 Back
Back6 PP
Study Guide - Smart Notes

Gases: Properties and Behavior
Nature of Gases
Gases are composed of particles (atoms or molecules) that move rapidly and occupy a large amount of empty space within their container. The movement of these particles is random and occurs in straight lines until they collide with either the container wall or another particle, after which they bounce off.
Low density: Gas molecules are far apart compared to solids and liquids.
Compressibility: Gases can be compressed easily due to the large spaces between particles.
Expansion: Gases expand to fill the shape and volume of their container.
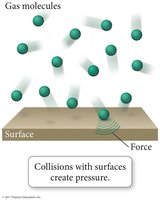
Gas Pressure
Pressure is the force exerted per unit area by gas molecules as they strike the surfaces around them. Each collision transfers momentum, creating measurable pressure.
Atmospheric pressure: The pressure exerted by the air in Earth's atmosphere.
Pressure and altitude: Pressure decreases with increasing altitude due to fewer gas particles in a given volume.
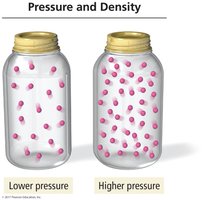
Pressure and Density
The pressure exerted by a gas depends on the number of gas particles in a given volume. Higher density (more particles per volume) results in higher pressure.
Lower pressure: Fewer gas particles in a container.
Higher pressure: More gas particles in a container.
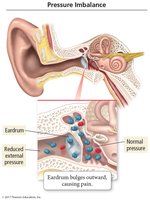
Pressure Imbalance in the Ear
If there is a difference in pressure across the eardrum membrane, the membrane will bulge outward or inward, causing discomfort or pain. This is commonly experienced during altitude changes, such as driving over mountains.
Units of Pressure
Pressure can be measured in several units. The most common units are:
Atmosphere (atm)
Millimeters of mercury (mmHg)
Torr
Pascals (Pa)
Conversion between units is essential for gas law calculations.
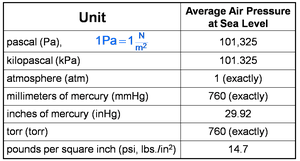
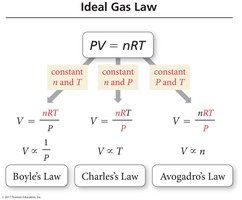
Gas Laws
Simple Gas Laws
The four basic properties of a gas are pressure (P), volume (V), temperature (T), and amount in moles (n). These properties are interrelated, and the simple gas laws describe the relationships between pairs of these properties.
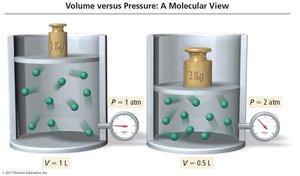
Boyle’s Law: Pressure and Volume
Boyle’s Law states that the pressure of a gas is inversely proportional to its volume at constant temperature and amount of gas.
Mathematical form:
Equation:
As pressure increases, volume decreases by the same factor.


Charles’s Law: Volume and Temperature
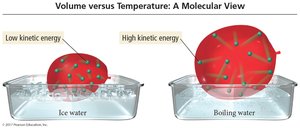
Charles’s Law states that the volume of a fixed amount of gas at constant pressure increases linearly with increasing temperature (in kelvins).
Mathematical form:
Equation:
Temperature must be in kelvins:
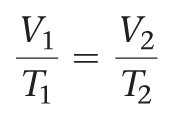
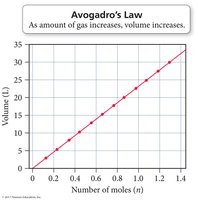
Avogadro’s Law: Volume and Amount
Avogadro’s Law states that the volume of a gas is directly proportional to the number of moles of gas at constant pressure and temperature.
Mathematical form:
Equal volumes of gases contain equal numbers of molecules (at given P and T).
Combined Gas Law and Ideal Gas Law
The relationships described by Boyle’s, Charles’s, and Avogadro’s laws can be combined into the Ideal Gas Law, which relates all four properties:
Equation:
R: Gas constant,
Standard Conditions and Molar Volume
Standard Temperature and Pressure (STP) is defined as 1 atm and 273 K (0 °C). At STP, one mole of any ideal gas occupies 22.4 L.
Molar volume:
At STP: for 1 mole


