 Back
BackChapter 4: Reactions in Aqueous Solutions – Concentration, Dilution, and Titration
Study Guide - Smart Notes

Reactions in Aqueous Solutions
Terminology and Key Concepts
This section introduces fundamental terms and concepts related to solutions and their reactions in aqueous environments. Understanding these terms is essential for mastering solution chemistry and its applications in laboratory and analytical contexts.
Concentration: The amount of solute dissolved in a given quantity of solvent or solution.
Molarity (M): A measure of concentration, defined as the number of moles of solute per liter of solution.
Dilution: The process of reducing the concentration of a solution by adding more solvent.
Equivalence Point: The stage in a titration where stoichiometrically equivalent quantities of reactants are combined.
Concentrations of Solutions
Concentration quantifies how much solute is present in a solution. Molarity is the most common unit used in chemistry for this purpose. Solutions can be prepared with varying concentrations by adjusting the amount of solute added to the solvent.
Molarity (M): Defined as moles of solute per liter of solution.
Formula:

Conversions: Grams to moles (using molar mass), moles to molarity, and vice versa.
Key Equations:

Worked Examples: Calculating Molarity
Applying the above formulas, we can solve typical concentration problems encountered in laboratory settings.
Example 1: Calculate the molarity of a solution prepared by dissolving 10.5 g of NaF in enough water to make 250 mL of solution (MMNaF = 42 g/mol).
Solution:
Convert grams to moles:
Convert mL to L:
Calculate molarity:
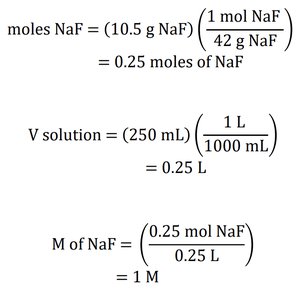
Example 2: How many moles of NaF are in 50 mL of 0.5 M NaF solution?
Solution:
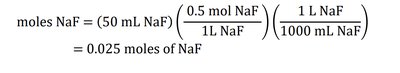
Example 3: How many grams of NaF are in 50 mL of 0.5 M NaF solution?
Solution:
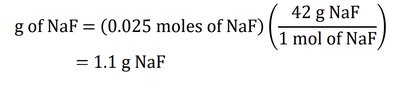
Example 4: How many mL of 0.5 M NaF are needed to get 0.01 mol of NaF?
Solution:
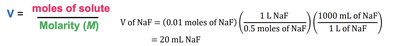
Concentration of Electrolyte Ions
When ionic compounds dissolve, the concentration of each ion depends on the compound's formula. This is crucial for understanding solution behavior and stoichiometry.
Example: 1.0 M NaCl yields 1.0 M Na+ and 1.0 M Cl- ions.
Example: 1.0 M Na2SO4 yields 2.0 M Na+ and 1.0 M SO42- ions.

Dilution of Solutions
Dilution is a common laboratory technique for preparing solutions of lower concentration from a stock solution. The number of moles of solute remains constant during dilution.
Key Equation:
Application: Used to calculate the volume of stock solution needed or the final concentration after dilution.

Example: How many mL of 4.0 M AgNO3 are needed to make 320 mL of 0.2 M AgNO3?
Solution:
Calculate moles in dilute solution:
Calculate volume of concentrated solution:

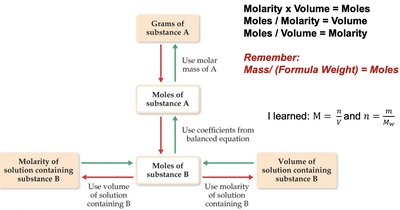
Solution Stoichiometry and Chemical Analysis
Stoichiometry in solution chemistry involves converting laboratory measurements to moles, using balanced equations to relate reactants and products, and then converting back to desired units. This process is essential for quantitative chemical analysis.
Steps:
Convert grams to moles using molar mass.
Convert volume or molarity to moles using .
Use stoichiometric coefficients from the balanced equation to relate reactants and products.
Convert moles back to grams, molarity, or volume as needed.
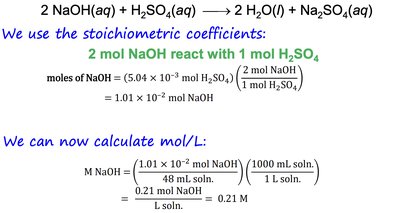
Example: How many grams of NaOH are needed to neutralize 20 mL of 0.150 M H2SO4?
Solution:
Balanced equation:
Convert volume to moles:
Use stoichiometry: reacts with
Calculate grams:

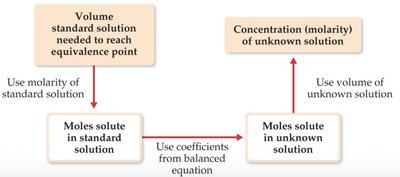
Titrations
Titration is an analytical technique used to determine the concentration of a solution by reacting it with a standard solution of known concentration. The equivalence point is reached when stoichiometrically equivalent amounts of reactants have reacted.
Types of Titrations: Neutralization, precipitation, and oxidation-reduction reactions.
Equivalence Point: The point at which the reaction is complete, often indicated by a color change.
Key Equation:

Example: What is the molarity of a NaOH solution if 48.0 mL neutralizes 35.0 mL of 0.144 M H2SO4?
Solution:
Convert volume to moles:
Use stoichiometry: reacts with
Calculate moles NaOH:
Calculate molarity:

Process Flow: Use the volume and molarity of the standard solution to determine moles, apply stoichiometry, and calculate the unknown concentration.

Summary Table: Solution Calculations
The following table summarizes key equations and their applications in solution chemistry:
Calculation | Equation (LaTeX) | Application |
|---|---|---|
Molarity | Concentration of solute | |
Grams to moles | Mass to moles conversion | |
Dilution | Preparing dilute solutions | |
Titration | Finding equivalence point |
