 Back
BackChemical Equilibrium: Expressions, Calculations, and Applications
Study Guide - Smart Notes

Chemical Equilibrium
Equilibrium Constants and Chemical Equations
Chemical equilibrium occurs when the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products. The equilibrium constant (K) quantifies the ratio of product and reactant concentrations at equilibrium.
Equilibrium Constant Expression: For a general reaction aA + bB → cC + dD, the equilibrium constant is given by:
Manipulating Equilibrium Constants: Reversing a reaction inverts the equilibrium constant, and multiplying the coefficients by a factor raises K to that power.
Example: For the synthesis and decomposition of ammonia:
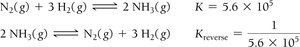
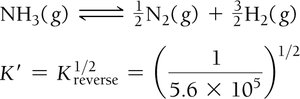
Relating Kc and Kp
For reactions involving gases, equilibrium constants can be expressed in terms of concentrations (Kc) or partial pressures (Kp). The relationship is:
, where is the change in moles of gas.
Example Calculation:

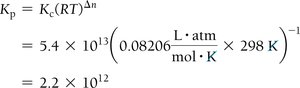
Finding Equilibrium Constants from Experimental Data
Experimental measurements of concentrations at equilibrium allow calculation of K using ICE tables (Initial, Change, Equilibrium).
ICE Table Example:
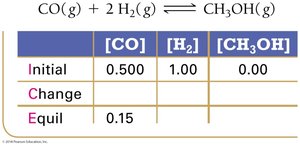
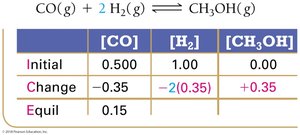
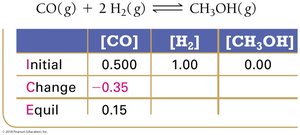
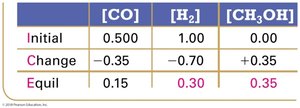
Equilibrium Constant Calculation:
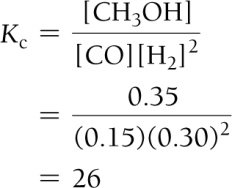
ICE Tables for Other Reactions
ICE tables are used for a variety of reactions to track changes and calculate equilibrium concentrations and constants.
Example: Methane Decomposition
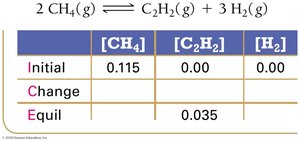
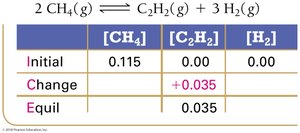
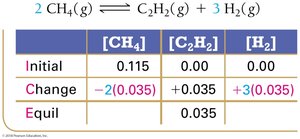
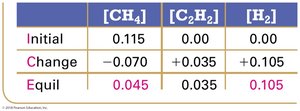
Equilibrium Constant Calculation:
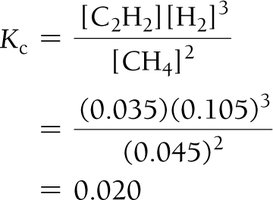
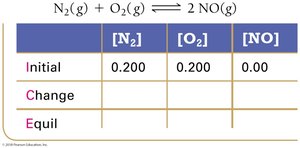
Reaction Quotient (Q) and Predicting Direction
The reaction quotient (Q) is calculated using initial concentrations or partial pressures. Comparing Q to K predicts the direction of the reaction:
If Q < K, the reaction proceeds to the right (toward products).
If Q > K, the reaction proceeds to the left (toward reactants).
Example:
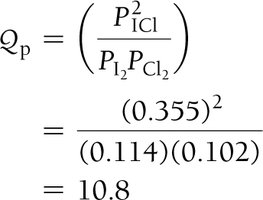
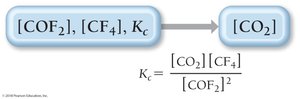
Finding Unknown Equilibrium Concentrations
If all but one equilibrium concentration is known, use the equilibrium constant expression to solve for the unknown.
Example:
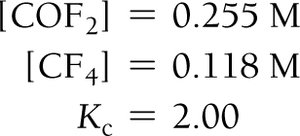
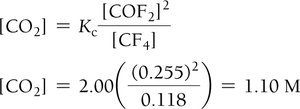
Solving for Equilibrium Concentrations with Initial Data
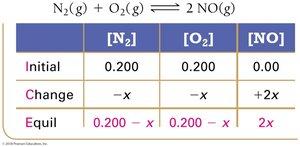
When only initial concentrations and the equilibrium constant are known, use ICE tables and algebraic methods (including quadratic equations) to solve for equilibrium concentrations.
Example: Nitrogen and Oxygen Reaction
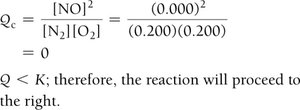
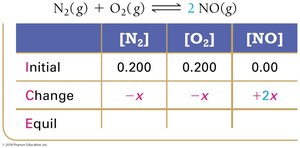
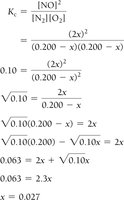
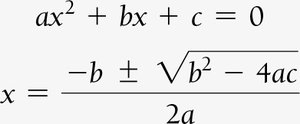

