 Back
BackChemical Equilibrium: Principles, Expressions, and Applications
Study Guide - Smart Notes

Chemical Equilibrium
Dynamic Equilibrium and Thermodynamics
Chemical equilibrium is a fundamental concept in chemistry, describing a state in which the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products. This dynamic process is closely related to thermodynamics, as it reflects the balance of energy and matter in a chemical system.
Dynamic Equilibrium: The system is not static; reactants and products continue to interconvert, but their concentrations remain unchanged.
Thermodynamic Connection: Equilibrium is the point at which the free energy of the system is minimized.
Reversible Reactions: Most chemical reactions are reversible, meaning they can proceed in both directions.

Hemoglobin and Oxygen Equilibrium
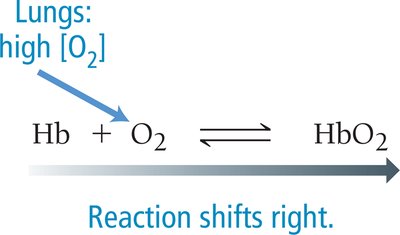
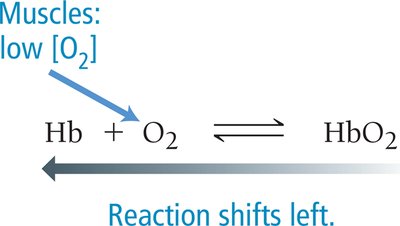
Hemoglobin (Hb) is a protein in red blood cells that binds oxygen (O2) in a reversible reaction, forming oxyhemoglobin (HbO2). This equilibrium is crucial for oxygen transport in the body.
Equilibrium Reaction: Hb + O2 ⇌ HbO2
Interdependence: The concentrations of Hb, O2, and HbO2 are interdependent and governed by the equilibrium constant, K.
Large K: Indicates a high concentration of products (HbO2) at equilibrium.
Shifts in Equilibrium: Changes in O2 concentration shift the equilibrium to favor either binding or release of O2.
Reaction Dynamics and Reaching Equilibrium
Progress of a Reaction
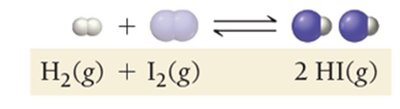
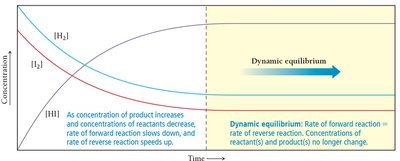
As a reaction proceeds, reactants are consumed and products are formed. The rates of the forward and reverse reactions change until equilibrium is reached.
Forward Reaction: Initially dominant as only reactants are present.
Reverse Reaction: Becomes significant as product concentration increases.
Equilibrium: Achieved when both rates are equal.
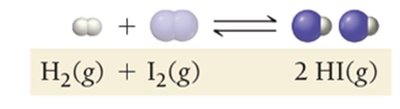
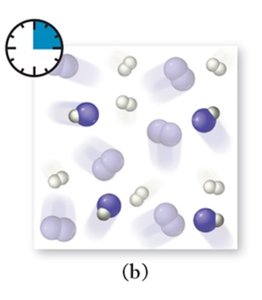
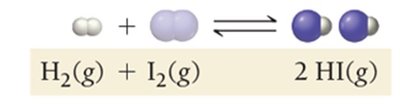
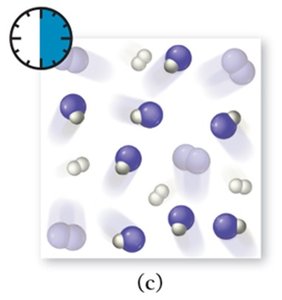
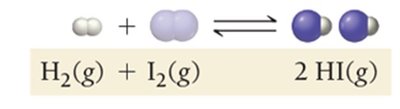
Dynamic Equilibrium: Key Characteristics
At equilibrium, the rates of the forward and reverse reactions are equal, but the concentrations of reactants and products are not necessarily equal.
Constant Concentrations: No net change in concentrations.
Position of Equilibrium: Can favor products or reactants depending on the value of K.
The Equilibrium Constant and the Law of Mass Action
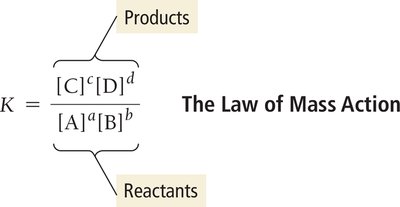
Defining the Equilibrium Constant (K)
The equilibrium constant, K, quantifies the ratio of product concentrations to reactant concentrations at equilibrium, each raised to the power of their coefficients in the balanced equation.
