 Back
BackChemical Equilibrium: Principles and Calculations
Study Guide - Smart Notes

Chemical Equilibrium
The Concept of Chemical Equilibrium
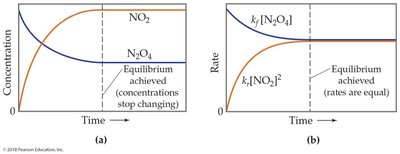
Chemical equilibrium is a fundamental concept in chemistry describing a state in which the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products. This balance is dynamic, meaning reactions continue to occur, but there is no net change in the amounts of substances.
Equilibrium State: At equilibrium, the system's composition remains unchanged over time.
Dynamic Nature: Both forward and reverse reactions proceed, but at equal rates.
Example: The dissociation of dinitrogen tetroxide (N2O4) into nitrogen dioxide (NO2) illustrates equilibrium as color changes stop when equilibrium is reached.
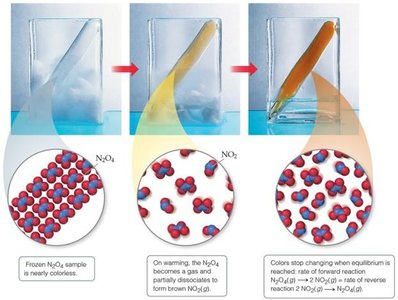
Equilibrium Constant and Its Expression
The equilibrium constant quantifies the ratio of product and reactant concentrations at equilibrium. It is denoted as K and depends on the balanced chemical equation.
Notation: The double arrow (
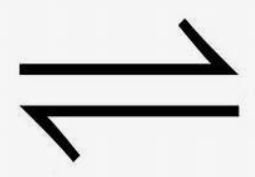 ) indicates a reversible reaction.
) indicates a reversible reaction.General Expression: For a reaction aA + bB → cC + dD, the equilibrium constant expression is:
Pressure-based Equilibrium: For gaseous reactions, equilibrium can also be expressed in terms of partial pressures (Kp).


Magnitude and Interpretation of K
The value of the equilibrium constant indicates the extent to which a reaction favors products or reactants.
K >> 1: Products predominate at equilibrium; equilibrium "lies to the right".
K << 1: Reactants predominate; equilibrium "lies to the left".

Stoichiometry and Equilibrium Constants
Changing the stoichiometry of a reaction affects the equilibrium constant:
Multiplying Equation: Raise K to the corresponding power.
Dividing Equation: Take the appropriate root of K.
Reversing Equation: Use the reciprocal of K.
Combining Equilibrium Expressions
When reactions are added, their equilibrium constants are multiplied to obtain the new constant for the overall reaction.
Example: If two reactions have constants K1 and K2, the combined reaction has K = K1 × K2.
Homogeneous and Heterogeneous Equilibria
Equilibria can involve substances in the same phase (homogeneous) or different phases (heterogeneous).
Homogeneous Equilibrium: All reactants and products are in the same phase.
Heterogeneous Equilibrium: At least one component is in a different phase. Concentrations of pure solids and liquids are omitted from the equilibrium expression.
Example: The decomposition of calcium carbonate:

Calculating Equilibrium Constants
To determine the equilibrium constant from experimental data:
Tabulate initial and equilibrium concentrations.
Calculate changes in concentration.
Use stoichiometry to relate changes among species.
Find equilibrium concentrations.
Substitute into the equilibrium expression to solve for K.


Reaction Quotient (Q) and Predicting Direction
The reaction quotient (Q) is calculated using current concentrations or pressures and compared to K to predict the direction of the reaction.
Q < K: Reaction proceeds toward products.
Q = K: System is at equilibrium.
Q > K: Reaction proceeds toward reactants.
Calculating Equilibrium Concentrations
Given initial concentrations and K, equilibrium concentrations can be found by setting up a table and solving for the change variable (often "x") using stoichiometry and the equilibrium expression.
Quadratic Equations: Sometimes, solving for x requires the quadratic formula.
Physical Meaning: Only positive, physically meaningful values for concentrations are accepted.
Le Châtelier’s Principle
Le Châtelier’s Principle states that if a system at equilibrium is disturbed, it will shift to counteract the disturbance and re-establish equilibrium.
Change in Concentration: Adding/removing reactants or products shifts equilibrium to use up or produce the component.
Change in Volume/Pressure: For gases, increasing volume (decreasing pressure) favors the side with more moles; decreasing volume favors the side with fewer moles.
Change in Temperature: For endothermic reactions, adding heat shifts equilibrium toward products (K increases); for exothermic reactions, adding heat shifts equilibrium toward reactants (K decreases).
Catalysts: Catalysts speed up attainment of equilibrium but do not affect the equilibrium composition or constant.
Summary Table: Types of Equilibrium and Effects
Type | Description | Effect on K |
|---|---|---|
Homogeneous | All substances in same phase | No effect |
Heterogeneous | Substances in different phases; solids/liquids omitted from K | No effect |
Change in Concentration | Shifts equilibrium position | K unchanged |
Change in Pressure/Volume | Shifts equilibrium for gases | K unchanged |
Change in Temperature | Shifts equilibrium; affects K | K changes |
Catalyst | Speeds up equilibrium attainment | K unchanged |
Additional info: The notes above expand on brief points from the original slides, providing definitions, examples, and academic context for clarity and completeness.
