 Back
BackChemical Bonds, Electronegativity, and Covalent Compounds: Study Notes
Study Guide - Smart Notes

Chemical Bonds and the Octet Rule
Introduction to Chemical Bonding
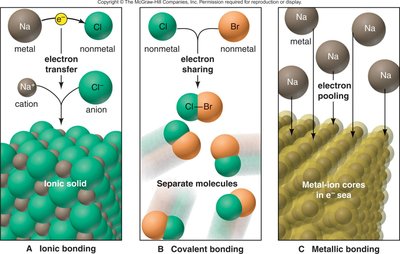
Chemical bonds are the attractive forces that hold atoms together in compounds. Atoms form bonds to achieve a stable electron configuration, often resembling the nearest noble gas (the octet rule). There are three primary types of chemical bonds: ionic, covalent, and metallic.
Ionic bonds: Involve the transfer of electrons from one atom (usually a metal) to another (usually a nonmetal), resulting in the formation of cations and anions.
Covalent bonds: Involve the sharing of electron pairs between nonmetal atoms.
Metallic bonds: Involve the pooling of electrons among a lattice of metal atoms.
Electronegativity and Covalent Bonds
Electronegativity: Definition and Trends
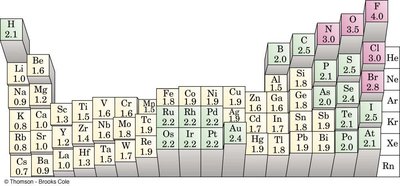
Electronegativity (EN) is the tendency of an atom to attract electrons in a chemical bond. It is a key factor in determining bond polarity and the type of bond formed between atoms.
Electronegativity increases across a period (left to right) and decreases down a group (top to bottom) in the periodic table.
Fluorine (F) is the most electronegative element; noble gases generally do not have EN values because they rarely form bonds.
Bond Polarity and Dipole Moments
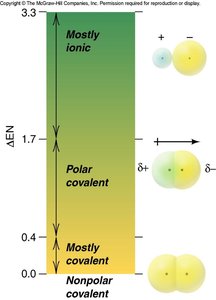
The difference in electronegativity (ΔEN) between two bonded atoms determines the bond's polarity:
Nonpolar covalent bond: ΔEN ≤ 0.4 (equal sharing of electrons)
Polar covalent bond: 0.4 < ΔEN ≤ 2.0 (unequal sharing of electrons)
Ionic bond: ΔEN > 2.0 (electron transfer)
A dipole moment (μ) is a measure of the separation of positive and negative charges in a molecule. It is calculated as:
where Q = charge, r = separation distance
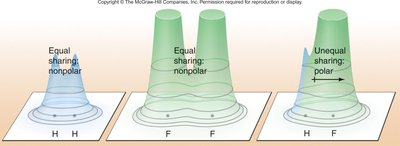
Electron Density and Molecular Polarity
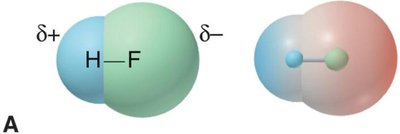
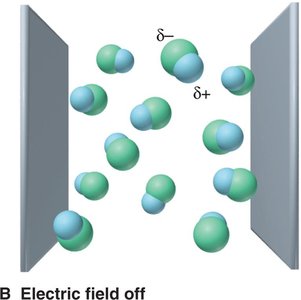
Electron density maps show how electrons are distributed in molecules. In polar bonds, electron density is shifted toward the more electronegative atom, creating partial charges (δ+ and δ−).
Lewis Structures and the Octet Rule
Drawing Lewis Structures
Lewis structures (or electron dot structures) represent the valence electrons of atoms within a molecule. They help visualize bonding and lone pairs.
Dots represent valence electrons.
Lines represent shared electron pairs (bonds).
Follow the octet rule: atoms (except H and He) tend to have 8 electrons in their valence shell.
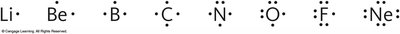

