 Back
BackChapter 9: Thermochemistry – Energy, Heat, and Enthalpy in Chemical Reactions
Study Guide - Smart Notes

Energy
Types of Energy
Energy is the ability to cause a change in a physical system. In chemistry, energy is fundamental to understanding how and why reactions occur. There are two main types of energy:
Kinetic energy: Associated with motion. An example is thermal energy, which is the energy of moving particles.
Potential energy: Associated with position or composition. Chemical energy is a form of potential energy stored in chemical bonds.
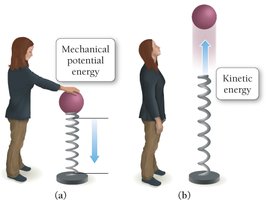
First Law of Thermodynamics
Law of Conservation of Energy
The first law of thermodynamics states that energy is neither created nor destroyed; it is constant in the universe. Energy can be transformed from one type to another or transferred between objects.
Transferring Energy
Work and Heat
Energy can be transferred in two main ways:
Work (w): Involves the action of a force through a distance.
Heat (q): Involves a change in temperature.
Both work and heat are path functions, meaning they depend on how the final state is reached.
Energy Transfer Terminology
System and Surroundings
In thermochemistry, the system refers to the chemicals involved in the reaction, while the surroundings are everything else.
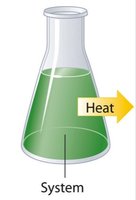
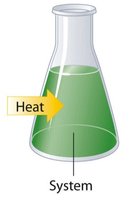
Internal Energy of a System (E)
State Function
The internal energy (E) of a system is the sum of the kinetic and potential energies of all particles in the system. It is a state function, meaning it depends only on the current state of the system, not on the path taken to reach that state.
Explaining State Functions
State functions are properties that depend only on the initial and final states, not on the process used to get there. For example, elevation is a state function; the route taken to climb a mountain does not affect the final elevation.
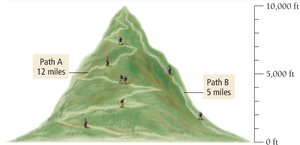
Change in Energy
Energy Change in Reactions
The change in internal energy (\( \Delta E \)) is calculated by comparing the initial and final conditions:
\( \Delta E = E_{final} - E_{initial} \)
For a reaction: \( \Delta E_{rxn} = E_{products} - E_{reactants} \)
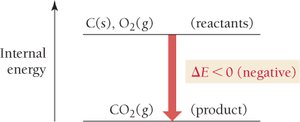
Importance of Sign
Negative \( \Delta E \): Energy is released by the system.
Positive \( \Delta E \): Energy is absorbed by the system.
Calculating \( \Delta E \)
Relationship to Work and Heat
\( \Delta E \) depends on the energy transfer functions:
Work (w)
Heat (q)
For a reaction:
\( \Delta E = q + w \)
\( \Delta E_{rxn} = q + w \)
Work (w)
Calculation and Units
Work is calculated by the change in volume due to applied force, primarily caused by gases in the system. The formula is:
\( w = -P \Delta V \)
SI Units:
Pressure: Pascals (Pa)
Volume: cubic meters (m3)
Work: joules (J)
Alternate Units:
Pressure: atmospheres (atm)
Volume: liters (L)
Work: liter-atmospheres (L·atm)
Direction of Transfer
Work done by the system (expansion): \( w < 0 \)
Work done on the system (contraction): \( w > 0 \)
Thermodynamics and Thermochemistry
Definitions
Thermodynamics is the study of energy transferred as heat. Thermochemistry focuses on the heat involved in chemical reactions.


