 Back
BackChapter 6: Electronic Structure of Atoms – Study Notes
Study Guide - Smart Notes

Electronic Structure of Atoms
Key Terminology
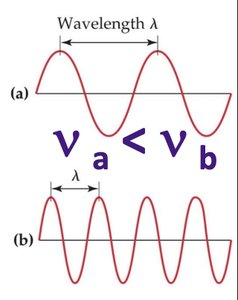
Frequency (\(\nu\)): The number of cycles (waves) that pass a point in one second, measured in hertz (Hz).
Wavelength (\(\lambda\)): The distance between successive peaks (or troughs) of a wave, typically measured in nanometers (nm).
Electromagnetic Spectrum: The range of all types of electromagnetic radiation, arranged by increasing wavelength or frequency.
Blackbody Radiation: Emission of light from hot objects.
Photoelectric Effect: Emission of electrons from metal surfaces when light shines on them.
Emission Spectra: Light emitted from electronically excited gas atoms, producing discrete lines.
Quantum: The smallest amount of energy that can be emitted or absorbed as electromagnetic radiation.
Valence Electrons: Electrons in the outermost shell, involved in chemical reactions.
Pauli Exclusion Principle: No two electrons in the same atom can have the same set of four quantum numbers.
The Wave Nature of Light
Properties of Electromagnetic Radiation
Understanding the electronic structure of atoms requires knowledge of electromagnetic radiation, which exhibits wave-like behavior. All waves have a characteristic wavelength (\(\lambda\)) and frequency (\(\nu\)).
Wavelength (\(\lambda\)): Measured in nanometers (1 nm = 1 × 10−9 m).
Frequency (\(\nu\)): Measured in hertz (Hz), where 1 Hz = 1 s−1.
The relationship between wavelength and frequency is given by:
The Electromagnetic Spectrum
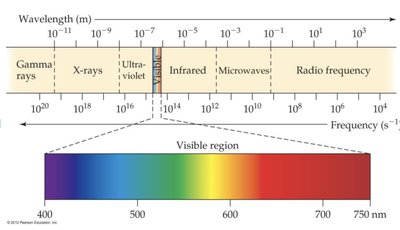
The electromagnetic spectrum displays all types of electromagnetic radiation, from gamma rays to radio waves, arranged by increasing wavelength and decreasing frequency. The visible region is a small part of this spectrum.
Sample Calculation: Frequency from Wavelength
To calculate the frequency of light given its wavelength, use the equation \(\nu = \frac{c}{\lambda}\):

Quantum Theory and the Nature of Energy
Blackbody Radiation and Quantization
The wave model of light cannot explain phenomena such as blackbody radiation. Max Planck proposed that energy is quantized and comes in discrete packets called quanta (singular: quantum).
Planck's constant (h): 6.626 × 10−34 J·s
The energy of a quantum is given by:

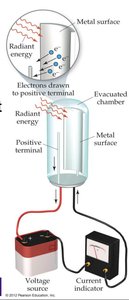
The Photoelectric Effect
When light of a certain frequency shines on a metal surface, electrons are ejected. Einstein explained this by proposing that light consists of photons, each with energy \(E = h\nu\). The photoelectric effect demonstrates the particle nature of light.
Energy of a photon can also be expressed as:
Atomic Emission Spectra
Continuous vs. Line Spectra
Atoms and molecules emit light at specific wavelengths, producing a line spectrum rather than a continuous spectrum. Each element has a unique line spectrum, which can be used for identification.
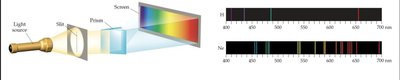
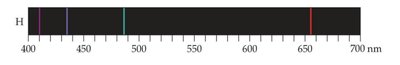
The Hydrogen Spectrum and Rydberg Formula
The wavelengths of visible lines in the hydrogen spectrum can be described by the Rydberg formula:
where \(R_H = 1.09677 \times 10^7\, \text{m}^{-1}\).
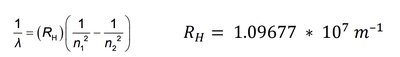
The Bohr Model of the Atom
Bohr's Postulates
Niels Bohr explained the hydrogen atom's line spectrum by proposing that electrons occupy only certain allowed orbits with specific energies. Energy is absorbed or emitted only when an electron transitions between these orbits.
Ground state: Lowest energy level (n = 1).
Excited state: Any energy level higher than the ground state.
Energy change for a transition:
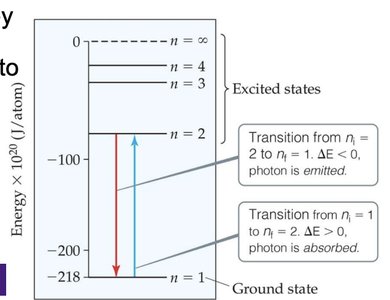
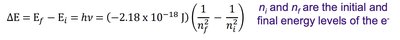
Limitations and Benefits of the Bohr Model
Only accurately describes hydrogen (one-electron systems).
Introduced the concept of quantized energy levels and quantum numbers.
Electrons do not move in fixed circular orbits as classical physics would suggest.
Quantum Mechanical Model of the Atom
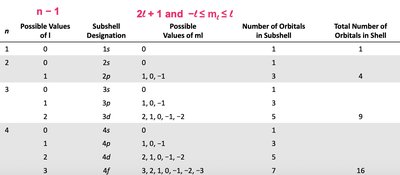
Quantum Numbers
The quantum mechanical model describes electrons in terms of wave functions (orbitals) and quantum numbers:
n | l | ml | ms |
|---|---|---|---|
1, 2, 3, ... | 0 to n-1 | -l to +l | +1/2 or -1/2 |
Shell (size) | Subshell (shape) | Orbital orientation | Spin |

