 Back
BackChapter 5: Thermochemistry – Study Notes for General Chemistry II
Study Guide - Smart Notes

Chapter 5: Thermochemistry
5.1 The Nature of Chemical Energy
Thermochemistry is the study of energy changes that occur during chemical reactions and physical transformations. Energy exists in various forms, primarily as kinetic energy (energy of motion) and potential energy (stored energy due to position or composition). In chemistry, we focus on the energy associated with atoms and molecules, including the energy stored in chemical bonds and the movement of particles.
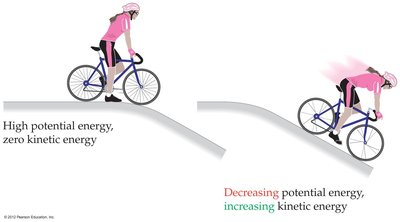
Kinetic Energy (Ek): The energy of an object in motion, calculated as where m is mass and v is velocity.
Potential Energy: Energy due to position or composition, such as energy stored in chemical bonds or a weight held above the ground.
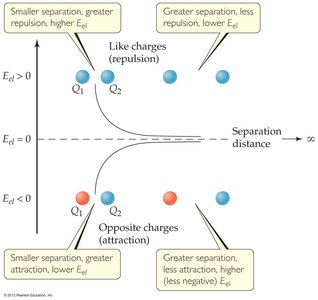
Electrostatic Potential Energy (Eel): Arises from interactions between charged particles, given by , where and are charges, is separation distance, and is a constant.
Example: A rock rolling down a hill converts potential energy to kinetic energy.
Electrostatic Potential Energy: Like charges repel (positive Eel), opposite charges attract (negative Eel), and energy approaches zero as separation increases.
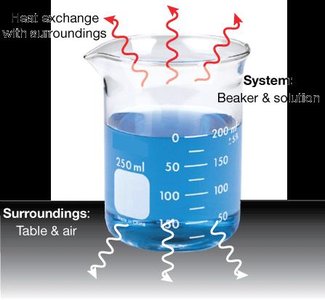
Transferring Energy: Energy can be transferred as work (mechanical movement) or heat (thermal energy).

5.1 System and Surroundings
To analyze energy changes, chemists define a system (the part under study) and surroundings (everything else). Systems can be classified based on their ability to exchange matter and energy:
Open System: Exchanges both matter and energy with surroundings.
Closed System: Exchanges only energy, not matter.
Isolated System: Exchanges neither matter nor energy.
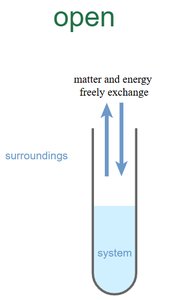
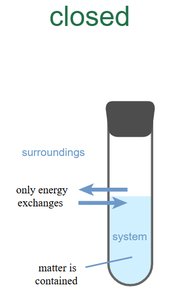
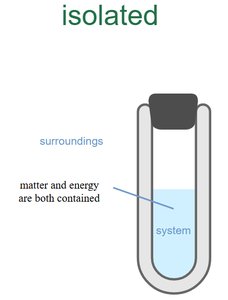
Example: An insulated thermos is an approximately isolated system.

5.2 The First Law of Thermodynamics
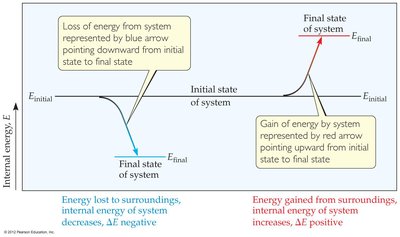
The first law states that energy can neither be created nor destroyed, only converted from one form to another. The internal energy (E) of a system is the sum of all kinetic and potential energies of its components. In thermochemistry, we focus on the change in internal energy ():
where q is heat and w is work
Sign Conventions:
Quantity | Positive (+) | Negative (-) |
|---|---|---|
q (heat) | System gains heat | System loses heat |
w (work) | Work done on system | Work done by system |
ΔE | Net gain of energy by system | Net loss of energy by system |

