 Back
BackChapter 5: Thermochemistry – Energy and Chemical Reactions
Study Guide - Smart Notes

Thermochemistry: The Study of Energy in Chemical Reactions
Introduction to Energy and Thermochemistry
Thermochemistry is a branch of thermodynamics that focuses on the energy changes, particularly heat, involved in chemical reactions. Understanding how energy is transferred and transformed is essential for predicting reaction behavior and for practical applications in chemistry and engineering.
Energy is the ability to do work or transfer heat.
Thermodynamics studies energy and its transformations.
Thermochemistry specifically examines the heat changes in chemical reactions.
The Nature of Chemical Energy
Kinetic and Potential Energy
Energy in a system can be classified as kinetic or potential energy. Kinetic energy is associated with motion, while potential energy is related to position or composition.
Kinetic Energy (E_k): The energy of motion, given by the formula:
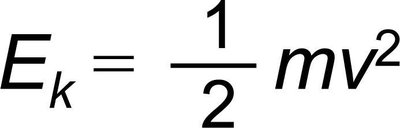
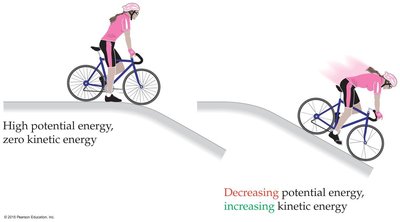
Potential Energy: Stored energy due to position or arrangement. In chemistry, the most important form is electrostatic potential energy between charged particles.
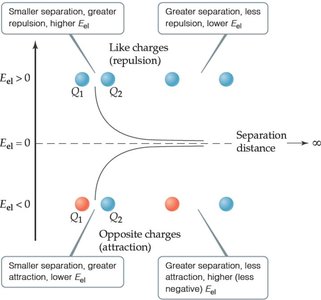
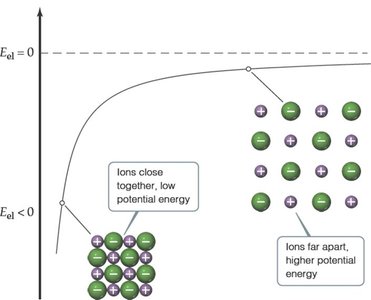
Electrostatic Potential Energy
Electrostatic interactions between charged particles are a major source of potential energy in chemical systems. The energy depends on the magnitude of the charges and their separation distance.
Energy is released when chemical bonds form (exothermic).
Energy is consumed when chemical bonds break (endothermic).
First Law of Thermodynamics
Law of Conservation of Energy
The first law states that energy can be converted from one form to another, but it cannot be created or destroyed. This principle underlies all energy changes in chemical reactions.
Examples: Chemical energy to heat (burning fuel), sunlight to chemical energy (photosynthesis).
System and Surroundings
In thermochemistry, we define a system (the part of the universe under study, e.g., reactants and products) and the surroundings (everything else).
Open system: Exchanges both matter and energy with surroundings.
Closed system: Exchanges energy but not matter.
Isolated system: Exchanges neither energy nor matter.
Work and Heat
Energy can be transferred as work (force acting over a distance) or as heat (energy transfer due to temperature difference).
Work:
Heat flows from warmer to cooler objects.
Internal Energy and State Functions
Internal Energy (E)
The internal energy of a system is the sum of all kinetic and potential energies of its components. The change in internal energy () is a state function, depending only on the initial and final states, not the path taken.
Thermodynamic Quantities: Number, Unit, and Sign
Thermodynamic values have a magnitude, a unit, and a sign.
Positive sign: System gains energy from surroundings.
Negative sign: System loses energy to surroundings.
Quantity | Positive (+) | Negative (−) |
|---|---|---|
q (heat) | System gains heat | System loses heat |
w (work) | Work done on system | Work done by system |
ΔE (internal energy) | Net gain of energy by system | Net loss of energy by system |
Heat Exchange: Endothermic and Exothermic Processes
When heat is absorbed by the system, the process is endothermic. When heat is released, it is exothermic.