Law of Mass Action: For a general reaction: aA + bB ⇌ cC + dD
Expression:
Unitless: K is a dimensionless quantity.
Interpreting the Value of K
K >> 1: Equilibrium favors products; high product concentration.
K << 1: Equilibrium favors reactants; high reactant concentration.
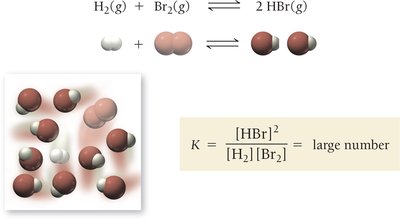
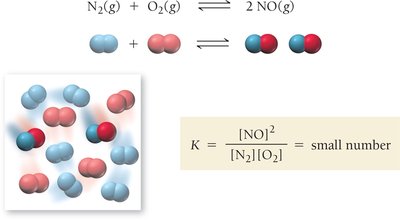
Relationships Between K and Chemical Equations
Manipulating Chemical Equations
Reverse Reaction: K is inverted ().
Multiplying Coefficients: K is raised to the power of the multiplying factor.
Adding Equations: K for the overall reaction is the product of the individual Ks.
Equilibrium Constants for Gaseous Reactions
Kc and Kp
For reactions involving gases, equilibrium constants can be expressed in terms of concentrations (Kc) or partial pressures (Kp).
Relationship: , where Δn is the change in moles of gas.
Kp = Kc: When Δn = 0.
Heterogeneous Equilibria
Solids and Liquids in Equilibrium Expressions
Pure solids and liquids are not included in equilibrium constant expressions because their concentrations do not change.
Example: For CO(g) + CO2(g) ⇌ C(s), only gases are included in K.
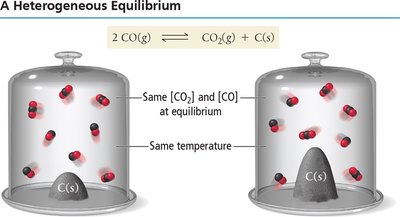
Calculating Equilibrium Constants
Using Measured Equilibrium Concentrations
Equilibrium constants can be determined by measuring the concentrations of reactants and products at equilibrium.
ICE Table: Initial, Change, Equilibrium concentrations are used to solve for unknowns.
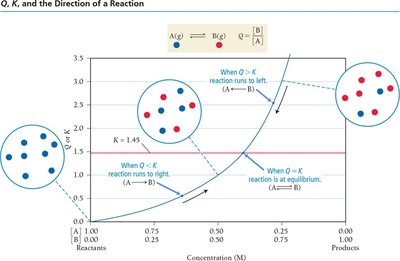
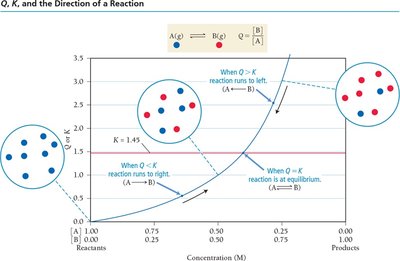
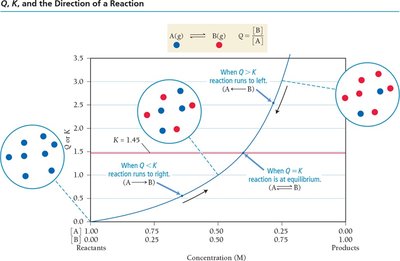
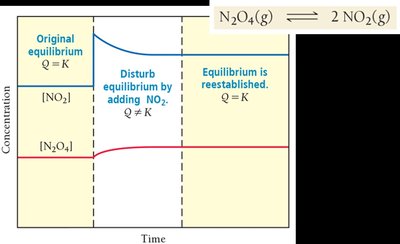
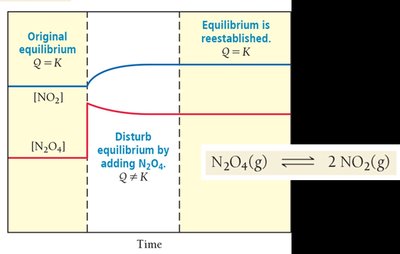
The Reaction Quotient (Q)
Predicting the Direction of Reaction
The reaction quotient, Q, is calculated using the same expression as K but with current concentrations. Comparing Q to K predicts the direction the reaction will proceed to reach equilibrium.
