 Back
BackChemical Equilibrium: Concepts, Calculations, and Applications
Study Guide - Smart Notes

Chemical Equilibrium
Introduction to Chemical Equilibrium
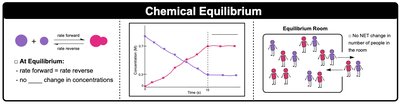
Chemical equilibrium is a fundamental concept in general chemistry, describing the state in which the rates of the forward and reverse reactions are equal, resulting in no net change in the concentrations of reactants and products over time. This dynamic process is central to understanding how reactions behave under various conditions.
Dynamic Process: At equilibrium, reactions continue in both directions, but the overall concentrations remain constant.
Equilibrium Representation: Reactions at equilibrium are denoted with a double arrow (⇌).
Types of Equilibrium:
Homogeneous Equilibrium: All reactants and products are in the same phase (e.g., all gases).
Heterogeneous Equilibrium: Reactants and products are in different phases (e.g., solids and gases).
Example: When a reaction reaches equilibrium, reactants form products as fast as products form reactants.
Equilibrium Constant (K)
Definition and Expression
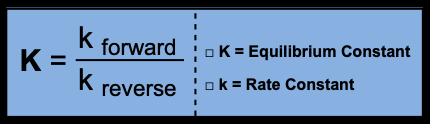
The equilibrium constant (K) quantifies the ratio of product to reactant concentrations at equilibrium. It is a central parameter for predicting the direction and extent of chemical reactions.
K vs. k: K is the equilibrium constant (unitless or depends on reaction), while k is the rate constant (units depend on reaction order).
Equilibrium Constant Expression: For a general reaction aA + bB ⇌ cC + dD, the equilibrium constant is:

Temperature Dependence: K is temperature dependent; changing temperature alters its value.
Exclusion of Solids and Pure Liquids: Solids and pure liquids are omitted from K expressions because their concentrations do not change.
Magnitude and Interpretation of K
The magnitude of K indicates whether products or reactants are favored at equilibrium.
K > 1: Products are favored.
K < 1: Reactants are favored.
K = 1: Neither direction is favored; significant amounts of both reactants and products are present.
Relation to Rate Constants:
Kp and Kc: Gas vs. Solution Equilibria
Relationship and Calculation
Equilibrium constants can be expressed in terms of partial pressures (Kp) for gases or concentrations (Kc) for solutions. The two are related by the following formula:
Δn: The change in moles of gas, calculated as Δn = moles of gaseous products - moles of gaseous reactants.
R: Gas constant (0.0821 L·atm·mol-1·K-1).
T: Temperature in Kelvin.

Manipulating Equilibrium Constants
Effect of Reaction Rearrangement
Changing the coefficients or direction of a reaction affects the equilibrium constant:
Multiplying Reaction: Raise K to the power of the multiplication factor.
Reversing Reaction: Take the inverse of K.
Dividing Reaction: Take the root of K according to the division factor.

Le Chatelier’s Principle
Response to Disturbances
Le Chatelier’s Principle states that if a system at equilibrium is disturbed, it will shift in a direction that minimizes the disturbance and re-establishes equilibrium. Common disturbances include changes in concentration, pressure, and addition of inert gases.
Adding/Removing Reactants or Products: Shifts equilibrium to counteract the change.
Changing Volume/Pressure: Affects gaseous equilibria; increasing pressure favors the side with fewer moles of gas.
Adding Inert Gas: No effect on equilibrium position if volume is constant.
Catalysts: Do not shift equilibrium; only affect the rate at which equilibrium is reached.
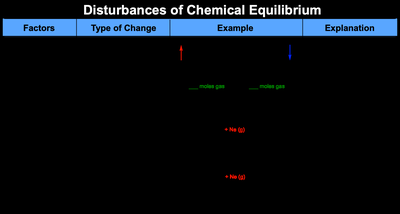
Temperature Changes
Changing the temperature affects the equilibrium position depending on whether the reaction is exothermic or endothermic.
Exothermic Reaction: Increasing temperature shifts equilibrium to the left (reactants).
Endothermic Reaction: Increasing temperature shifts equilibrium to the right (products).
Equilibrium Constant: K changes with temperature.

ICE Charts (Initial, Change, Equilibrium)
Calculating Equilibrium Amounts
ICE charts are used to systematically calculate equilibrium concentrations or partial pressures for reactions. They are especially useful when initial amounts and equilibrium constants are known, but some equilibrium values are missing.
Steps:
Set up the ICE chart with initial values.
Define changes using variables (usually x).
Express equilibrium values in terms of x.
Set up the equilibrium constant expression and solve for x.
Shortcuts: When both numerator and denominator are squared, use the root method. If the ratio of K to initial concentration is very small or large, use the approximation method.

Reaction Quotient (Q)
Comparing Q and K
The reaction quotient (Q) is calculated the same way as K, but using concentrations or pressures at any point in time, not necessarily at equilibrium. Comparing Q to K predicts the direction the reaction will shift:
Q < K: Reaction shifts right (towards products).
Q > K: Reaction shifts left (towards reactants).
Q = K: System is at equilibrium.
Example: For the reaction N2 (g) + 3 H2 (g) ⇌ 2 NH3 (g), if Q < K, more NH3 will form.
Summary Table: Key Equilibrium Concepts
Concept | Definition | Formula |
|---|---|---|
Chemical Equilibrium | State where forward and reverse rates are equal | -- |
Equilibrium Constant (K) | Ratio of product to reactant concentrations at equilibrium | |
Kp vs Kc | Gas-phase vs solution-phase equilibrium constants | |
Le Chatelier's Principle | System shifts to minimize disturbance | -- |
ICE Chart | Method for calculating equilibrium values | -- |
Reaction Quotient (Q) | Ratio at any time, predicts direction of shift | Same as K formula |
