 Back
BackChemical Bonding I: Lewis Structures, Electronegativity, and Molecular Geometry
Study Guide - Smart Notes

Lewis Theory & Lewis Structures
Lewis Symbols
The Lewis symbol is a simple representation of an atom's valence electrons. The chemical symbol represents the nucleus and core electrons, while dots around the symbol represent valence electrons. For main-group elements, the number of valence electrons equals the group number for s- and p-block elements.
Example: Nitrogen (N), Phosphorus (P), Arsenic (As), Antimony (Sb), Bismuth (Bi) all have five valence electrons, so their Lewis symbols have five dots.
Aluminum (Al) is in group 13 (3 dots), Selenium (Se) in group 16 (6 dots), Argon (Ar) in group 18 (8 dots).

Lewis Structures of Ionic Compounds
Lewis structures can also represent the transfer of electrons in ionic compounds. The approach is to write the Lewis symbols and determine how many electrons each atom must gain or lose to acquire a noble-gas configuration.
Example: In BaCl2, Ba loses two electrons (forming Ba2+), and each Cl gains one electron (forming two Cl-).
In MgCl2, Mg loses two electrons (Mg2+), and two Cl atoms each gain one electron.
In Al2O3, two Al atoms each lose three electrons (Al3+), and three O atoms each gain two electrons (O2-).

Writing Simple Lewis Structures
To write a Lewis structure for a molecule, count the number of valence electrons for each atom and arrange them to satisfy the octet rule (except for hydrogen, which requires only two electrons).
Example: Ammonia (NH3): N has 5 valence electrons, each H has 1. The structure is assembled so that N has an octet and each H has two electrons.

Covalent Bonding and Electronegativity
Bond Polarity and Electronegativity
Electronegativity (EN) is the ability of an atom in a molecule to attract electrons toward itself. The difference in EN between two atoms determines the bond type:
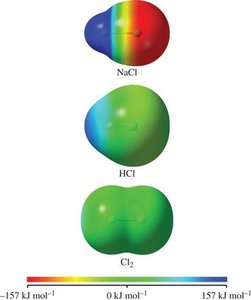
Nonpolar covalent bond: Electrons are shared equally (e.g., Cl2).
Polar covalent bond: Electrons are shared unequally (e.g., HCl).
Ionic bond: Electrons are transferred (e.g., NaCl).
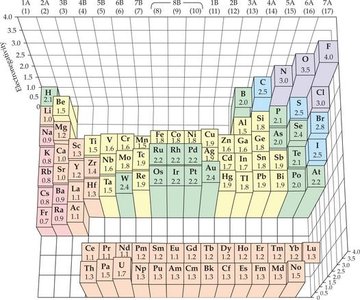
Electronegativity Trends
Electronegativity increases across a period and decreases down a group in the periodic table. Fluorine is the most electronegative element.
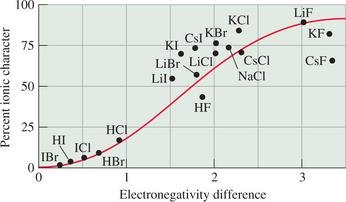
Percent Ionic Character
The percent ionic character of a bond increases with the difference in electronegativity between the two atoms. Bonds with a large EN difference are more ionic in character.
Assessing Electronegativity Differences and Bond Polarity
To determine which bond is more polar, compare the EN values of the atoms involved.
Percent ionic character can be estimated from EN difference using reference graphs.
Example: H–Cl bond is more polar than H–O; H–Cl is about 18% ionic, H–O about 39% ionic.

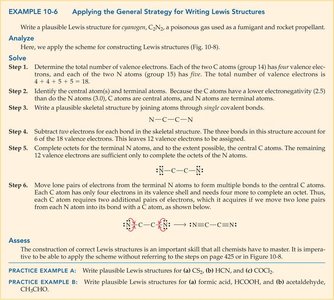
Writing Lewis Structures: General Strategy
Stepwise Approach
Count total valence electrons.
Draw a skeletal structure, connecting atoms with single bonds.
Complete octets for terminal atoms, then central atoms.
Place remaining electrons as lone pairs on central atom if needed.
If octets are incomplete, form multiple bonds as necessary.
Example: Cyanogen (C2N2): Place N atoms at ends, C atoms in the center, connect with triple bonds to satisfy octets.
Lewis Structures for Polyatomic Ions
For polyatomic ions, add or subtract electrons according to the ion's charge. Enclose the structure in brackets and indicate the charge.
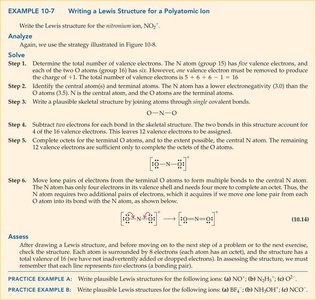
Example: Nitronium ion (NO2+): Count electrons, draw skeletal structure, complete octets, and adjust for charge.
Formal Charge
Formal charge (FC) is used to determine the most stable Lewis structure. It is calculated as:
The sum of formal charges must equal the overall charge of the molecule or ion.
The best Lewis structure has the smallest formal charges and places negative charges on the most electronegative atoms.

