 Back
BackChem121A Exam 1 Study Guide: Matter, Atoms, and Chemical Reactions
Study Guide - Smart Notes

Chapter 1: Matter and Measurements
Physical States of Matter
The three physical states of matter—solid, liquid, and gas—are fundamental to understanding chemical substances. Each state has unique properties, but all are composed of atoms and molecules.
Solid: Definite shape and volume; particles are closely packed and vibrate in place.
Liquid: Definite volume but no definite shape; particles are less tightly packed and can move past each other.
Gas: No definite shape or volume; particles are far apart and move freely.
Similarity: All states are made of atoms/molecules.
Difference: Arrangement and movement of particles.
Classification of Matter
Matter can be classified as pure substances or mixtures.
Pure Substances:
Elements: Cannot be broken down further (e.g., O2, Fe).
Compounds: Composed of two or more elements chemically combined (e.g., H2O).
Mixtures:
Homogeneous: Uniform composition (e.g., salt water).
Heterogeneous: Non-uniform composition (e.g., sand and water).
Chemical vs. Physical Changes
Understanding the difference between chemical and physical changes is essential for identifying reactions and processes.
Chemical Change: Alters the chemical composition (e.g., rusting of iron).
Physical Change: Alters physical form but not composition (e.g., melting ice).
Example: Burning wood (chemical), dissolving sugar in water (physical).
Intensive vs. Extensive Properties
Properties of matter are classified as intensive or extensive.
Intensive Properties: Independent of amount (e.g., density, boiling point).
Extensive Properties: Dependent on amount (e.g., mass, volume).
Example: Density is intensive; mass is extensive.
SI Base Units and Metric Prefixes
The International System of Units (SI) provides standard units for scientific measurements.
Length: Meter (m)
Mass: Kilogram (kg)
Temperature: Kelvin (K)
Time: Second (s)
Amount: Mole (mol)
Metric prefixes modify base units to represent multiples or fractions.
Example: 1 milligram (mg) = grams (g)
Common Prefixes: Mega (M), kilo (k), centi (c), milli (m), micro (μ), nano (n), pico (p)
Temperature Conversions
Temperature can be converted between Celsius, Fahrenheit, and Kelvin using formulas:
Precision, Accuracy, and Significant Figures
Measurements must be reported with the correct number of significant figures to reflect precision and accuracy.
Precision: Consistency of repeated measurements.
Accuracy: Closeness to the true value.
Significant Figures: Digits that carry meaning in a measurement.
Rules:
All nonzero digits are significant.
Zeros between nonzero digits are significant.
Leading zeros are not significant.
Trailing zeros are significant if after a decimal point.
Rounding:
Multiplication/Division: Round to the least number of sig figs.
Addition/Subtraction: Round to the least number of decimal places.
Scientific Notation and Dimensional Analysis
Scientific notation is used to express very large or small numbers. Dimensional analysis is a method for converting units using conversion factors.
Example: nm = m
Dimensional Analysis: Multiply by conversion factors to cancel units.
Chapter 2: Atoms, Molecules, and Ions
Subatomic Particles
Atoms are composed of three subatomic particles:
Proton: Positive charge, found in nucleus.
Neutron: Neutral charge, found in nucleus.
Electron: Negative charge, found outside nucleus; smallest mass.
Isotopes and Atomic Mass
Isotopes are atoms of the same element with different numbers of neutrons.
Average Atomic Mass: Weighted average of isotopes.
Formula:
Example: If an element has two isotopes, calculate using their masses and abundances.
Atomic Number and Atomic Mass
Atomic Number (Z): Number of protons; determines element identity.
Atomic Mass: Sum of protons and neutrons.
Neutral Atom: Number of electrons equals number of protons.
Atomic Symbols and Ions
Atomic symbols provide information about the element, its atomic number, and mass. Ions are atoms with a net charge due to loss or gain of electrons.
Cation: Positive ion; electrons lost.
Anion: Negative ion; electrons gained.
Variable-Charge Ions: Transition metals can have multiple charges; indicated by Roman numerals in names.
Empirical and Molecular Formulas
Empirical formulas show the simplest ratio of elements; molecular formulas show the actual number of atoms.
