 Back
BackChapter 7: Thermochemistry and Thermodynamics Study Notes
Study Guide - Smart Notes

Thermodynamics: Key Concepts
System, Surroundings, and Universe
Thermodynamics is the study of energy changes, particularly in chemical reactions and physical processes. Understanding the distinction between the system and its surroundings is fundamental:
System: The part of the universe under study, typically the chemical reaction or physical process of interest.
Surroundings: Everything else in the universe outside the system.
Universe: The sum of the system and its surroundings.
Key Principle: The total energy of the universe is constant. Energy lost by the system is gained by the surroundings and vice versa.
Mathematical Expression:



Sign Conventions in Thermodynamics
Thermodynamic quantities such as heat (q), work (w), and change in internal energy (ΔE) are assigned positive or negative signs depending on the direction of energy flow:
Quantity | Sign (System) | Change for System | Sign (Surroundings) | Change for Surroundings |
|---|---|---|---|---|
q | + | Absorbing heat (Endothermic) | - | Losing heat |
q | - | Losing heat (Exothermic) | + | Absorbing heat |
w | + | Work done on system (decreasing volume) | - | Doing work (increasing volume) |
w | - | Work done by system (increasing volume) | + | Work done on surroundings (decreasing volume) |
ΔE | + | Gaining energy | - | Losing energy |
ΔE | - | Losing energy | + | Gaining energy |

Internal Energy and Its Calculation
Definition and Formula
Internal energy (E) is the total energy contained within a system. The change in internal energy (ΔE) is given by:
q: Heat exchanged with the surroundings
w: Work done by or on the system
Sample Problems
Example 1: A gas absorbs 5.973 kJ of heat and does 0.589 kJ of work by pushing a piston. What is ΔE?
Example 2: A 10.0 L balloon loses 206 J of heat, and ΔE = -485 J. What is the value of work (w)? Did the balloon expand or contract?

Heat Capacity and Calorimetry
Specific Heat and Molar Heat Capacity
Specific heat (C_s): The amount of heat required to raise the temperature of one gram of a substance by one degree Celsius.
Molar heat capacity (C_m): The amount of heat required to raise the temperature of one mole of a substance by one degree Celsius.
Formula:
q: Heat absorbed or released
m: Mass of the substance
C_s: Specific heat capacity
ΔT: Change in temperature
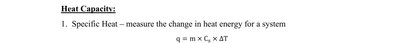
Sample Calorimetry Problems
Calculate the heat required to warm 420.0 g of ethylene glycol from 22.2°C to 100.0°C. (C_s = 2.78 J/g°C, d = 1.1132 g/mL)
Find the final temperature of 5.009 g of gold at 37.0°C after absorbing 11.04 J of heat. (C_s = 0.129 J/g°C)
Determine the heat absorbed by 1.0 tablespoon of chicken broth heated from 22.2°C to 100.0°C. (Assume properties of water: d = 0.99875 g/mL, C_s = 4.184 J/g°C)
Calculate the temperature change for a 63.1 g silver spoon absorbing the same heat as above. (C_s = 0.240 J/g°C)
Find the final temperature of the silver spoon.
A hot piece of iron at 275.5°C is placed in 200 g of water at 22.2°C. Final temperature is 28.1°C. What is the mass of the iron? (C_s,Fe = 0.449 J/g°C, C_s,water = 4.184 J/g°C)

Pressure-Volume Work
Definition and Formula
Pressure-volume (P-V) work occurs when the volume of a system changes against an external pressure. The work done by the system is:
Where:
Pext: External pressure
ΔV: Change in volume
To convert between units:
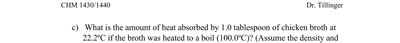
Sample Problem
Calculate the work done (in Joules) by inflating a balloon from 2.0 L to 3.8 L on the moon, where atmospheric pressure is 0.00500 atm.
Calorimetry: Measuring Energy Changes
Constant Volume Calorimetry (Bomb Calorimeter)
Bomb calorimetry is used to measure the change in internal energy (ΔE) of a reaction at constant volume. In this setup:
The system is sealed, so no matter can enter or leave.
All energy from the reaction is transferred as heat, since ΔV = 0 and no P-V work is done.
The heat absorbed by the calorimeter equals the energy released or absorbed by the reaction.

Relationship: (no heat is lost to the surroundings)
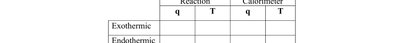
Temperature and Energy Relationships
Process | q (Reaction) | T (Reaction) | q (Calorimeter) | T (Calorimeter) |
|---|---|---|---|---|
Exothermic | - | Decreases | + | Increases |
Endothermic | + | Increases | - | Decreases |

Sample Bomb Calorimeter Calculations
Determine ΔErxn for the combustion of ethanol if 1.015 g of ethanol causes the calorimeter temperature to rise from 22.8°C to 35.2°C. (Calorimeter heat capacity = 5.936 kJ/°C)
Determine ΔErxn for the combustion of CH3OH if 1.015 g of CH3OH causes the temperature to rise from 21.4°C to 34.0°C. (Calorimeter heat capacity = 4.90 kJ/°C)
Find the calorimeter constant if 1.009 g of butane causes a 10.6°C temperature change. (ΔErxn for butane = -2878 kJ/mol)
Constant Pressure Calorimetry (Coffee Cup Calorimeter)
Used for reactions open to the atmosphere (constant pressure). The heat measured (qp) is related to the enthalpy change (ΔH) of the reaction.
Cannot assume all work is zero; only pressure-volume work is considered.
ΔH is the thermodynamic property measured at constant pressure.
Enthalpy (H)
Definition and Equations
Enthalpy (H): A thermodynamic property defined as the sum of the internal energy and the product of pressure and volume.
The change in enthalpy (ΔH) is:
If ΔV is negligible, then ΔH ≈ ΔE.

Sample Enthalpy Calculation
The reaction of one mole of sodium with water produces 367.5 kJ of heat and does 2.5 kJ of work. Calculate ΔH and ΔE for the reaction.
Coffee Cup Calorimeter Calculations
For aqueous reactions, ΔHrxn can be determined using constant pressure calorimetry.
System: chemical reaction; Surroundings: water and calorimeter.
Summary Table: Key Thermodynamic Quantities
Quantity | Symbol | Definition | Units |
|---|---|---|---|
Internal Energy | E | Total energy of a system | J (Joules) |
Change in Internal Energy | ΔE | q + w | J (Joules) |
Enthalpy | H | E + PV | J (Joules) |
Change in Enthalpy | ΔH | ΔE + PΔV | J (Joules) |
Heat | q | Energy transferred due to temperature difference | J (Joules) |
Work | w | Energy transferred when an object is moved by a force | J (Joules) |
Additional info: These notes cover the core concepts of thermochemistry and thermodynamics as outlined in a general chemistry curriculum, including definitions, sign conventions, calorimetry, and sample calculations. The included images directly support the explanation of key concepts, formulas, and apparatus.