Q < K: Reaction proceeds forward (toward products).
Q > K: Reaction proceeds in reverse (toward reactants).
Q = K: Reaction is at equilibrium.
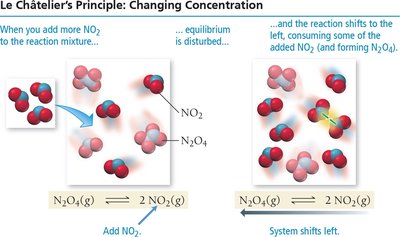
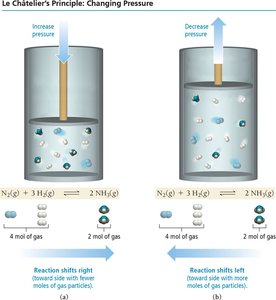
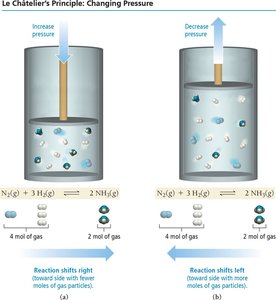
Le Châtelier’s Principle
Disturbing and Restoring Equilibrium
Le Châtelier’s principle states that if a system at equilibrium is disturbed, it will shift to minimize the disturbance and restore equilibrium.
Concentration: Adding reactant shifts equilibrium toward products; removing reactant shifts toward reactants.
Volume/Pressure: Decreasing volume (increasing pressure) shifts equilibrium toward the side with fewer gas molecules.
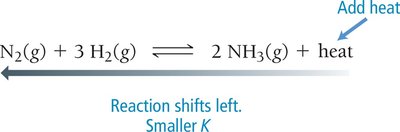
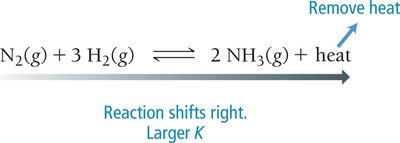
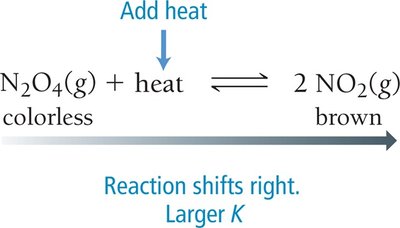
Temperature: For exothermic reactions, adding heat shifts equilibrium toward reactants; for endothermic reactions, adding heat shifts toward products.

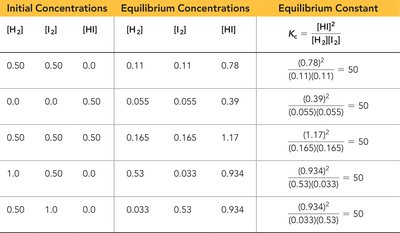
Summary Table: Calculating Equilibrium Constants
The following table summarizes how equilibrium constants are calculated from measured concentrations:
Initial Concentrations | Equilibrium Concentrations | Equilibrium Constant |
|---|---|---|
[H2] = 0.50, [I2] = 0.50 | [H2] = 0.11, [I2] = 0.11, [HI] = 0.78 | Kc = (0.78)2/(0.11*0.11) = 50 |
[H2] = 0.00, [I2] = 0.50 | [H2] = 0.055, [I2] = 0.055, [HI] = 0.39 | Kc = (0.39)2/(0.055*0.055) = 50 |
[H2] = 0.50, [I2] = 0.50 | [H2] = 0.165, [I2] = 0.165, [HI] = 1.17 | Kc = (1.17)2/(0.165*0.165) = 50 |
[H2] = 1.0, [I2] = 0.53 | [H2] = 0.033, [I2] = 0.033, [HI] = 0.934 | Kc = (0.934)2/(0.033*0.033) = 50 |
[H2] = 0.50, [I2] = 1.0 | [H2] = 0.033, [I2] = 0.53, [HI] = 0.934 | Kc = (0.934)2/(0.033*0.53) = 50 |
Additional info: These notes expand on the original outline by providing definitions, formulas, and examples, as well as visual aids to reinforce key concepts in chemical equilibrium. The content is structured to be self-contained and suitable for exam preparation in a general chemistry course.
