 Back
BackChapter 4: Reactions in Aqueous Solution – Study Notes
Study Guide - Smart Notes

Reactions in Aqueous Solution
4.1 Molarity and Solution Calculations
Molarity (M) is a measure of the concentration of a solution, defined as the number of moles of solute per liter of solution. Calculating molarity and related quantities is fundamental for preparing solutions and performing stoichiometric calculations in aqueous chemistry.
Molarity formula:
Calculating moles from molarity and volume:
Calculating mass of solute:
Example: To prepare 0.500 L of 1.00 M NaCl solution, dissolve 0.500 mol NaCl (29.2 g) in enough water to make 0.500 L.
4.2 Dilution of Solutions
When a solution is diluted, the amount of solute remains constant, but the volume increases, lowering the concentration. The dilution equation relates the initial and final concentrations and volumes:
Dilution equation:
Example: To make 250 mL of 0.100 M HCl from 1.00 M HCl, use L = 25.0 mL.
4.3 Electrolytes and Nonelectrolytes
Substances dissolved in water can be classified based on their ability to conduct electricity:
Strong electrolytes: Completely dissociate into ions (e.g., NaCl, HCl).
Weak electrolytes: Partially dissociate (e.g., CH3COOH, NH3).
Nonelectrolytes: Do not dissociate (e.g., sugar, ethanol).
Example: NaCl is a strong electrolyte; C6H12O6 (glucose) is a nonelectrolyte.
4.4 Concentration of Ions in Solution
Strong electrolytes dissociate completely, so the concentration of each ion can be calculated from the formula and the solution's molarity.
Example: 0.20 M Na2SO4 yields 0.40 M Na+ and 0.20 M SO42−.
4.5 Types of Aqueous Reactions
Reactions in aqueous solution are classified as:
Precipitation reactions: Formation of an insoluble solid (precipitate).
Acid-base (neutralization) reactions: Reaction of acid and base to form water and a salt.
Redox reactions: Transfer of electrons between species.
4.6 Writing Ionic Equations
Chemical equations for aqueous reactions can be written in three forms:
Molecular equation: All species written as compounds.
Total ionic equation: Strong electrolytes written as ions.
Net ionic equation: Only species undergoing change are shown.
Example: For NaCl(aq) + AgNO3(aq) → AgCl(s) + NaNO3(aq): Net ionic: Ag+(aq) + Cl−(aq) → AgCl(s)
4.7 Solubility Guidelines
Solubility rules help predict whether an ionic compound will dissolve in water or form a precipitate. The following table summarizes common solubility guidelines and exceptions:

4.8 Predicting Precipitation Reactions
To predict if a precipitation reaction occurs, use solubility guidelines to determine if any product is insoluble. If so, a precipitate forms.
Example: Mixing solutions of BaCl2 and Na2SO4 forms BaSO4(s) (insoluble).
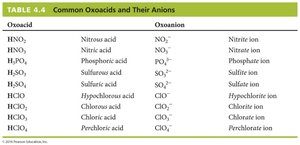
4.9 Oxoacids: Names and Formulas
Oxoacids are acids containing hydrogen, oxygen, and another element. The following table lists common oxoacids and their corresponding anions:
4.10 Arrhenius Theory of Acids and Bases
According to Arrhenius theory:
Acids produce H+ ions in water.
Bases produce OH− ions in water.
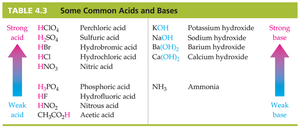
Acids and bases are classified as strong or weak based on their degree of dissociation in water. The following table summarizes common acids and bases and their relative strengths:
4.11 Acid-Base Neutralization Equations
In a neutralization reaction, an acid reacts with a base to produce water and a salt. The net ionic equation for a strong acid and strong base is:
4.12 Molarity in Stoichiometry and Titrations
Molarity is used to relate moles and volume in stoichiometric calculations, especially in titrations, where the concentration of an unknown solution is determined by reacting it with a solution of known concentration.
Titration formula: (for 1:1 stoichiometry)
4.13 Oxidation Numbers and Redox Reactions
Assigning oxidation numbers helps identify redox reactions, the species oxidized/reduced, and the oxidizing/reducing agents.
Oxidation: Increase in oxidation number (loss of electrons).
Reduction: Decrease in oxidation number (gain of electrons).
Example: In Zn + CuSO4 → ZnSO4 + Cu, Zn is oxidized, Cu2+ is reduced.
4.14 Predicting Redox Reactions
The activity series and periodic table position help predict if a redox reaction will occur. A more active metal will reduce the ion of a less active metal.
4.15 Redox Titrations
Redox titrations are used to determine the concentration of an oxidizing or reducing agent in solution by reacting it with a titrant of known concentration until the reaction is complete.
Example: Titrating Fe2+ with KMnO4 to determine Fe2+ concentration.