Principal quantum number (n): Indicates the energy level and size of the orbital.
Angular momentum quantum number (l): Defines the shape of the orbital (0 = s, 1 = p, 2 = d, 3 = f).
Magnetic quantum number (ml): Specifies the orientation of the orbital.
Spin quantum number (ms): Specifies the spin of the electron (+1/2 or -1/2).
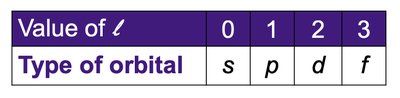
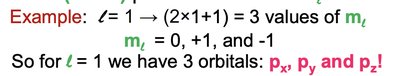
Shapes and Types of Orbitals
s orbitals (l = 0): Spherical shape, one per energy level.
p orbitals (l = 1): Dumbbell-shaped, three orientations (px, py, pz).
d orbitals (l = 2): Four have cloverleaf shapes, one has a unique shape.
f orbitals (l = 3): Complex shapes, seven orientations.
Electron Configurations
Writing Electron Configurations
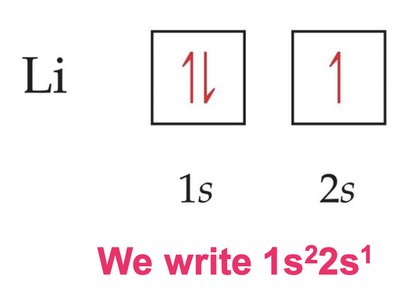
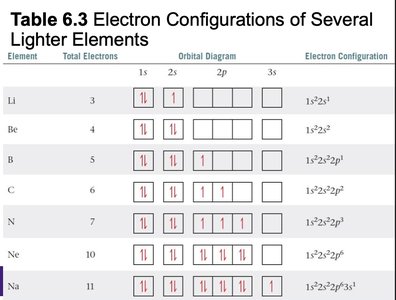
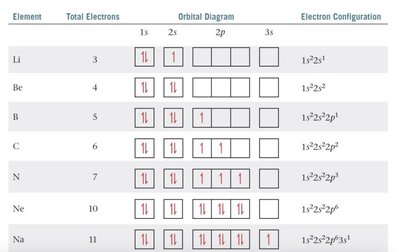
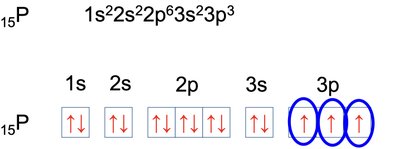
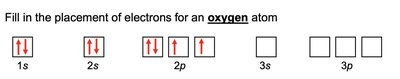
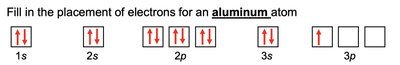
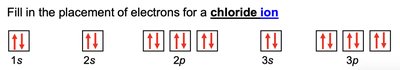
Electron configuration describes the arrangement of electrons in an atom. The notation consists of the energy level (n), the type of orbital (l), and the number of electrons in that orbital as a superscript (e.g., 1s2).
Electrons fill orbitals in order of increasing energy (Aufbau principle).
No more than two electrons per orbital (Pauli exclusion principle).
Electrons fill degenerate orbitals singly before pairing (Hund's rule).
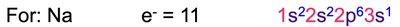
Order of Orbital Filling
The order in which orbitals are filled can be determined using the diagonal rule or the periodic table:
1s, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, etc.
Condensed Electron Configurations
Condensed notation uses the symbol of the nearest noble gas in brackets to represent core electrons, followed by the remaining valence electrons.
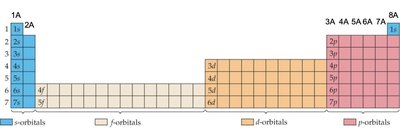
Electron Configurations and the Periodic Table
The periodic table is a guide for electron configurations. The period number corresponds to the principal quantum number (n), and the block (s, p, d, f) indicates the type of orbital being filled.


Important Anomalies in Electron Configurations
Some elements have electron configurations that differ from the expected order due to the stability of half-filled and fully filled d subshells. For example:
Chromium (Cr): [Ar] 4s1 3d5 instead of [Ar] 4s2 3d4
Copper (Cu): [Ar] 4s1 3d10 instead of [Ar] 4s2 3d9
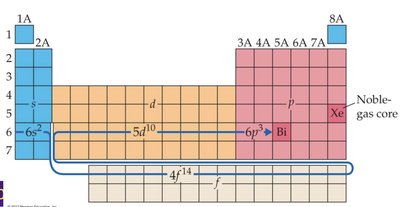
Valence Electrons
Valence electrons are those in the outermost shell beyond the noble gas core. They are responsible for chemical reactivity and bonding.
