 Back
BackChemical Equilibrium, Acids, Bases, and Salt Solutions: Exam 3 Study Guide
Study Guide - Smart Notes

Chemical Equilibrium
Equilibrium Constants and Expressions
Chemical equilibrium occurs when the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products. The equilibrium constant quantifies the ratio of product to reactant concentrations at equilibrium.
Equilibrium Constant (K): Represents the ratio of concentrations (Kc) or partial pressures (Kp) of products to reactants at equilibrium.
Writing Kc and Kp: Only gases and aqueous species are included; pure solids and liquids are excluded.
Homogeneous Equilibria: All reactants and products are in the same phase.
Heterogeneous Equilibria: Reactants and products are in different phases.
General Expression:
Interpreting the Value of K
The magnitude of the equilibrium constant indicates the position of equilibrium:
K > 1: Products are favored; equilibrium lies to the right.
K < 1: Reactants are favored; equilibrium lies to the left.
K ≈ 1: Significant amounts of both reactants and products are present.
Calculating Equilibrium Constants
To calculate Kc or Kp, substitute equilibrium concentrations or partial pressures into the equilibrium expression.
Example: For the reaction , use measured equilibrium values.
Reaction Quotient (Q) and Predicting Direction
The reaction quotient (Q) uses current concentrations or pressures to predict the direction of the reaction:
Q < K: Reaction proceeds toward products.
Q > K: Reaction proceeds toward reactants.
Q = K: System is at equilibrium.
Manipulating Equilibrium Constants
Equilibrium constants can be adjusted for related reactions:
Reversing a reaction: Take the reciprocal of K.
Multiplying a reaction by a factor: Raise K to that power.
Adding reactions: Multiply the K values.
Le Châtelier’s Principle
Le Châtelier’s principle states that a system at equilibrium will shift to counteract changes:
Concentration: Adding/removing reactants or products shifts equilibrium to oppose the change.
Pressure/Volume: For gases, increasing pressure shifts equilibrium toward fewer gas molecules; decreasing pressure shifts it toward more gas molecules.
Temperature: Increasing temperature favors the endothermic direction; decreasing temperature favors the exothermic direction. Temperature changes affect both equilibrium position and K.
Catalysts: Speed up attainment of equilibrium but do not affect its position.
Inert Gas Addition: At constant volume, does not affect equilibrium position.
Calculating Equilibrium Concentrations
Given Kc or Kp and initial conditions, use an ICE table (Initial, Change, Equilibrium) to solve for unknown concentrations or pressures.
"x is small" Assumption: Used when K is very small and initial concentration is much larger than x. Neglect x in calculations to simplify algebra.
Check Validity: If is less than 5%, the assumption is valid.
Acids and Bases
Strong Acids and Strong Bases
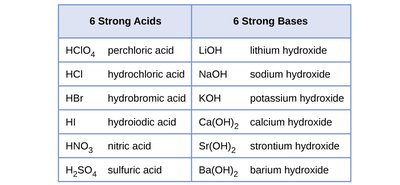
Strong acids and bases dissociate completely in water. Knowing common strong acids and bases is essential for predicting solution behavior.
Strong Acids: HClO4, HCl, HBr, HI, HNO3, H2SO4
Strong Bases: LiOH, NaOH, KOH, Ca(OH)2, Sr(OH)2, Ba(OH)2
Acid and Base Definitions
Acids and bases can be defined in two main ways:
Arrhenius Definition: Acids produce H+ (or H3O+) in water; bases produce OH-.
Brønsted–Lowry Definition: Acids are proton donors; bases are proton acceptors.
Example: NH3 is a Brønsted–Lowry base but not an Arrhenius base.
Conjugate Acid–Base Pairs
Conjugate pairs differ by one proton:
Conjugate Base: What remains after an acid donates a proton.
Conjugate Acid: What forms when a base accepts a proton.
Example: In , HA and A- are a conjugate acid–base pair.
Acid and Base Ionization Constants (Ka and Kb)
The strength of acids and bases is quantified by their ionization constants:
Ka: Acid ionization constant; larger Ka means stronger acid.
Kb: Base ionization constant; larger Kb means stronger base.
Inverse Relationship: Stronger acid (larger Ka) has weaker conjugate base; stronger base (larger Kb) has weaker conjugate acid.
Relationship Between Ka and Kb
For a conjugate acid–base pair, the product of Ka and Kb equals the ionization constant of water:
This relationship allows calculation of Kb from Ka and vice versa, and illustrates the inverse relationship between acid and base strength.
Calculating pH and pOH
For strong acids and bases, use the following formulas:
pH:
pOH:
Relationship: at 25°C
pH + pOH = 14
pOH Calculation for Strong Bases
For strong bases, the concentration of OH- depends on the number of hydroxide ions produced per formula unit:
NaOH: Produces 1 OH- per unit.
Ba(OH)2: Produces 2 OH- per unit; [OH-] is twice the molar concentration.
pH and pOH of Weak Acids and Bases
Weak acids and bases only partially dissociate. Use an ICE table and the appropriate Ka or Kb to determine equilibrium concentrations before calculating pH or pOH.
Salt Solutions
Nature of Salts and Their Solutions
Salts are ionic compounds formed from acid–base neutralization. The pH of a salt solution depends on the strengths of the parent acid and base:
Neutral Salt: Strong acid + strong base (e.g., NaCl) → neutral solution.
Acidic Salt: Strong acid + weak base (e.g., NH4Cl) → acidic solution.
Basic Salt: Weak acid + strong base (e.g., NaC2H3O2) → basic solution.
Calculating pH of Salt Solutions
To determine the pH:
Identify if the salt is acidic, basic, or neutral.
For acidic/basic salts, write the hydrolysis reaction, set up an ICE table, and solve for equilibrium [H+] or [OH-].
For neutral salts, pH = 7.0; no calculation needed.
Additional info: Academic context and examples were added to clarify and expand upon brief study guide points, ensuring completeness and self-contained explanations.