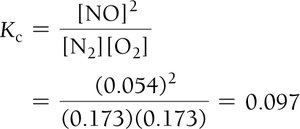
Example: Dinitrogen Tetraoxide and Nitrogen Dioxide
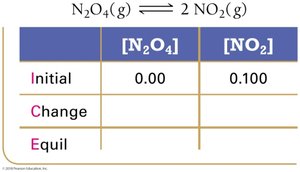
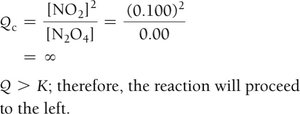
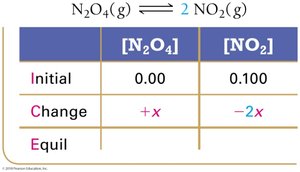
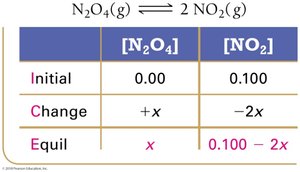
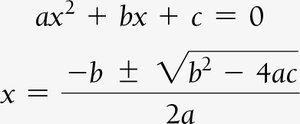
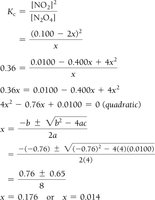
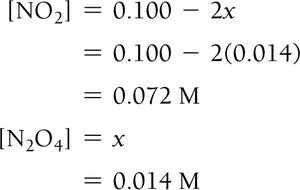
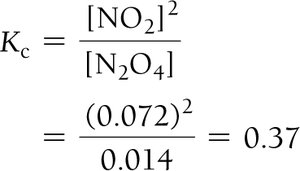
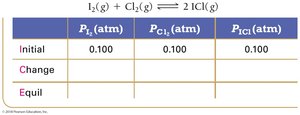
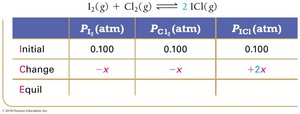
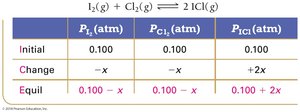
Equilibrium Partial Pressures
For reactions involving gases, partial pressures can be used in ICE tables and equilibrium expressions.
Example: Iodine and Chlorine Reaction
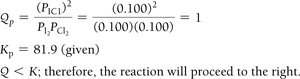
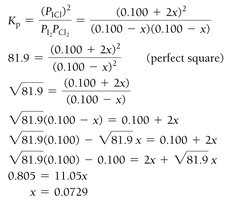
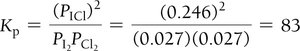
Small Equilibrium Constants and Approximations
When K is very small, the reaction does not proceed far toward products. Approximations can simplify calculations, but must be checked for validity.
Example: Hydrogen Disulfide Decomposition
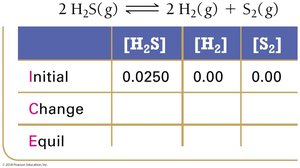
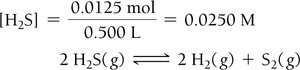
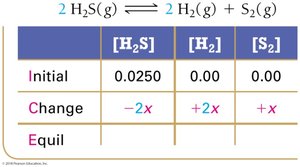
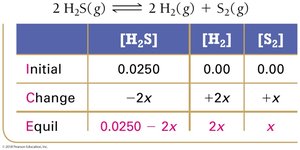
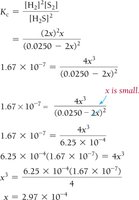

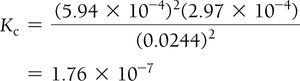
Example: Lower Initial Concentration
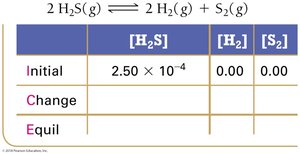
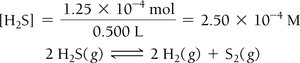
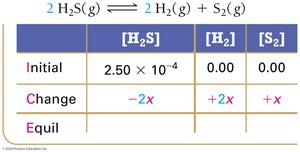
Summary Table: ICE Table Structure
ICE tables are a fundamental tool for equilibrium calculations. They organize the initial, change, and equilibrium values for each species in a reaction.
Step | Description |
|---|---|
Initial | Starting concentrations or partial pressures |
Change | Change in each species (based on stoichiometry) |
Equilibrium | Final concentrations or partial pressures at equilibrium |
Quadratic equations and approximations are often used to solve for unknowns in equilibrium calculations.
Additional info: These notes expand on the original examples by providing definitions, formulas, and stepwise explanations for equilibrium calculations, including the use of ICE tables, reaction quotients, and the relationship between Kc and Kp.
