 Back
BackChapter 5: Thermochemistry – Energy, Heat, and Chemical Reactions
Study Guide - Smart Notes

Thermochemistry and Energy
Introduction to Thermochemistry
Thermochemistry is the branch of chemistry that studies the energy and heat changes associated with chemical reactions and physical transformations. Understanding energy flow is essential for predicting reaction behavior and designing chemical processes.
Energy: The capacity to do work or transfer heat.
Thermodynamics: The study of energy transformations.
Thermochemistry: Focuses on energy changes during chemical reactions.
Forms of Energy
Kinetic Energy (KE): Energy of motion. Calculated as where m is mass and v is velocity.
Potential Energy (PE): Stored energy due to position or composition.
Units of Energy: The SI unit is the joule (J), where .
The First Law of Thermodynamics
Law Statement and Implications
The first law of thermodynamics states that energy can be converted from one form to another but cannot be created or destroyed. This principle is also known as the law of conservation of energy.
Example: Chemical energy in fuel is converted to heat to warm a house.
Example: Sunlight is converted to chemical energy in plants via photosynthesis.
System and Surroundings
Definitions
In thermochemistry, it is important to distinguish between the system and its surroundings:
System: The part of the universe chosen for study (e.g., the chemicals in a reaction vessel).
Surroundings: Everything outside the system (e.g., the container, air, etc.).
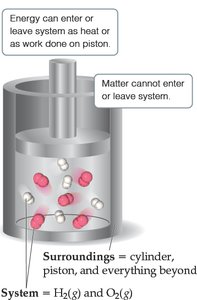
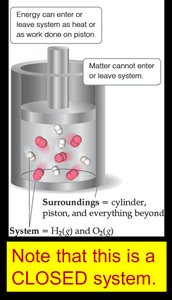
Types of Systems
Open System: Can exchange both matter and energy with surroundings.
Closed System: Can exchange energy (as heat or work) but not matter with surroundings.
Isolated System: Cannot exchange either matter or energy with surroundings.
Internal Energy (E)
Definition and Calculation
The internal energy of a system (E) is the sum of all kinetic and potential energies of its components. While the absolute value of E is not usually known, changes in internal energy (ΔE) are measurable and significant in chemical processes.
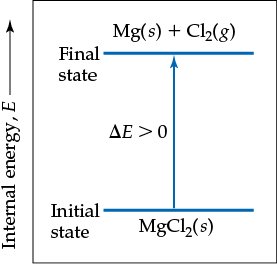
If , the system absorbs energy (endothermic).
If , the system releases energy (exothermic).
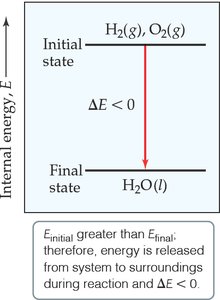
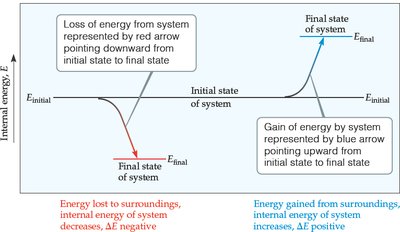
Energy Diagrams
Energy diagrams visually represent the change in internal energy during a reaction. For example, if the products have higher energy than the reactants, (energy absorbed).
Thermodynamic Quantities
Components of a Measurement
Every thermodynamic quantity consists of three parts:
A number (magnitude)
A unit (e.g., J, kJ)
A sign (indicating direction of energy flow)
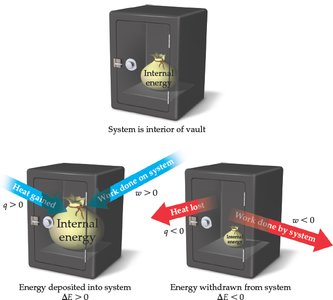
A positive means the system gains energy; a negative $\Delta E$ means the system loses energy.
Heat (q) and Work (w)
Energy is exchanged between system and surroundings as either heat (q) or work (w):
q: Heat absorbed (+) or released (−) by the system
w: Work done on (+) or by (−) the system
Sign Conventions
Quantity | Positive (+) | Negative (−) |
|---|---|---|
q (heat) | System gains heat | System loses heat |
w (work) | Work done on system | Work done by system |
ΔE (internal energy) | Net gain of energy by system | Net loss of energy by system |
Endothermic and Exothermic Processes
Definitions
Endothermic Process: Heat is absorbed by the system from the surroundings ().
Exothermic Process: Heat is released by the system to the surroundings ().


Examples
Endothermic: Ice melting, boiling soup (system absorbs heat).
Exothermic: Combustion, freezing water (system releases heat).
Calorimetry and Heat Measurement
Heat Capacity and Specific Heat
Heat Capacity (C): Amount of heat required to raise the temperature of an object by 1°C or 1 K.
Molar Heat Capacity: Heat capacity per mole of substance.
Specific Heat (Cs): Heat capacity per gram of substance.
Formulas:
Where q is heat, C is heat capacity, Cs is specific heat, m is mass, and ΔT is the temperature change.
Properties of Heat Capacity
Heat capacity is an extensive property (depends on amount of substance).
Specific heat is an intensive property (independent of amount, unique to each substance).
State Functions
Definition and Examples
A state function is a property that depends only on the current state of the system, not on the path taken to reach that state. Examples include internal energy (E), pressure (P), temperature (T), and volume (V).
ΔE is a state function; q and w are not (they depend on the process path).
Additional info: In thermochemistry, understanding the distinction between state functions and path functions is crucial for analyzing energy changes in chemical reactions and physical processes.
