 Back
BackChapter 5: Chemical Bonding I – Lewis Structures, Molecular Shapes, and Bond Properties
Study Guide - Smart Notes

Chemical Bonding I: Lewis Structures and Molecular Shapes
Introduction to Chemical Bonding
Chemical bonding is fundamental to understanding how atoms combine to form molecules and compounds. The shape and structure of molecules influence their physical and chemical properties, including reactivity, polarity, and biological activity.
Bonding Theories and Molecular Structure
Lewis Dot Model and Lewis Structural Model
The Lewis dot model is a simple representation of atoms and their valence electrons. It helps predict how atoms bond and the resulting molecular shapes. The Lewis structural model expands on this by showing bonds and lone pairs explicitly.
Valence Shell Electron Pair Repulsion (VSEPR) Theory: Predicts the 3D shape of molecules based on repulsion between electron groups.
Bonding Theories: Explain why certain combinations of atoms are stable and others are not.
Applications: Used to predict molecular shapes, chemical properties, and physical properties.
Example: Lewis Dot Diagram for HF
Hydrogen fluoride (HF) is a diatomic molecule, meaning it consists of two atoms. The Lewis dot diagram shows the shared pair of electrons between H and F.

Electronegativity and Bond Polarity
Definition and Trends
Electronegativity is the ability of an atom to attract bonding electrons to itself. It increases across a period (left to right) and decreases down a group (top to bottom) in the periodic table. Fluorine is the most electronegative element, while francium is the least.
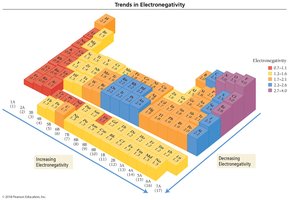
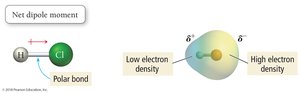
Bond Polarity and Electronegativity Difference
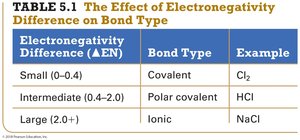
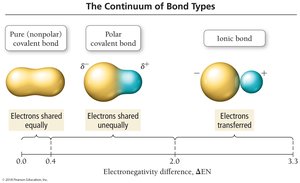
The difference in electronegativity between two bonded atoms determines the bond type:
Pure Covalent (Nonpolar): Electrons are shared equally (ΔEN = 0).
Polar Covalent: Electrons are shared unequally (ΔEN = 0.1–1.9).
Ionic: Electrons are transferred (ΔEN ≥ 2.0).
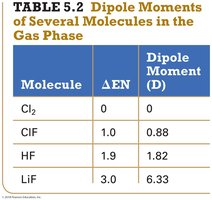
Bond Dipole Moments
A dipole moment (μ) measures bond polarity and is calculated as:
where q is the magnitude of the partial charges and r is the distance between them. Dipole moments are measured in Debyes (D).
Writing Lewis Structures for Molecular Compounds
Steps for Drawing Lewis Structures
Lewis structures are drawn by following these steps:
Write the correct skeletal structure: The less electronegative atom is usually the central atom; hydrogen is always terminal.
Calculate the total number of valence electrons: Sum the valence electrons from each atom and account for any charges.
Distribute electrons: Place bonding pairs first, then lone pairs to complete octets (or duets for hydrogen).
Form double or triple bonds: If any atom lacks an octet, create multiple bonds as needed.
Check: Ensure the total number of electrons matches the calculated sum.
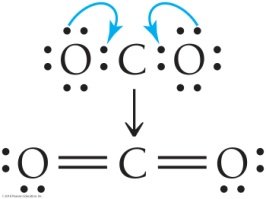
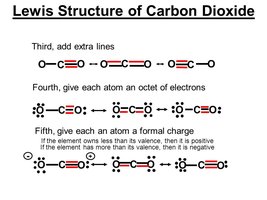
Example: Lewis Structure for CO2
Step 1: Carbon is the central atom. Step 2: Calculate total valence electrons.

Step 3: Distribute electrons and form double bonds to satisfy octets.
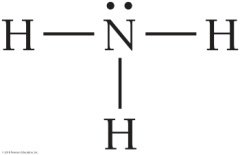
Example: Lewis Structure for NH3
Step 1: Nitrogen is the central atom. Step 2: Calculate total valence electrons.

Step 3: Distribute electrons; all atoms have octets or duets.
Resonance and Formal Charge
Resonance Structures
Resonance occurs when two or more valid Lewis structures can be drawn for the same compound, differing only in electron positions. The most stable resonance contributor has the maximum number of atoms with full octets and minimal charge separation.

