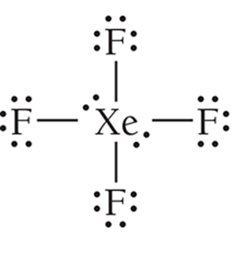Back
BackChemical Bonding II: Molecular Shapes, Valence Bond Theory, and Molecular Orbital Theory
Study Guide - Smart Notes

Chemical Bonding II: Molecular Shapes, Valence Bond Theory, and Molecular Orbital Theory
Introduction to Molecular Shapes and Bonding Theories
This chapter explores advanced concepts in chemical bonding, focusing on how molecules adopt three-dimensional shapes, the theoretical models used to describe these shapes, and the implications for molecular properties. Understanding these concepts is essential for predicting molecular behavior, reactivity, and physical properties.

Valence Shell Electron Pair Repulsion (VSEPR) Theory
Basic Principles of VSEPR Theory
VSEPR theory is a model used to predict the geometry of molecules based on the repulsion between electron groups around a central atom. Electron groups include lone pairs, single bonds, double bonds, and triple bonds. The theory states that these groups arrange themselves in three dimensions to minimize repulsion, resulting in specific molecular shapes.
Electron group: Any region of electron density (bond or lone pair) around a central atom.
Repulsion: Electron groups repel each other due to electrostatic forces.

Counting Electron Groups
To determine the geometry, count the number of electron groups around the central atom. Each bond (single, double, triple) and lone pair counts as one group.
Electronic and Molecular Geometries
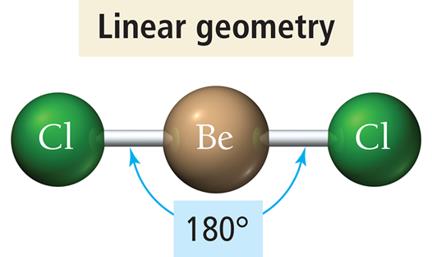
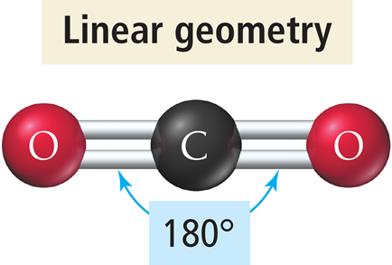
Linear Geometry (2 Electron Groups)
When two electron groups surround a central atom, they adopt a linear geometry with a bond angle of 180°.
Example: BeCl2 and CO2
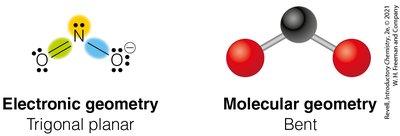
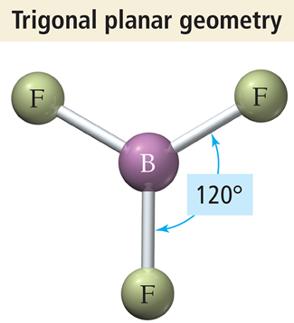
Trigonal Planar Geometry (3 Electron Groups)
Three electron groups arrange themselves in a flat triangle, resulting in trigonal planar geometry with bond angles of 120°.
Example: BF3 and formaldehyde (H2CO)