Resonance Structures
Some molecules cannot be represented by a single Lewis structure. Resonance structures are multiple valid Lewis structures for the same molecule, differing only in the placement of electrons. The actual structure is a resonance hybrid.
Example: Acetate ion (CH3COO-): Two resonance structures with the negative charge delocalized over two oxygen atoms.

VSEPR Theory and Molecular Geometry
Valence-Shell Electron Pair Repulsion (VSEPR) Theory
VSEPR theory predicts the shapes of molecules based on the repulsion between electron pairs (bonding and lone pairs) around a central atom. Electron pairs arrange themselves to minimize repulsion, determining the molecular geometry.
Electron group geometry: Arrangement of all electron groups (bonding and lone pairs).
Molecular geometry: Arrangement of only the atoms (nuclei).

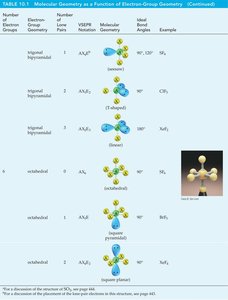
Common Molecular Geometries
Number of Electron Groups | Electron-Group Geometry | Molecular Geometry | Bond Angles | Example |
|---|---|---|---|---|
2 | Linear | Linear | 180° | BeCl2 |
3 | Trigonal planar | Trigonal planar | 120° | BF3 |
4 | Tetrahedral | Tetrahedral | 109.5° | CH4 |
5 | Trigonal bipyramidal | Trigonal bipyramidal | 90°, 120° | PCl5 |
6 | Octahedral | Octahedral | 90° | SF6 |

Predicting Molecular Shapes with VSEPR
To predict the shape of a molecule:
Draw the Lewis structure.
Count the number of electron groups around the central atom.
Determine the electron-group geometry.
Determine the molecular geometry by considering only the positions of the atoms.
Multiple Central Atoms
For molecules with more than one central atom, determine the geometry around each central atom separately and describe the overall shape.

Molecular Polarity and Dipole Moments
Bond Dipoles and Molecular Dipole Moments
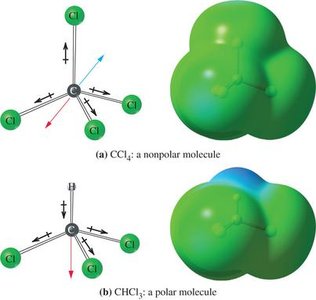
A dipole moment arises when there is a separation of charge in a molecule. The overall molecular dipole is the vector sum of individual bond dipoles. Symmetrical molecules may have polar bonds but no net dipole moment if the bond dipoles cancel.
Example: CCl4 is nonpolar (symmetrical), CHCl3 is polar (asymmetrical).
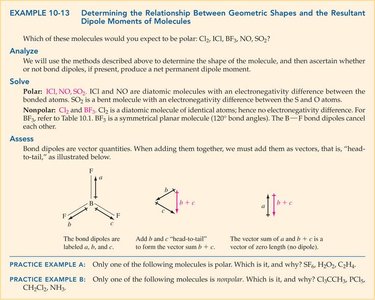
Relationship Between Shape and Dipole Moment
To determine if a molecule is polar, consider both the polarity of individual bonds and the molecular geometry. If the vector sum of bond dipoles is nonzero, the molecule is polar.
Bond Order, Bond Length, and Bond Energy
Bond Order
Bond order is the number of chemical bonds between a pair of atoms. Single bond = 1, double bond = 2, triple bond = 3. Higher bond order generally means a shorter and stronger bond.
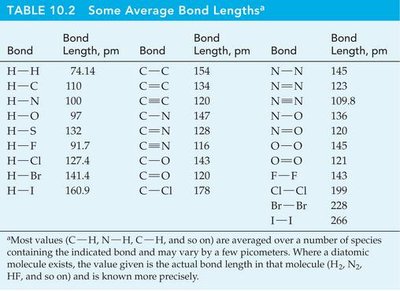
Bond Length
The bond length is the distance between the nuclei of two bonded atoms. It can be estimated as the sum of the covalent radii of the two atoms.
Bond | Bond Length (pm) |
|---|---|
H–H | 74.1 |
C–C | 154 |
C=C | 134 |
C≡C | 120 |
N–N | 145 |
O–O | 145 |
F–F | 142 |
Bond Energy
Bond energy is the energy required to break one mole of a bond in a molecule in the gas phase. It is a measure of bond strength. The enthalpy change for a reaction can be estimated using bond energies:
Breaking bonds requires energy (endothermic, positive ΔH).
Forming bonds releases energy (exothermic, negative ΔH).
Summary
This guide covers the foundational concepts of chemical bonding, including Lewis structures, electronegativity, molecular geometry, and bond properties. Mastery of these topics is essential for understanding the structure and reactivity of molecules in general chemistry.