State Functions vs. Path Functions
State functions (like internal energy, enthalpy) depend only on the state of the system, not on how it got there. Heat (q) and work (w) are path-dependent.
Enthalpy (H)
Definition and Properties
Enthalpy is a thermodynamic quantity defined as the internal energy plus the product of pressure and volume: .
At constant pressure, the change in enthalpy () equals the heat gained or lost by the system.
Endothermic: (system absorbs heat)
Exothermic: (system releases heat)
Pressure–Volume Work
Most chemical reactions involve pressure–volume work, especially when gases are involved. The work done by the system is negative (energy leaves the system).
Enthalpies of Reaction and Calorimetry
Enthalpy of Reaction ()
The enthalpy change for a reaction is the difference between the enthalpy of the products and the reactants:
Enthalpy is an extensive property (depends on amount).
Reversing a reaction changes the sign of.
Enthalpy depends on the physical states of reactants and products.
Calorimetry
Calorimetry is the measurement of heat flow. A calorimeter is used to measure the heat exchanged in a reaction.
Heat capacity: Energy required to raise the temperature of a substance by 1 K.
Specific heat: Heat capacity per gram.
Molar heat capacity: Heat capacity per mole.
Substance | Specific Heat (J/g·K) |
|---|---|
N2 (gas) | 1.04 |
H2O (liquid) | 4.18 |
Al (solid) | 0.90 |
CH4 (gas) | 2.20 |
Fe (solid) | 0.45 |
CO2 (gas) | 0.84 |
Hg (liquid) | 0.14 |
CaCO3 (solid) | 0.82 |
Constant-Pressure and Bomb Calorimetry
Constant-pressure calorimetry: Used for reactions in solution; heat change is measured by temperature change in water.
Bomb calorimetry: Used for reactions at constant volume; measures change in internal energy ().
Hess’s Law and Enthalpies of Formation
Hess’s Law
Hess’s law states that if a reaction is carried out in a series of steps, the overall enthalpy change is the sum of the enthalpy changes for the individual steps. This is possible because enthalpy is a state function.
Standard Enthalpies of Formation ()
The standard enthalpy of formation is the enthalpy change when one mole of a compound is formed from its elements in their standard states. By definition, the of an element in its standard state is zero.
Substance | Formula | (kJ/mol) |
|---|---|---|
Acetylene | C2H2 (g) | 226.7 |
Ammonia | NH3 (g) | -46.19 |
Benzene | C6H6 (l) | 49.0 |
Calcium carbonate | CaCO3 (s) | -1207.1 |
Carbon dioxide | CO2 (g) | -393.5 |
Water (liquid) | H2O (l) | -285.8 |
Water (gas) | H2O (g) | -241.8 |
Calculating Enthalpy Changes Using Standard Enthalpies of Formation
The enthalpy change for a reaction can be calculated as:
Where n and m are stoichiometric coefficients.
Bond Enthalpies
Definition and Use
Bond enthalpy is the energy required to break one mole of a specific bond in a gaseous molecule. It is always positive, as breaking bonds requires energy. The greater the bond enthalpy, the stronger the bond.
Bond | Bond Enthalpy (kJ/mol) |
|---|---|
C–H | 413 |
C–C | 348 |
C=C | 614 |
O–H | 463 |
O=O | 495 |
H–H | 436 |
Cl–Cl | 242 |
Energy is released when bonds form (exothermic).
Energy is required to break bonds (endothermic).
Estimating Enthalpy of Reaction Using Bond Enthalpies
The enthalpy change for a reaction can be estimated by subtracting the total bond energies of bonds formed from those broken:
If more energy is released in forming bonds than is required to break them, the reaction is exothermic.
Additional info: This summary covers the core concepts of thermochemistry, including energy, the first law of thermodynamics, internal energy, enthalpy, calorimetry, Hess's law, standard enthalpies of formation, and bond enthalpies, as presented in a typical general chemistry curriculum.
