Back
BackChemical Equilibrium: Principles, Calculations, and Applications
Study Guide - Smart Notes

Chemical Equilibrium
Dynamic Equilibrium
Chemical equilibrium is a state in which the forward and reverse reactions occur at the same rate, resulting in constant concentrations of reactants and products. This state is dynamic, meaning that both reactions continue to occur, but there is no net change in the system.
Dynamic equilibrium involves two opposing processes occurring at the same rate.
At equilibrium, the system exhibits sameness (equal rates) and constancy (unchanging concentrations).
Equilibrium can be illustrated by analogies, such as traffic flow in opposite directions at equal rates.

Equilibrium and Living Systems
Living organisms maintain a controlled disequilibrium with their environment, which is essential for life. In contrast, non-living systems, such as a cup of hot water, eventually reach equilibrium with their surroundings without control.
Living things regulate internal conditions to remain out of equilibrium with their environment.
Upon death, organisms lose this control and come into equilibrium with their surroundings.
The Rate of a Chemical Reaction
The rate of a chemical reaction measures how quickly reactants are converted to products. Reaction rates are crucial for understanding how equilibrium is established.
Reaction rate is defined as the amount of reactant converted to product per unit time.
Fast reactions reach equilibrium quickly; slow reactions take longer.
Factors affecting reaction rate include concentration, temperature, and the presence of a catalyst.
Collision Theory of Chemical Reactions
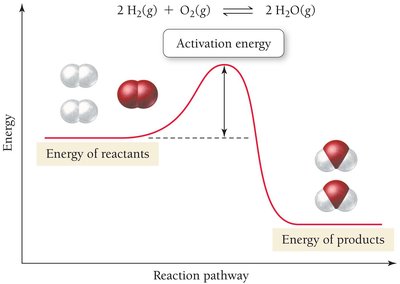
According to collision theory, chemical reactions occur when molecules or atoms collide with sufficient energy to overcome the activation energy barrier.
Activation energy is the minimum energy required for a reaction to occur.
High-energy collisions are more likely to result in product formation.
Factors influencing the number of high-energy collisions include reactant concentration and temperature.
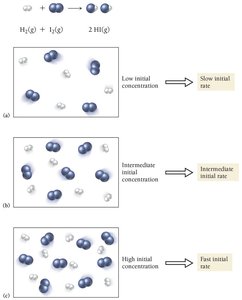
Effect of Concentration on Reaction Rate
Increasing the concentration of reactants generally increases the reaction rate by raising the frequency of collisions.
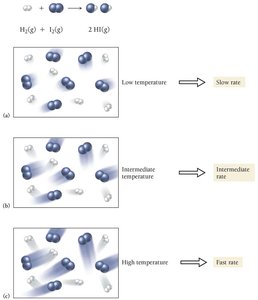
Effect of Temperature on Reaction Rate
Raising the temperature increases the kinetic energy of molecules, leading to more frequent and energetic collisions, thus increasing the reaction rate.
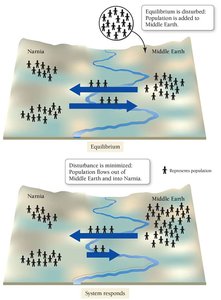
Establishing Chemical Equilibrium
In a reversible reaction, equilibrium is reached when the rates of the forward and reverse reactions are equal. At this point, the concentrations of reactants and products remain constant.
Equilibrium is not static; both reactions continue at equal rates.
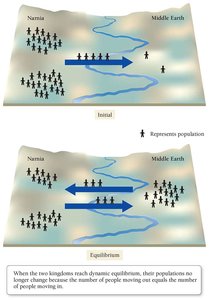
The system can be modeled by analogies, such as population movement between two regions.
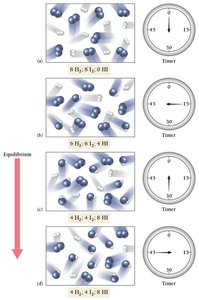
The Equilibrium Constant (Keq)
The equilibrium constant quantifies the ratio of product and reactant concentrations at equilibrium for a given reaction.