Endothermic vs. Exothermic:
Endothermic: System absorbs heat (ΔE positive)
Exothermic: System releases heat (ΔE negative)
Example: Melting ice is endothermic; combustion of gasoline is exothermic.
Pressure-Volume Work
When gases expand or contract, work is done. Pressure-volume work (P-V work) is calculated as:
(at constant pressure)
If gas expands: ,
If gas contracts: ,
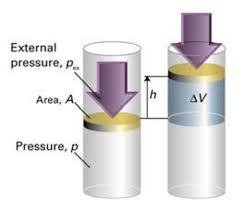
Example: Calculate work when volume changes at constant pressure.
State Functions
State functions are properties that depend only on the current state of the system, not the path taken to reach that state. Examples include pressure, volume, temperature, and internal energy. Heat and work are not state functions.
Change in state function:
5.3 Enthalpy
Enthalpy (H) is the heat content of a system at constant pressure. It is defined as:
Change in enthalpy:
Enthalpy is a state function.
5.4 Enthalpies of Reaction
The enthalpy of reaction () is the heat released or absorbed during a chemical reaction at constant pressure. Thermochemical equations show the enthalpy change for a reaction.
If , is negative (exothermic).
If , is positive (endothermic).
Enthalpy is an extensive property (depends on sample size).
Enthalpy change for reverse reaction is equal in magnitude, opposite in sign.
Enthalpy change depends on physical states of reactants and products.
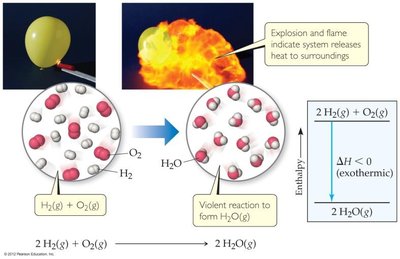
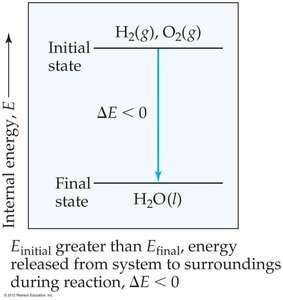
Example: CH4 (g) + 2 O2 (g) → CO2 (g) + 2 H2O (l), kJ
Relating ΔH to Quantities of Reactants and Products
To calculate heat released or absorbed, use stoichiometry and thermochemical equations. For example, burning 4.50 g of methane:
Convert grams to moles using molar mass.
Multiply by enthalpy change per mole.
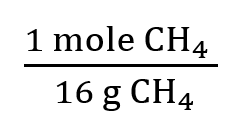
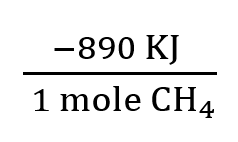
5.5 Calorimetry
Calorimetry is the measurement of heat flow during a reaction. A calorimeter is used to measure heat transfer. The heat capacity is the amount of heat required to raise the temperature of a material by 1°C (or 1 K).
Molar Heat Capacity: Heat capacity for one mole (J/mol·K).
Specific Heat Capacity (Cs): Heat capacity for one gram (J/g·K).
Formula: or
Example: Water has a high specific heat (4.18 J/g·K), making it valuable as a coolant.
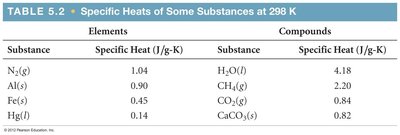
Substance | Specific Heat (J/g·K) |
|---|---|
H2O (l) | 4.18 |
CH4 (g) | 2.20 |
CO2 (g) | 0.84 |
CaCO3 (s) | 0.82 |
N2 (g) | 1.04 |
Al (s) | 0.90 |
Fe (s) | 0.45 |
Hg (l) | 0.14 |
Relating Heat, Temperature Change, and Heat Capacity:
To warm 250 g of water from 22°C to 98°C: J or 79.42 kJ
To determine specific heat of a metal:
Example: How much energy does it take to raise the body temperature 2.5ºC for someone weighing 50 kg, with average body specific heat capacity of 3 J/g·ºC? J = 375 kJ
Additional info: Sections 5.6 (Hess’s Law) and 5.7 (Enthalpies of formation) are outlined but not covered in detail in the provided materials.
