 Back
BackChemical Equilibrium: Principles, Calculations, and Applications
Study Guide - Smart Notes

Chemical Equilibrium
Introduction to Chemical Equilibrium
Chemical equilibrium occurs when the rates of the forward and reverse reactions in a chemical system are equal, resulting in constant concentrations of reactants and products. This state is dynamic, meaning that reactions continue to occur, but there is no net change in the amounts of substances.
Dynamic Equilibrium: Both forward and reverse reactions proceed at equal rates.
Equilibrium Mixture: The composition of the mixture remains constant over time.
Equilibrium Constant (K): Quantifies the ratio of product and reactant concentrations at equilibrium.
Equilibrium Constant Expressions
Writing Equilibrium Constant Expressions
The equilibrium constant expression for a reaction is derived from the balanced chemical equation. For a general reaction:
The equilibrium constant expression is:
[ ]: Denotes concentration in mol/L (molarity).
Kc: Used for concentrations; Kp is used for partial pressures in gaseous systems.

Calculating Equilibrium Concentrations
To solve for unknown equilibrium concentrations, set up an ICE (Initial, Change, Equilibrium) table. Substitute equilibrium values into the K expression and solve for the unknown.
Set up the balanced equation and write the K expression.
List initial concentrations, changes, and equilibrium concentrations.
Solve for unknowns using algebraic methods (factoring, quadratic formula, or approximation).


Factors Affecting Equilibrium
Le Châtelier's Principle
Le Châtelier's Principle states that if a system at equilibrium is disturbed, the system will shift in the direction that counteracts the disturbance to re-establish equilibrium.
Concentration: Adding/removing reactants or products shifts equilibrium to consume or produce them.
Pressure (for gases): Increasing pressure favors the side with fewer moles of gas; decreasing pressure favors the side with more moles of gas.
Temperature: Increasing temperature favors the endothermic direction; decreasing temperature favors the exothermic direction.
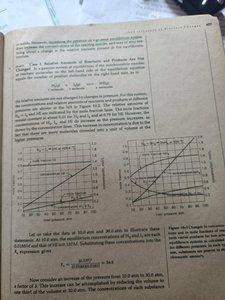
Effect of Pressure Changes
Pressure changes affect equilibria involving gases. If the number of moles of gas changes during the reaction, altering the pressure will shift the equilibrium position.
For reactions where the number of moles of gas is the same on both sides, pressure changes have no effect.
For reactions with different numbers of gas moles, increasing pressure shifts equilibrium toward fewer moles of gas.


Equilibrium Constants for Gaseous Systems
Equilibrium Constant in Terms of Pressure (Kp)
For gaseous reactions, the equilibrium constant can be expressed in terms of partial pressures:
P: Partial pressure of each gas (in atm or other pressure units).
Relationship between and :
where is the change in moles of gas, is the gas constant, and is temperature in Kelvin.

Thermodynamics and Equilibrium
Relationship Between ΔG and K
The standard free energy change () is related to the equilibrium constant by:
If , (products favored at equilibrium).
If , (reactants favored at equilibrium).

Influence of Temperature and Catalysts
Temperature Effects
Changing temperature alters the value of the equilibrium constant (). For endothermic reactions, increasing temperature increases $K$; for exothermic reactions, increasing temperature decreases $K$.
Temperature changes shift the equilibrium position and change the value of .

Effect of a Catalyst
A catalyst increases the rate at which equilibrium is achieved but does not affect the position of equilibrium or the value of the equilibrium constant. It lowers the activation energy for both forward and reverse reactions equally.
Chapter Review
Summary of Key Concepts
Chemical equilibrium is dynamic, with no net change in concentrations of reactants and products.
The equilibrium constant () quantifies the ratio of products to reactants at equilibrium.
Le Châtelier's Principle predicts the direction of shift when a system at equilibrium is disturbed.
Pressure, temperature, and concentration changes can affect equilibrium position; only temperature changes .
Thermodynamics links and , providing insight into reaction spontaneity and equilibrium position.
Catalysts speed up the attainment of equilibrium but do not alter its position.

