 Back
Back5 PP
Study Guide - Smart Notes

Solutions and Solution Concentration
Definitions and Types of Solutions
Solutions are homogeneous mixtures composed of a solvent (majority component) and a solute (minority component). When water is the solvent, the solution is called an aqueous solution.
Solvent: The substance present in the largest amount in a solution.
Solute: The substance present in a smaller amount, dissolved in the solvent.
Homogeneous mixture: Uniform composition throughout; also called a solution.
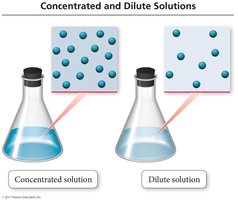
Concentrated vs. Dilute Solutions
Solutions are described as concentrated or dilute based on the amount of solute relative to solvent.
Concentrated solution: Contains a large amount of solute.
Dilute solution: Contains a small amount of solute.
Quantitative Description of Solution Concentration
Molarity (M)
Molarity is the most common unit for expressing solution concentration in chemistry. It is defined as the number of moles of solute per liter of solution.
Formula:
Used as a conversion factor between moles and volume.
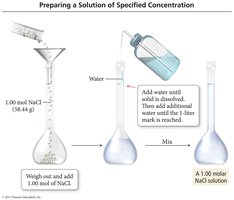
Preparing a Molar Solution
To prepare a solution of specified molarity, weigh the required amount of solute, dissolve it in water, and dilute to the desired volume.
Using Molarity in Calculations
Molarity allows conversion between moles and liters of solution:
To find moles from volume:
To find volume from moles:


Solution Stoichiometry and Dilution
Dilution of Solutions
Stock solutions are often diluted to prepare solutions of lower concentration. The amount of solute remains constant during dilution.
Dilution equation:
and are the concentration and volume of the stock solution; and are those of the diluted solution.
Electrolytes and Nonelectrolytes
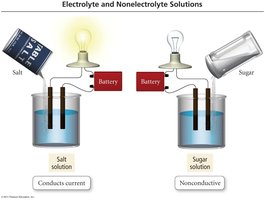
Conductivity of Solutions
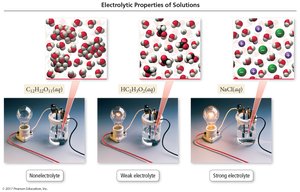
Electrolytes are substances that dissolve in water to produce a solution that conducts electricity, while nonelectrolytes do not.
Electrolyte: Dissolves to form ions; conducts electricity.
Nonelectrolyte: Dissolves as molecules; does not conduct electricity.
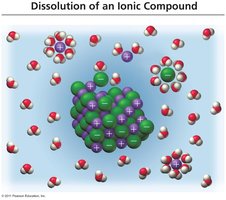
Dissolution of Ionic and Molecular Compounds
Ionic compounds dissociate into ions when dissolved in water, while molecular compounds typically do not.
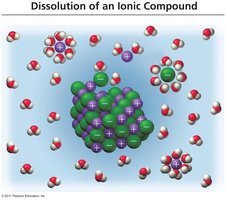
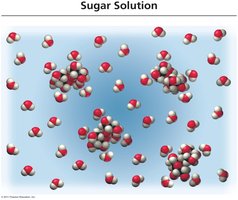
Strong and Weak Electrolytes
Electrolytes are classified as strong or weak based on the extent of ionization.
Strong electrolytes: Completely dissociate into ions (e.g., NaCl, strong acids).
Weak electrolytes: Partially dissociate; most molecules remain intact (e.g., acetic acid).
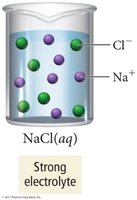
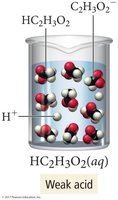
Solubility of Ionic Compounds
Soluble vs. Insoluble Compounds
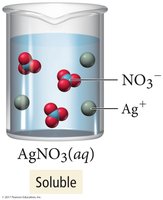
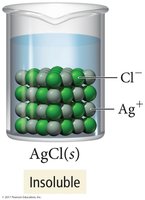
Solubility describes whether a compound dissolves in water. Soluble compounds form aqueous solutions; insoluble compounds remain as solids.
Soluble: Dissolves in water (e.g., NaCl, AgNO3).
Insoluble: Does not dissolve (e.g., AgCl).
Solubility Rules
Solubility rules help predict whether an ionic compound will dissolve in water.
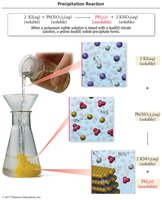
Precipitation Reactions
Formation of Precipitates
Precipitation reactions occur when two aqueous solutions combine to form an insoluble product (precipitate).
Double displacement: Exchange of ions between reactants.
Precipitate: The insoluble product formed.
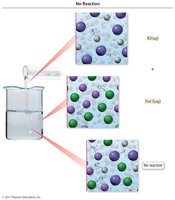
No Reaction Scenario
If no insoluble product forms, no precipitation reaction occurs.
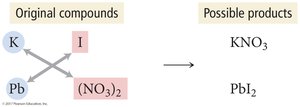
Predicting Precipitation Reactions
Write formulas of possible products by exchanging ions.
Determine solubility using solubility rules.
If a product is insoluble, it precipitates; otherwise, write "no reaction."
Balance the equation.
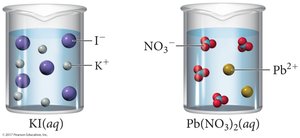
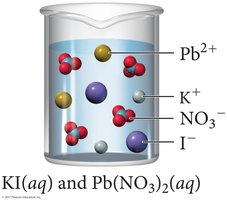
Molecular, Complete Ionic, and Net Ionic Equations
Types of Equations
Molecular equation: Shows all reactants and products as compounds.
Complete ionic equation: Shows all strong electrolytes as ions.
Net ionic equation: Shows only the ions and molecules directly involved in the reaction; spectator ions are omitted.