For a general reaction:
The equilibrium constant expression is:
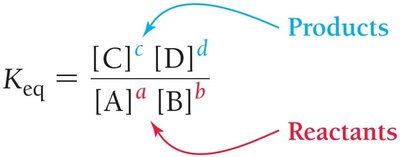
The exponents correspond to the stoichiometric coefficients in the balanced equation.
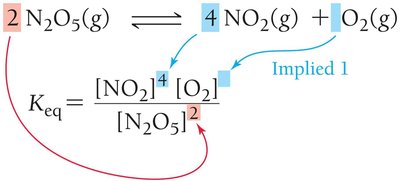
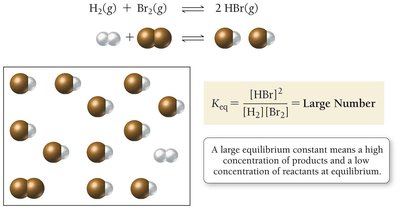
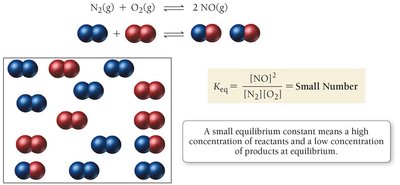
Significance of Keq
If is large (), products predominate at equilibrium.
If is small (), reactants predominate at equilibrium.
If , significant amounts of both reactants and products are present.
Calculating Equilibrium Constants
To calculate , substitute the equilibrium concentrations of all species into the equilibrium expression.
Example: For , with M, M, M:
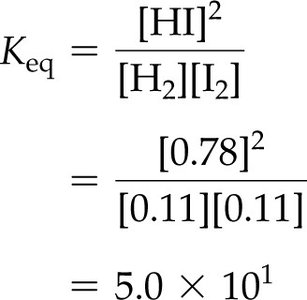
Using Keq in Calculations
Given and the concentrations of all but one species, you can solve for the unknown concentration.
Example: For , , M, M, find :
M
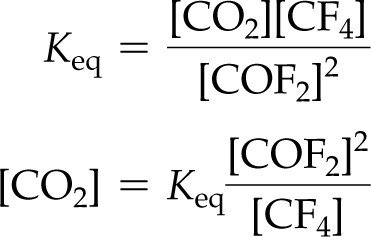
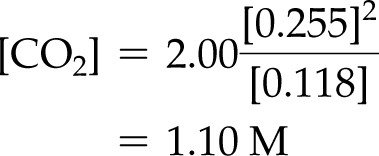
Le Châtelier’s Principle
Le Châtelier’s Principle states that if a system at equilibrium is disturbed, it will shift in the direction that minimizes the disturbance.
Adding reactants or products, changing volume, or changing temperature can disturb equilibrium.
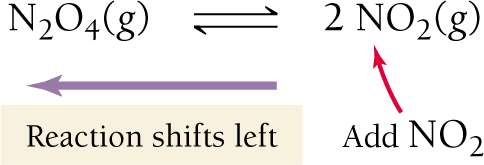
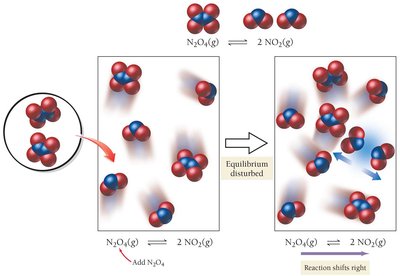
Effect of Adding Products or Reactants
Adding a product shifts the reaction toward the reactants (left).
Adding a reactant shifts the reaction toward the products (right).
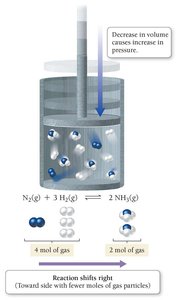
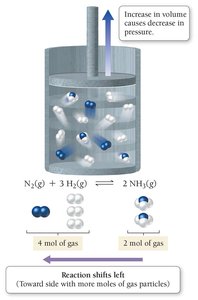
Effect of Volume and Pressure Changes
For reactions involving gases, changing the volume affects the pressure and can shift the equilibrium.
Decreasing volume (increasing pressure) shifts equilibrium toward the side with fewer moles of gas.