Steps for Constructing Lewis Structures
Find the total number of valence electrons by summing the group numbers of all atoms.
Place the least electronegative atom in the center (except hydrogen).
Connect atoms with single bonds, then complete the octets of surrounding atoms.
Place remaining electrons on the central atom. If the central atom lacks an octet, form double or triple bonds as needed (except for Be, B, F, and metals).
Calculate formal charges to determine the most reasonable structure.
Exceptions to the Octet Rule
Incomplete octet: Some elements (e.g., B, Be) can have fewer than 8 electrons.
Odd-electron species: Molecules with an odd number of electrons (e.g., NO).
Expanded octet: Elements in period 3 or higher can have more than 8 electrons due to available d orbitals.
Resonance Structures
Some molecules cannot be represented by a single Lewis structure. Resonance structures are two or more valid Lewis structures for the same molecule, with electrons delocalized over multiple atoms. The actual structure is a resonance hybrid.
Formal Charge
Formal charge helps identify the most stable Lewis structure. It is calculated as:
Structures with the smallest formal charges (closest to zero) are preferred.
Negative formal charges should reside on the most electronegative atoms.
Naming Covalent Compounds
Rules for Naming Binary Covalent Compounds
Binary covalent compounds are composed of two nonmetals. The naming rules are:
Name the less electronegative element first.
Name the second element with the root and the suffix –ide.
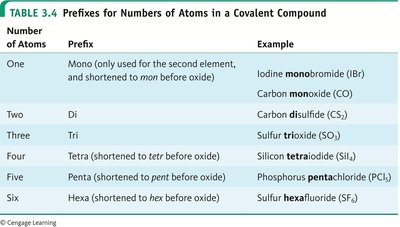
Use numerical prefixes to indicate the number of atoms (mono-, di-, tri-, etc.), but do not use "mono-" for the first element.
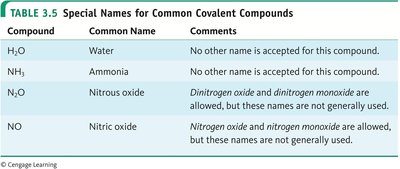
Special Names for Common Covalent Compounds
Certain covalent compounds have common names that are accepted universally, such as water (H2O) and ammonia (NH3).
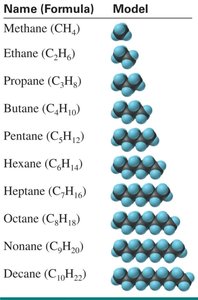
Naming Hydrocarbons
Hydrocarbons are compounds containing only carbon and hydrogen. The simplest type, alkanes, are named with a root indicating the number of carbons and the suffix –ane.
Recognizing Ionic and Molecular Compounds
Distinguishing Features
Ionic and molecular (covalent) compounds can be distinguished by their composition and properties:
Ionic compounds: Contain a metal and a nonmetal, or contain polyatomic ions (e.g., NH4+).
Molecular compounds: Contain only nonmetals.

Comparison Table: Ionic vs. Covalent Compounds
Property | Ionic Compounds | Covalent Compounds |
|---|---|---|
Type of elements | Metal and nonmetal | Nonmetals only |
Bonding | Ionic (attraction between anions and cations) | Covalent (sharing of electrons) |
Representative unit | Formula unit | Molecule |
Physical state at room temperature | Solid | Gas, liquid, or solid |
Water solubility | Usually high | Variable |
Melting and boiling points | Generally high | Generally low |
Electrical conductivity | Good when molten or in solution | Poor |
State when dissolved in water | Separates into ions | Remains molecules |
Summary
This guide covers the essential concepts of chemical bonding, electronegativity, Lewis structures, resonance, formal charge, and the systematic naming and recognition of covalent and ionic compounds. Mastery of these topics is fundamental for success in general chemistry.