Density and Molar Mass of Gases
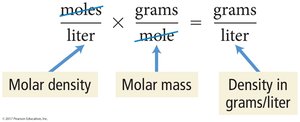
Density of a Gas
Density is the ratio of mass to volume and is generally given in g/L for gases. The density of a gas is directly proportional to its molar mass.
Equation:
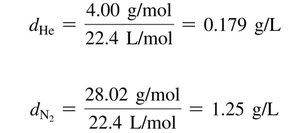

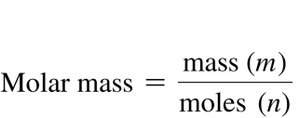

Mixtures of Gases and Partial Pressures
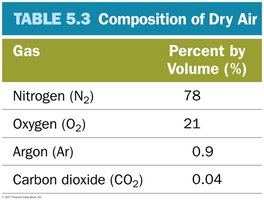
Composition of Air
Dry air is a mixture of several gases, primarily nitrogen, oxygen, argon, and carbon dioxide.
Gas | Percent by Volume (%) |
|---|---|
Nitrogen (N2) | 78 |
Oxygen (O2) | 21 |
Argon (Ar) | 0.9 |
Carbon dioxide (CO2) | 0.04 |
Dalton’s Law of Partial Pressures
In a mixture of gases, each gas exerts its own pressure, called partial pressure. The total pressure is the sum of the partial pressures of all components.
Equation:
Partial pressure can be calculated using the ideal gas law for each component.
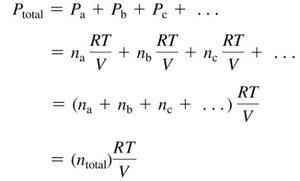
Mole Fraction
The mole fraction is the ratio of the number of moles of a component to the total number of moles in the mixture. For gases, the mole fraction is equivalent to its percent by volume divided by 100%.
Equation:


Stoichiometry and Reactions Involving Gases
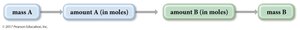
Stoichiometric Calculations
Stoichiometry involves relationships between amounts in moles. For reactions involving gases, the ideal gas law can be used to determine the amounts in moles from the volumes, or to determine the volumes from the amounts in moles.
Kinetic Molecular Theory
Postulates of Kinetic Molecular Theory
The kinetic molecular theory explains the behavior of ideal gases:
The size of a particle is negligibly small.
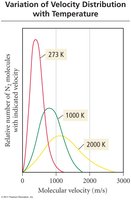
The average kinetic energy of gas particles is proportional to the Kelvin temperature.
Collisions between particles and with container walls are completely elastic.

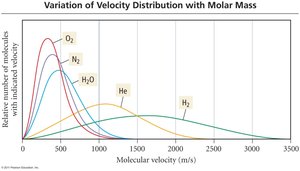
Kinetic Energy and Molecular Velocities
The average kinetic energy of gas molecules depends on their mass and velocity. At the same temperature, lighter particles move faster than heavier particles.
Equation:
Mean Free Path, Diffusion, and Effusion
Definitions
Mean Free Path: The average distance a molecule travels between collisions.
Diffusion: The process of gas molecules spreading out in response to a concentration gradient. Heavier molecules diffuse slower than lighter ones.
Effusion: The process of gas molecules escaping from a container to a vacuum through a small hole. Heavier molecules effuse slower than lighter ones.
Real Gases
Deviation from Ideal Behavior
Real gases deviate from ideal behavior because their particles have volume and experience intermolecular forces. These effects cause real gases to occupy larger volumes and exert lower pressures than predicted by the ideal gas law, especially at high pressures and low temperatures.