Increasing volume (decreasing pressure) shifts equilibrium toward the side with more moles of gas.
Effect of Temperature Changes
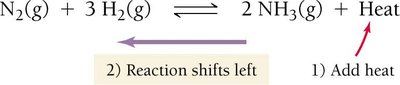
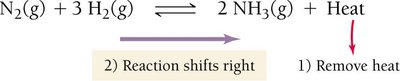
The effect of temperature depends on whether the reaction is exothermic or endothermic.
For exothermic reactions (heat as a product): Increasing temperature shifts equilibrium left; decreasing temperature shifts right.
For endothermic reactions (heat as a reactant): Increasing temperature shifts equilibrium right; decreasing temperature shifts left.
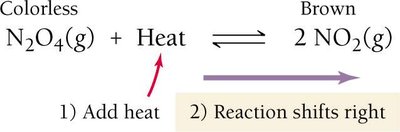
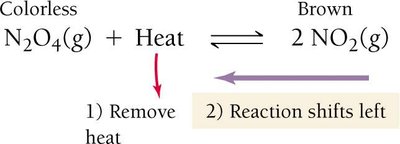

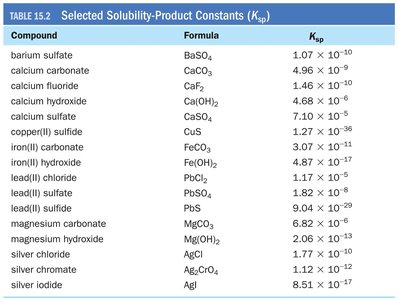
The Solubility-Product Constant (Ksp)
The solubility-product constant, , describes the equilibrium between a solid ionic compound and its dissolved ions in solution.
Solids are omitted from the equilibrium expression.
A large indicates high solubility; a small $K_{sp}$ indicates low solubility.
Calculating Molar Solubility from Ksp
For :
Given , M
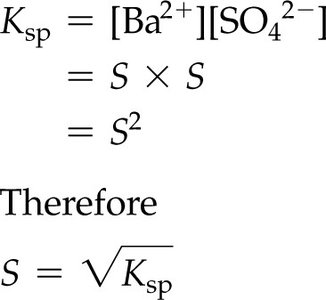
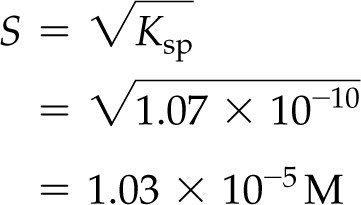
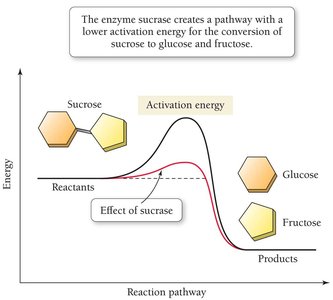
Reaction Pathways and Catalysts
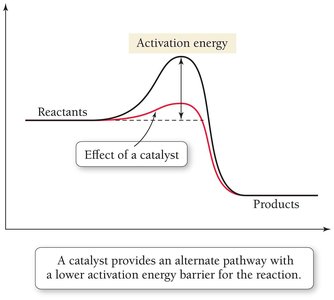
The equilibrium constant determines the extent of a reaction, while the activation energy determines the rate. Catalysts increase reaction rates by lowering the activation energy barrier without affecting the equilibrium position.
High activation energy means a slower reaction rate.
Catalysts provide an alternative pathway with lower activation energy.

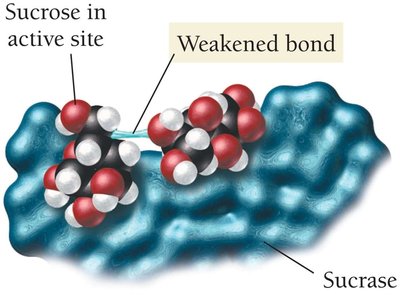
Enzymes: Biological Catalysts
Enzymes are biological catalysts that speed up biochemical reactions by lowering activation energies, allowing reactions to proceed rapidly at body temperature.