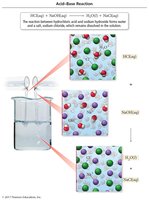
Acid-Base Reactions
Neutralization Reactions
Acid-base reactions involve the neutralization of an acid by a base, producing a salt and water.
General net ionic equation:
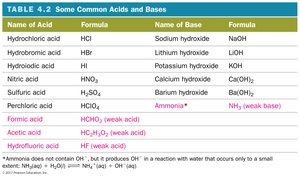
Common Acids and Bases
Acids ionize to produce H+ ions; bases dissociate to produce OH- ions.
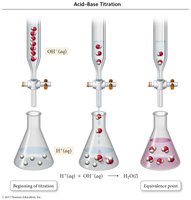
Acid-Base Titration
Titration is a technique to determine the concentration of an unknown solution by reacting it with a solution of known concentration until the endpoint is reached.
Burette: Instrument used to add titrant.
Indicator: Detects the endpoint.
Gas-Evolving Reactions
Formation of Gases
Some reactions produce a gas either directly or by decomposition of an intermediate product.
Example:

Oxidation-Reduction (Redox) Reactions
Electron Transfer and Oxidation States
Redox reactions involve the transfer of electrons between reactants. Oxidation is the loss of electrons; reduction is the gain of electrons.
Oxidation state: Imaginary charge assigned to atoms based on rules.
Oxidizing agent: Causes oxidation; is reduced.
Reducing agent: Causes reduction; is oxidized.

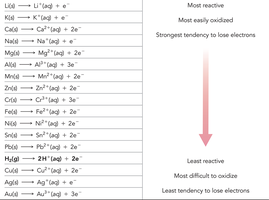
Activity Series and Spontaneity of Redox Reactions
The activity series ranks metals by their tendency to lose electrons (undergo oxidation). Reactions are spontaneous if the metal higher in the series is oxidized.
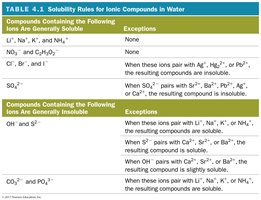
Summary Table: Solubility Rules for Ionic Compounds in Water
Compounds Containing the Following Ions Are Generally Soluble | Exceptions |
|---|---|
Li+, Na+, K+, and NH4+ | None |
NO3-, and C2H3O2- | None |
Cl-, Br-, and I- | When these ions pair with Ag+, Hg22+, or Pb2+, the resulting compound is insoluble |
SO42- | When SO42- pairs with Sr2+, Ba2+, Pb2+, or Ca2+, the resulting compound is insoluble |
Compounds Containing the Following Ions Are Generally Insoluble | Exceptions |
OH- and S2- | When these ions pair with Li+, Na+, K+, or NH4+, the resulting compounds are soluble. When S2- pairs with Ca2+, Sr2+, or Ba2+, the resulting compounds are soluble. When OH- pairs with Ca2+, Sr2+, or Ba2+, the resulting compounds are slightly soluble. |
CO32- and PO43- | When these ions pair with Li+, Na+, K+, or NH4+, the resulting compounds are soluble. |
