of carbon and hydrogen, often with oxygen, nitrogen, phosphorus, or sulfur. Carbon's ability to form four covalent bonds and create chains and rings makes organic chemistry a vast and diverse field.
