 Back
BackChapter 7: Periodic Properties of the Elements – Study Notes
Study Guide - Smart Notes

Chapter 7: Periodic Properties of the Elements
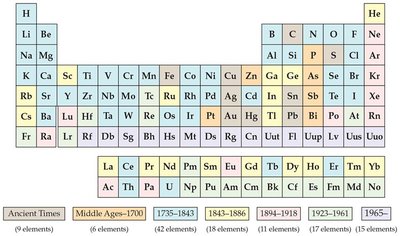
7.1 Development of the Periodic Table
The periodic table is a systematic arrangement of elements based on their atomic properties. Its development was a major milestone in chemistry, allowing scientists to predict the properties of elements and their compounds.
Mendeleev and Meyer: Dmitri Mendeleev and Lothar Meyer independently proposed that elements should be grouped according to recurring (periodic) properties.
Mendeleev's Predictions: Mendeleev is especially credited because he used chemical properties to organize the table and predicted the existence and properties of undiscovered elements, such as germanium.
Atomic Number: Originally, the table was organized by atomic mass. Later, Henry Moseley established that atomic number (number of protons) is the fundamental property determining periodicity.
Periodicity: The periodic repetition of chemical and physical properties of the elements is called periodicity.
7.2 Effective Nuclear Charge
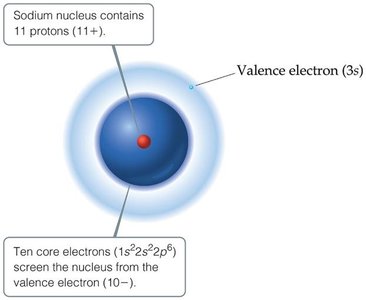
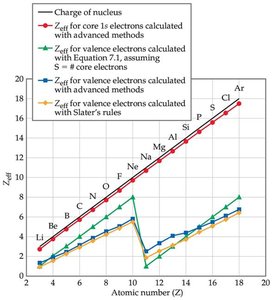
Many atomic properties depend on the attraction between valence electrons and the nucleus, which is reduced by the repulsion from other electrons. This net positive charge experienced by valence electrons is called the effective nuclear charge (Zeff).
Definition: where is the atomic number and is the screening constant (approximate number of core electrons).
Trends: Effective nuclear charge increases across a period (left to right) and changes little down a group.
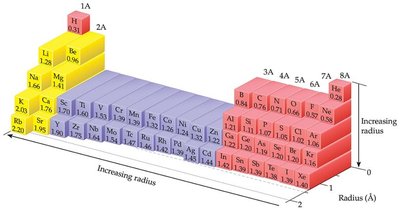
7.3 Sizes of Atoms and Ions
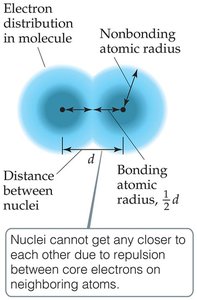
The size of an atom is determined by the space occupied by its electrons. Atomic size can be described in several ways:
Nonbonding atomic radius (van der Waals radius): Half the shortest distance between nuclei during collisions.
Bonding atomic radius (covalent radius): Half the distance between nuclei in a molecule.
Trends: Atomic radius decreases across a period (due to increasing Zeff) and increases down a group (due to increasing principal quantum number, n).
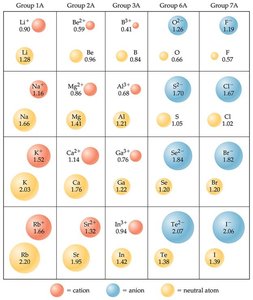
Sizes of Ions (Ionic Radii)
Cations: Smaller than their parent atoms because electrons are removed, reducing electron-electron repulsion.
Anions: Larger than their parent atoms due to added electrons and increased repulsion.
Isoelectronic Series: Ions with the same number of electrons; size decreases with increasing nuclear charge.
7.4 Ionization Energy and Electron Affinity
These properties describe the energy changes associated with gaining or losing electrons.
Ionization Energy (I)
Definition: The minimum energy required to remove an electron from a gaseous atom or ion.
First Ionization Energy (I1): Energy to remove the first electron.
Trends: Increases across a period, decreases down a group. There are exceptions due to sublevel filling and electron pairing.
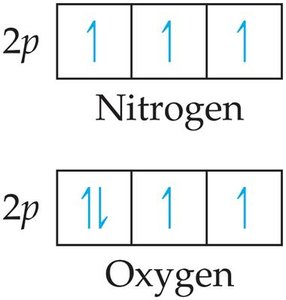

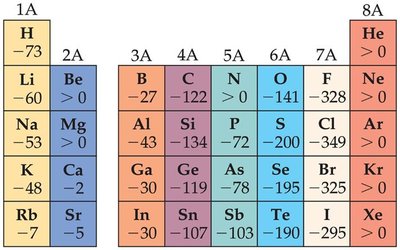
Electron Affinity (EA)
Definition: The energy change when an electron is added to a gaseous atom. Usually exothermic (negative value).
Trends: Generally becomes more negative across a period, with exceptions (e.g., Group 2A, 5A, 8A).
7.5 Metals, Nonmetals, and Metalloids
Elements are classified based on their physical and chemical properties as metals, nonmetals, or metalloids.
Metals
Properties: Shiny, good conductors, malleable, ductile, low ionization energies, form cations.
Chemistry: Form ionic compounds with nonmetals; metal oxides are basic.


Nonmetals
Properties: Can be solid, liquid, or gas; solids are dull, brittle, poor conductors; large negative electron affinities, form anions.
Chemistry: Form molecular compounds; nonmetal oxides are acidic.


Metalloids
Properties: Exhibit properties intermediate between metals and nonmetals; some are semiconductors.

Comparison Table: Metals vs Nonmetals
Metals | Nonmetals |
|---|---|
Shiny luster; various colors, mostly silvery | No luster; various colors |
Malleable and ductile | Brittle solids; some hard, some soft |
Good conductors of heat and electricity | Poor conductors |
Most oxides are basic | Most oxides are acidic |
Form cations in solution | Form anions or oxyanions in solution |
7.6 Group Trends
Elements in the same group (vertical column) of the periodic table have similar properties. Important groups include:
Group 1A: Alkali Metals – Soft, metallic, low density, low melting points, very reactive, form +1 cations.
Group 2A: Alkaline Earth Metals – Harder, higher melting points and densities than alkali metals, form +2 cations.
Group 6A: Oxygen Group – Includes nonmetals (O, S, Se), a metalloid (Te), and a metal (Po); increasing metallic character down the group.
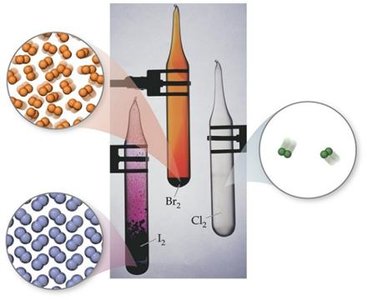
Group 7A: Halogens – Typical nonmetals, highly negative electron affinities, form anions, very reactive.
Group 8A: Noble Gases – Monatomic gases, very unreactive, high ionization energies, positive electron affinities.




Summary Table: Key Periodic Trends
Property | Across a Period (→) | Down a Group (↓) |
|---|---|---|
Atomic Radius | Decreases | Increases |
Ionization Energy | Increases | Decreases |
Electron Affinity | Becomes more negative | Becomes less negative |
Metallic Character | Decreases | Increases |
