Back
BackChapter 4: Reactions in Aqueous Solution – Study Notes
Study Guide - Smart Notes

Reactions in Aqueous Solution
Introduction to Solutions
Solutions are homogeneous mixtures composed of two or more pure substances. The component present in the greatest amount is called the solvent, while the other components are solutes. When water is the solvent, the solution is termed an aqueous solution.
Types of Solutes in Water
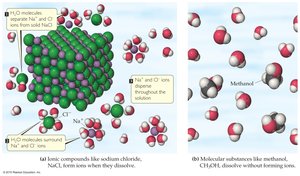
Ionic Compounds dissolve by dissociation, where water molecules surround and separate the ions.
Molecular Compounds may dissolve without dissociation, interacting with water but not forming ions.
Some molecular substances react with water upon dissolving, forming ions.
Electrolytes and Nonelectrolytes
An electrolyte is a substance that dissociates into ions in water, allowing the solution to conduct electricity. A nonelectrolyte dissolves without forming ions and does not conduct electricity.

Classification of Electrolytes
Strong Electrolytes: Completely dissociate in water (e.g., most ionic salts, strong acids, strong bases).
Weak Electrolytes: Partially dissociate in water (e.g., weak acids, weak bases).
Nonelectrolytes: Do not dissociate in water (e.g., most molecular compounds like sugar).
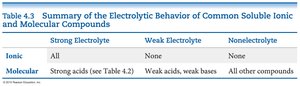
Strong Electrolyte | Weak Electrolyte | Nonelectrolyte | |
|---|---|---|---|
Ionic | All | None | None |
Molecular | Strong acids | Weak acids, weak bases | All other compounds |
Solubility of Ionic Compounds
Not all ionic compounds are soluble in water. Solubility rules help predict whether a compound will dissolve. These rules are based on the ions present in the compound.
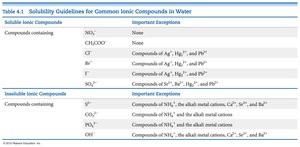
Soluble Ionic Compounds | Important Exceptions |
|---|---|
NO3-, CH3COO- | None |
Cl-, Br-, I- | Ag+, Hg22+, Pb2+ |
SO42- | Sr2+, Ba2+, Hg22+, Pb2+ |
Insoluble Ionic Compounds | Important Exceptions |
S2-, CO32-, PO43-, OH- | NH4+, alkali metal cations, Ca2+, Sr2+, Ba2+ |
Precipitation Reactions
When two solutions containing soluble salts are mixed, an insoluble salt (precipitate) may form. This is a precipitation reaction.
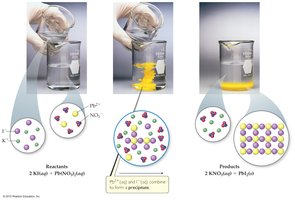
Metathesis (Exchange) Reactions
In metathesis reactions, ions in the reactants exchange partners to form new compounds. The general form is:
Example:
Steps to Complete and Balance Metathesis Equations
Identify ions present in reactants.
Write formulas for possible products by exchanging ions.
Use solubility rules to determine if a precipitate forms.
Balance the equation.
Writing Ionic Equations
Molecular Equation: Shows reactants and products as compounds.
Complete Ionic Equation: Shows all strong electrolytes as ions.
Net Ionic Equation: Shows only the species that actually change during the reaction (removes spectator ions).
Example (Net Ionic Equation for CaCl2 + Na2CO3):
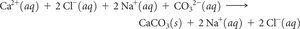
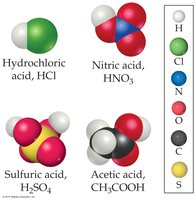
Acids and Bases
Acids are substances that increase the concentration of H+ in water (Arrhenius) or donate protons (Brønsted-Lowry). Bases increase OH- concentration or accept protons.

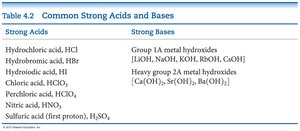
Strong and Weak Acids/Bases
Strong acids/bases dissociate completely in water.
Weak acids/bases only partially dissociate.
Strong Acids | Strong Bases |
|---|---|
HCl, HBr, HI, HNO3, HClO4, H2SO4 | Group 1A and heavy Group 2A metal hydroxides |
Acid-Base Reactions (Neutralization)
In a neutralization reaction, an acid reacts with a base to produce water and a salt. The net ionic equation for a strong acid and strong base is:
Gas-Forming Reactions
Some metathesis reactions produce a gas, such as CO2, H2S, or SO2. For example:
Oxidation-Reduction (Redox) Reactions
Oxidation is the loss of electrons; reduction is the gain of electrons. These processes always occur together in redox reactions.
Assign oxidation numbers to determine if redox has occurred.
Rules: Elements in elemental form = 0; monatomic ion = charge; sum in compound = 0; sum in polyatomic ion = ion charge.
Displacement and Activity Series
In displacement reactions, a more active element displaces a less active one from a compound. The activity series ranks metals by their tendency to be oxidized.

Concentration of Solutions: Molarity
Molarity (M) is defined as the number of moles of solute per liter of solution:

Calculating Molarity
To calculate molarity, convert grams of solute to moles and volume to liters, then use the formula above.
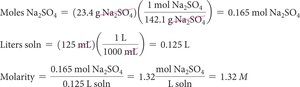
Calculating Ion Concentrations
Use the chemical formula to determine the ratio of ions produced per formula unit dissolved.
Using Molarity in Calculations
To find the amount of solute in a given volume of solution:
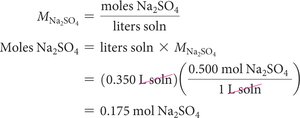
Dilution of Solutions
To dilute a solution, add solvent to decrease concentration. The relationship is:
where c = concentrated, d = dilute.

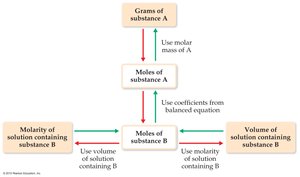
Stoichiometry in Solution Reactions
Stoichiometric calculations in solution use molarity and volume to relate reactants and products. The general approach is:
Calculate moles of known substance using molarity and volume.
Use balanced equation to relate moles of reactants and products.
Convert moles to grams or volume as needed.
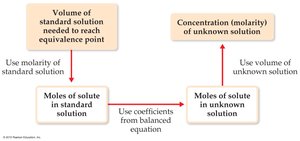
Titration
Titration is an analytical technique to determine the concentration of a solute. A solution of known concentration (standard solution) is added to react completely with the analyte. The point at which the reaction is complete is the equivalence point.