Heat (q)
Heat Flow and Reaction Types
Heat flows from high to low temperatures. Types of reactions:
Endothermic: Energy enters the system (\( q > 0 \), +q).
Exothermic: Energy exits the system (\( q < 0 \), -q).
Heat and Temperature
Heat Capacity and Specific Heat
Heat capacity (c) is the amount of heat required to raise the temperature of a substance by 1°C. Specific heat (C_{sp}) is the heat required to raise the temperature of 1 gram of a substance by 1°C.
Formula: q = c × deltaT
Molar heat capacity (C) is the heat required to raise the temperature of 1 mole of a substance by 1°C.
Mixing Substances at Different Temperatures
Insulated Mixture Example
When substances at different temperatures are mixed in an insulated container, heat is transferred until thermal equilibrium is reached. The final temperature (Tf) can be calculated using the heat capacities of the substances.
Enthalpy (H)
Definition and Properties
Enthalpy (H) is a state function that represents the heat content of a system at constant pressure. The change in enthalpy (\( \Delta H \)) for a reaction is:
\( \Delta H_{rxn} = \Delta E_{rxn} + \Delta (PV)_{rxn} \)
At constant pressure: \( \Delta H_{rxn} = \Delta E_{rxn} + P \Delta V_{rxn} \)
Enthalpy is also known as the heat of reaction and is an extensive property, meaning it depends on the amount of substance.
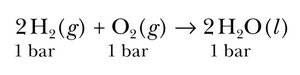
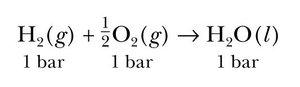
Calorimetry
Measuring Enthalpy Changes
Calorimetry is the experimental measurement of heat changes in chemical reactions. A coffee-cup calorimeter is commonly used for reactions at constant pressure.
Example: Magnesium reacts with hydrochloric acid, and the temperature change is used to calculate \( \Delta H_{rxn} \).
Hess’s Law
Combining Chemical Equations
Hess’s Law states that the enthalpy change for a reaction is the sum of the enthalpy changes for individual steps. This allows calculation of \( \Delta H_{rxn} \) for complex reactions by combining simpler reactions.
If a reaction is reversed, the sign of \( \Delta H_{rxn} \) is reversed.
If a reaction is multiplied by a coefficient, \( \Delta H_{rxn} \) is multiplied by the same coefficient.
Standard Enthalpy Change (\( \Delta H^\circ_{rxn} \))
Definition
Standard enthalpy change is measured when all reactants and products are in their standard states (1 bar for gases, pure solids/liquids, 1 M for solutions). It is indicated with a superscript "°" and measured in kJ/mol.
Standard Enthalpy of Formation (\( \Delta H^\circ_f \))
Definition and Calculation
The standard enthalpy of formation is the enthalpy change for the formation of 1 mole of a compound from its elements in their standard states. For stable elements, \( \Delta H^\circ_f = 0 \).

\( \Delta H^\circ_{rxn} = \sum \Delta H^\circ_f [products] - \sum \Delta H^\circ_f [reactants] \)
Bond Energies
Bond Energy and Reaction Enthalpy
During a chemical reaction, bonds are broken and new bonds are formed. The energy required to break bonds is absorbed, while the energy released during bond formation is evolved as heat.
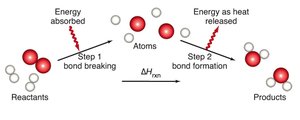
Average bond energy is the energy required to break a specific type of bond in a molecule.
Estimating \( \Delta H_{rxn} \) Using Bond Energies
To estimate the enthalpy change of a reaction using bond energies:
\( \Delta H_{rxn} \approx \text{(energy required to break all bonds in reactants)} - \text{(energy released in forming bonds in products)} \)

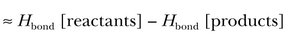
\( \Delta H_{rxn} \approx H_{bond}[reactants] - H_{bond}[products] \)
Summary Table: Key Thermochemistry Terms
Term | Definition |
|---|---|
Energy | Ability to cause change in a system |
Kinetic Energy | Energy of motion |
Potential Energy | Energy of position/composition |
System | Chemicals in the reaction |
Surroundings | Everything else |
Internal Energy (E) | Total kinetic and potential energy of system |
Enthalpy (H) | Heat content at constant pressure |
Work (w) | Energy transfer via force and distance |
Heat (q) | Energy transfer via temperature change |
State Function | Depends only on current state, not path |
Path Function | Depends on how state is reached |
Heat Capacity (c) | Heat needed to raise temp by 1°C |
Specific Heat (Csp) | Heat needed to raise 1g by 1°C |
Molar Heat Capacity (C) | Heat needed to raise 1 mol by 1°C |
Standard Enthalpy of Formation (\( \Delta H^\circ_f \)) | Enthalpy change for forming 1 mol from elements in standard states |
Bond Energy | Energy required to break a bond |