Example: Molecular formula C6H12O6; empirical formula CH2O.
Monoatomic and Polyatomic Ions
Monoatomic ions are single atoms with a charge; polyatomic ions are groups of atoms with a charge.
Example: Na+ (monoatomic), SO42- (polyatomic).
Naming Ionic and Molecular Compounds
Different rules apply for naming ionic and molecular compounds.
Ionic Compounds:
Binary: Metal + nonmetal (e.g., NaCl).
Polyatomic: Contains polyatomic ions (e.g., Na2SO4).
Variable-charge: Use Roman numerals (e.g., FeCl2 is iron(II) chloride).
Molecular Compounds:
Use prefixes to indicate number of atoms (e.g., CO2 is carbon dioxide).
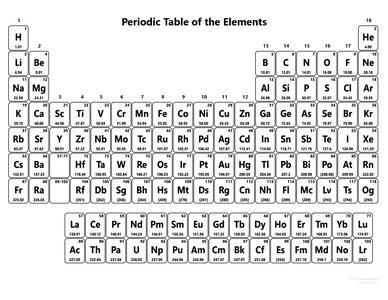
Periodic Table and Element Classification
The periodic table organizes elements by atomic number and groups elements with similar properties together. Metals and nonmetals are distinguished by their location and properties.
Groups: Vertical columns; elements in a group share chemical properties.
Metals: Left and center; conduct electricity, malleable.
Nonmetals: Right; poor conductors, brittle.
Chapter 3: Chemical Reactions and Stoichiometry
Law of Conservation of Mass
The law of conservation of mass states that mass is neither created nor destroyed in a chemical reaction.
Example: Total mass of reactants equals total mass of products.
Balancing Chemical Equations
Balancing equations ensures the same number of atoms of each element on both sides.
Steps:
Write the unbalanced equation.
Count atoms of each element.
Add coefficients to balance.
Types of Chemical Reactions
Three broad classes of reactions:
Combination: Two or more substances form one product.
Decomposition: One substance breaks into two or more products.
Combustion: Substance reacts with oxygen, producing heat and often CO2 and H2O.
Formula Weight and Percent Composition
Formula weight (FW) is the sum of atomic masses in a compound; percent composition is the percentage of each element.
Formula Weight:
Percent Composition:
The Mole and Molar Mass
The mole is a counting unit for atoms/molecules; Avogadro’s number is particles per mole.
Molar Mass: Mass of one mole of a substance (g/mol).
Conversion: Use molar mass to convert between grams and moles.
Stoichiometry and Limiting Reagents
Stoichiometry involves calculations based on balanced equations to determine amounts of reactants and products.
Molar Ratios: Coefficients in equations indicate ratios.
Limiting Reagent: Reactant that determines the maximum amount of product.
Theoretical Yield: Maximum possible product.
Percent Yield:
Empirical Formula Determination
Empirical formulas are determined from the amounts of each element in a compound.
Steps:
Convert mass to moles.
Divide by smallest number of moles.
Multiply to get whole numbers if needed.
Dimensional Analysis in Stoichiometry
Dimensional analysis is used to convert between units (grams, moles, molecules) in stoichiometry problems.
Example: Convert grams of reactant to moles, use molar ratio, convert to grams of product.
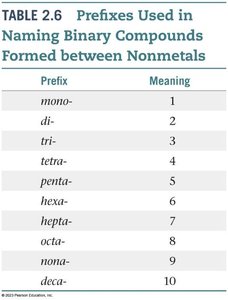
Summary Table: Prefixes Used in Naming Binary Compounds
Prefixes are used to indicate the number of atoms in molecular compounds:
Prefix | Meaning |
|---|---|
mono- | 1 |
di- | 2 |
tri- | 3 |
tetra- | 4 |
penta- | 5 |
hexa- | 6 |
hepta- | 7 |
octa- | 8 |
nona- | 9 |
deca- | 10 |
Summary Table: Periodic Table of the Elements
The periodic table is a reference for atomic numbers, element symbols, and atomic masses.

Additional info: Tables and images included are directly relevant to naming rules and element classification. Academic context has been expanded for clarity and completeness.
