 Back
BackCHEM 1113: Covalent Bonding, Molecular Shape, and Intermolecular Forces Study Guide
Study Guide - Smart Notes

Q5. Which of these Lewis structures are incorrect?
Background
Topic: Lewis Structures and the Octet Rule
This question tests your ability to evaluate Lewis structures for correctness, focusing on the octet rule, formal charges, and proper electron placement.
Key Terms and Concepts:
Lewis Structure: A diagram showing the arrangement of valence electrons around atoms in a molecule.
Octet Rule: Most main-group elements tend to form bonds until they are surrounded by eight valence electrons.
Formal Charge: The charge assigned to an atom in a Lewis structure, calculated as:
Step-by-Step Guidance
Examine each Lewis structure (1, 2, and 3) for the following:
Does each atom (especially central atoms) follow the octet rule?
Are all valence electrons accounted for (including lone pairs)?
Are formal charges minimized and assigned correctly?
For each structure, count the total number of valence electrons and compare to the expected total for the molecule or ion.
Check if any atom (especially central atoms like S, N, or I) has more or fewer than 8 electrons, unless it is an exception (e.g., expanded octet for elements in period 3 or higher).
Assign formal charges to each atom in the structure and see if the most stable (lowest energy) arrangement is shown.
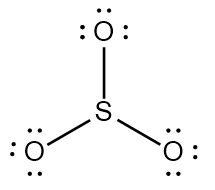
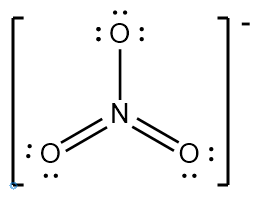
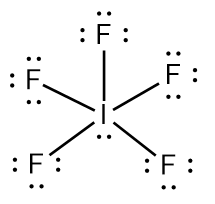
Try solving on your own before revealing the answer!
Final Answer: B. 2 and 3 are incorrect
Structure 2 (NO3-) does not show all resonance forms or the correct number of electrons for the ion, and structure 3 (IF4-) does not properly account for the lone pairs on the central atom, violating the octet rule for iodine.
Structure 1 (SO3) is correct, as sulfur can have an expanded octet and all atoms have appropriate formal charges.