Formal Charge
Formal charge is an electron bookkeeping system used to evaluate alternative Lewis structures. The formula is:
Structures with fewer and smaller formal charges are preferred, and negative formal charges should reside on the more electronegative atom.
Exceptions to the Octet Rule
Odd-Electron Species, Incomplete Octets, and Expanded Octets
Odd-electron species: Molecules with an odd number of electrons (free radicals).
Incomplete octets: Atoms like hydrogen and boron may have fewer than eight electrons.
Expanded octets: Atoms in the third period or below can have more than eight electrons when they are the central atom.
Bond Energies and Bond Lengths
Bond Energies
The bond energy is the energy required to break one mole of a bond in the gaseous state. In general, the more electrons two atoms share, the stronger and shorter the bond.
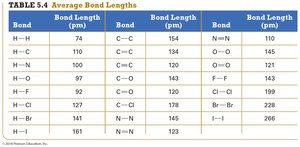
Bond Lengths
Bond length is the distance between the nuclei of bonded atoms. It decreases as the number of shared electrons increases and varies with atomic size.
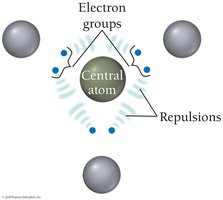
VSEPR Theory and Molecular Geometry
VSEPR Theory
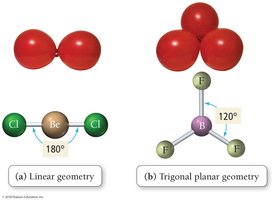
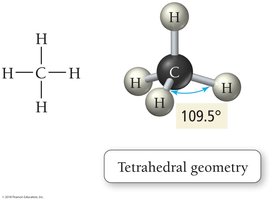
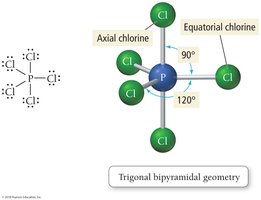
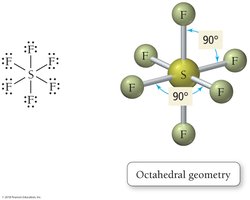
VSEPR theory states that electron groups around a central atom will be most stable when they are as far apart as possible, determining the geometric arrangement and bond angles.
Electron Group Arrangements
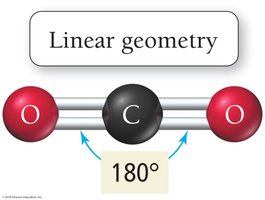
Linear: 2 electron groups, bond angle 180°
Trigonal planar: 3 electron groups, bond angle 120°
Tetrahedral: 4 electron groups, bond angle 109.5°
Trigonal bipyramidal: 5 electron groups, bond angles 90° and 120°
Octahedral: 6 electron groups, bond angle 90°
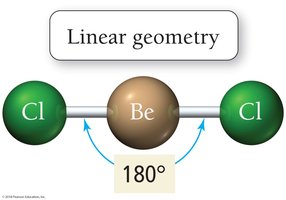

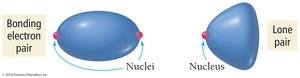
Effect of Lone Pairs on Molecular Structure
Lone pairs occupy more space on the central atom than bonding pairs, causing bond angles to decrease. The repulsive force interactions are ranked as:
Lone pair–Lone pair > Lone pair–Bonding pair > Bonding pair–Bonding pair
Molecular Shape and Polarity
Polarity and Dipole Moments
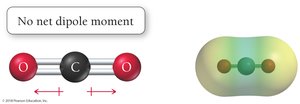
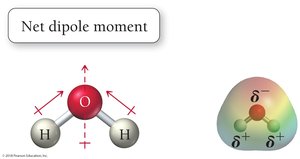
A molecule is polar if it contains polar bonds (due to electronegativity differences) and has an unsymmetrical shape so that dipole moments do not cancel out. Polarity affects intermolecular forces, boiling points, and solubility.
Examples of Molecular Polarity
HCl: Polar molecule due to bond dipole moment.
CO2: Nonpolar molecule; dipole moments cancel due to linear geometry.
H2O: Polar molecule; dipole moments do not cancel due to bent geometry.
Summary Table: Electronegativity Difference and Bond Type
Electronegativity Difference (ΔEN) | Bond Type | Example |
|---|---|---|
Small (0–0.4) | Covalent | Cl2 |
Intermediate (0.4–2.0) | Polar covalent | HCl |
Large (2.0+) | Ionic | NaCl |
Summary Table: Dipole Moments of Molecules
Molecule | ΔEN | Dipole Moment (D) |
|---|---|---|
Cl2 | 0 | 0 |
ClF | 1.0 | 0.88 |
HF | 1.9 | 1.82 |
LiF | 3.0 | 6.33 |
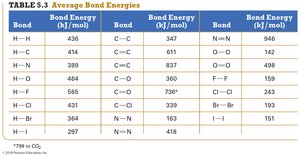
Summary Table: Average Bond Energies
Bond | Bond Energy (kJ/mol) |
|---|---|
H–H | 436 |
C–C | 347 |
C≡C | 837 |
H–O | 464 |
H–Cl | 431 |
Br–Br | 193 |
N≡N | 946 |
Summary Table: Average Bond Lengths
Bond | Bond Length (pm) |
|---|---|
H–H | 74 |
C–C | 154 |
C≡C | 120 |
H–O | 97 |
H–Cl | 127 |
Br–Br | 228 |
N≡N | 110 |
Additional info: These notes cover the essential concepts of chemical bonding, Lewis structures, molecular geometry, bond energies, and polarity, as outlined in Chapter 5 of a general chemistry course.