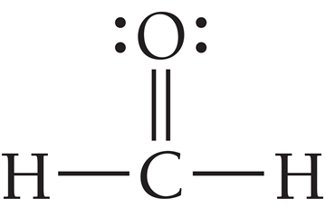
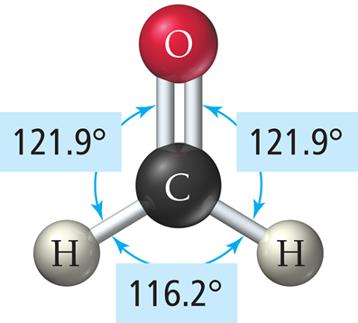
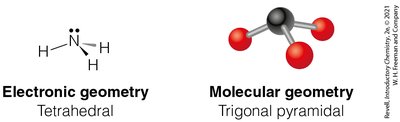
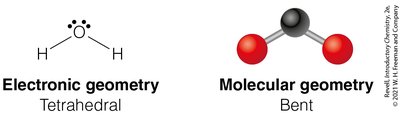
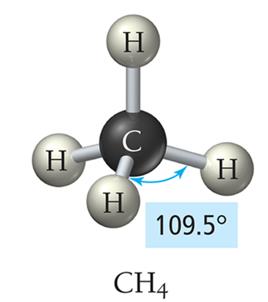
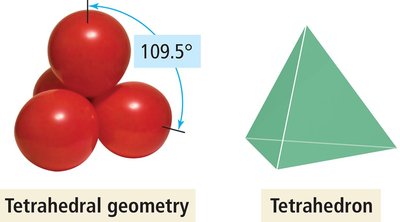
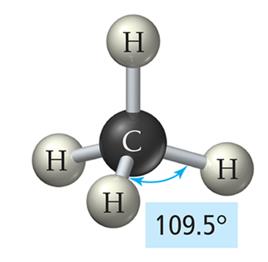
Tetrahedral Geometry (4 Electron Groups)
Four electron groups form a tetrahedral geometry with bond angles of 109.5°.
Example: Methane (CH4)
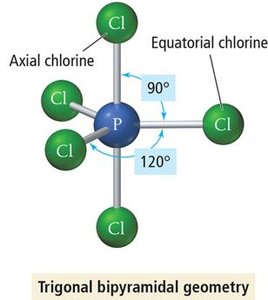
Trigonal Bipyramidal Geometry (5 Electron Groups)
Five electron groups form a trigonal bipyramidal geometry. Three groups lie in a plane (equatorial), and two are above and below (axial). Bond angles are 120° (equatorial) and 90° (axial).
Example: PCl5
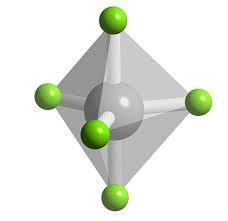
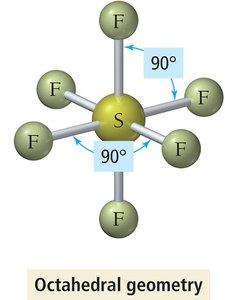
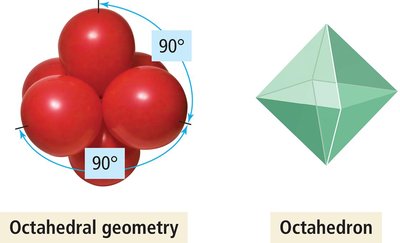
Octahedral Geometry (6 Electron Groups)
Six electron groups form an octahedral geometry with all bond angles at 90°.
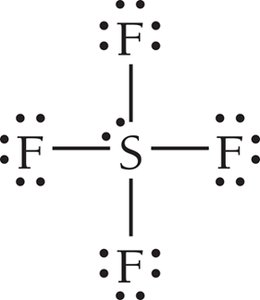
Example: SF6
Summary Table: Electron and Molecular Geometries
The following table summarizes the main electronic and molecular geometries, bond angles, and examples:
Electron Groups | Bonding Groups | Lone Pairs | Electron Geometry | Molecular Geometry | Approximate Bond Angles | Example | Example |
|---|---|---|---|---|---|---|---|
2 | 2 | 0 | Linear | Linear | 180° | O=C=O |
|
3 | 3 | 0 | Trigonal Planar | Trigonal Planar | 120° | BF3 |
|
3 | 2 | 1 | Trigonal Planar | Bent | <120° | NO2- |
|
4 | 4 | 0 | Tetrahedral | Tetrahedral | 109.5° | CH4 |
|
4 | 3 | 1 | Tetrahedral | Trigonal Pyramidal | <109.5° | NH3 |
|
4 | 2 | 2 | Tetrahedral | Bent | <109.5° | H2O |
|
5 | 5 | 0 | Trigonal Bipyramidal | Trigonal Bipyramidal | 120° (eq), 90° (ax) | PCl5 |
|
6 | 6 | 0 | Octahedral | Octahedral | 90° | SF6 |
|
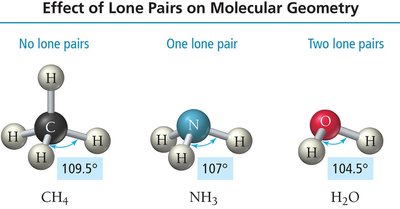
Effect of Lone Pairs on Molecular Geometry
Lone Pair Repulsion and Geometry Distortion
Lone pairs occupy more space than bonding pairs, causing greater repulsion and distortion of bond angles. This leads to variations in molecular geometry compared to electronic geometry.
Trigonal pyramidal: 3 bonding groups, 1 lone pair (e.g., NH3)
Bent: 2 bonding groups, 2 lone pairs (e.g., H2O)
Representing 3D Molecular Shapes
Drawing Conventions
To represent three-dimensional shapes on paper, chemists use straight lines for bonds in the plane, solid wedges for bonds coming out of the page, and hatched wedges for bonds going into the page.

