 Back
BackCHEM 172 Exam I – General Chemistry Equilibrium, Thermodynamics, and Solutions Guidance
Study Guide - Smart Notes

Q8. Which of the following is true? The critical point...
Background
Topic: Phase Diagrams and Critical Point
This question tests your understanding of phase diagrams, specifically the meaning and location of the critical point, and how it relates to the different phases of matter (solid, liquid, vapor).
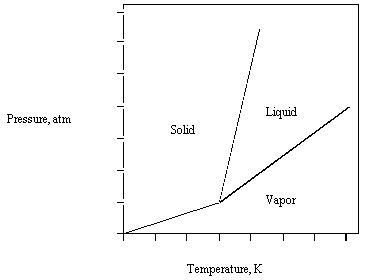
Key Terms and Concepts:
Phase Diagram: A graphical representation showing the stability of phases of a substance as a function of temperature and pressure.
Critical Point: The end point of the liquid-vapor equilibrium curve, beyond which the liquid and vapor phases become indistinguishable.
Triple Point: The unique set of conditions where all three phases (solid, liquid, vapor) coexist in equilibrium.
Step-by-Step Guidance
Examine the phase diagram and identify the regions labeled as solid, liquid, and vapor. Notice where the boundaries between these phases meet.
Recall that the critical point is found at the end of the liquid-vapor boundary, where the distinction between liquid and vapor disappears.
Consider the definitions of the options provided: (a) where the liquid-vapor boundary crosses standard pressure, (b) where vapor reaches the same density as liquid, (c) where all three phase boundaries meet.
Think about which of these options matches the definition of the critical point, based on your understanding of phase diagrams and the behavior of substances at high temperature and pressure.
Try solving on your own before revealing the answer!
Final Answer: (b) is where the vapor reaches the same density as a liquid.
At the critical point, the properties of the liquid and vapor phases become identical, and there is no longer a distinction between them.