Molecular Polarity
Determining Molecular Polarity
Molecular polarity depends on both the presence of polar bonds and the overall shape of the molecule. If the polar bonds do not cancel out, the molecule is polar and has a net dipole moment.
Polar molecules: Have a net dipole moment (e.g., H2O)
Nonpolar molecules: No net dipole moment (e.g., CO2)
Valence Bond Theory and Hybridization
Valence Bond Theory
Valence bond theory describes chemical bonding as the overlap of atomic or hybrid orbitals, resulting in shared electron pairs. The geometry of overlapping orbitals determines the shape of the molecule.
Hybridization: Mixing of atomic orbitals to form new hybrid orbitals with specific shapes and energies.
Types of hybridization: sp, sp2, sp3, sp3d, sp3d2
Example: Carbon in methane (CH4) undergoes sp3 hybridization, resulting in a tetrahedral geometry.
Sigma and Pi Bonds
Single bonds are sigma (σ) bonds, formed by end-to-end overlap. Double and triple bonds contain one sigma bond and one or two pi (π) bonds, formed by side-to-side overlap of p orbitals.
Sigma (σ) bond: Stronger, allows free rotation.
Pi (π) bond: Weaker, restricts rotation.
Molecular Orbital (MO) Theory
Basic Concepts of MO Theory
Molecular orbital theory applies quantum mechanics to molecules, describing electrons as occupying molecular orbitals that extend over the entire molecule. These orbitals are formed by the linear combination of atomic orbitals (LCAO).
Bonding orbitals: Lower energy, stabilize the molecule.
Antibonding orbitals: Higher energy, destabilize the molecule.
Bond Order Calculation
Bond order is a measure of bond strength and stability, calculated as:
Bond order > 0: Stable molecule
Bond order = 0: Unstable, molecule does not form
Paramagnetic vs. Diamagnetic Molecules
MO theory predicts magnetic properties based on electron pairing:
Paramagnetic: Unpaired electrons, attracted to magnetic fields (e.g., O2)
Diamagnetic: All electrons paired, not attracted to magnetic fields
Summary Table: Electron and Molecular Geometries (Expanded)
Electron Groups | Bonding Groups | Lone Pairs | Electron Geometry | Molecular Geometry | Approximate Bond Angles | Example | Example |
|---|---|---|---|---|---|---|---|
2 | 2 | 0 | Linear | Linear | 180° | CO2 |
|
3 | 3 | 0 | Trigonal Planar | Trigonal Planar | 120° | BF3 |
|
3 | 2 | 1 | Trigonal Planar | Bent | <120° | NO2- |
|
4 | 4 | 0 | Tetrahedral | Tetrahedral | 109.5° | CH4 |
|
4 | 3 | 1 | Tetrahedral | Trigonal Pyramidal | <109.5° | NH3 |
|
4 | 2 | 2 | Tetrahedral | Bent | <109.5° | H2O |
|
5 | 5 | 0 | Trigonal Bipyramidal | Trigonal Bipyramidal | 120° (eq), 90° (ax) | PCl5 |
|
5 | 4 | 1 | Trigonal Bipyramidal | Seesaw | <120° (eq), <90° (ax) | SF4 |
|
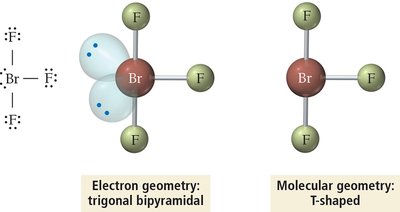
5 | 3 | 2 | Trigonal Bipyramidal | T-shaped | <90° | BrF3 |
|
5 | 2 | 3 | Trigonal Bipyramidal | Linear | 180° | XeF2 |
|
6 | 6 | 0 | Octahedral | Octahedral | 90° | SF6 |
|
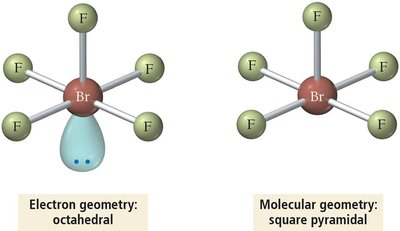
6 | 5 | 1 | Octahedral | Square Pyramidal | <90° | BrF5 |
|
6 | 4 | 2 | Octahedral | Square Planar | 90° | XeF4 |
|
Key Points to Memorize
Names and bond angles of all electronic and molecular geometries
How to identify geometry from Lewis structures
Effect of lone pairs on bond angles and molecular shape
Types of hybridization and their associated geometries
Bond order calculation and its significance