 Back
BackChemical Equilibrium: Principles, Expressions, and Calculations
Study Guide - Smart Notes

Chemical Equilibrium
Introduction to Chemical Equilibrium
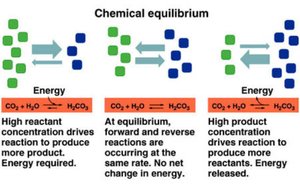
Chemical equilibrium occurs when the rates of the forward and reverse reactions in a chemical system are equal, resulting in constant concentrations of reactants and products over time. This concept is fundamental in understanding how reactions behave under different conditions and is central to many areas of chemistry.
Dynamic equilibrium: Both forward and reverse reactions continue to occur, but there is no net change in concentrations.
Equilibrium position: Indicates the relative concentrations of reactants and products at equilibrium.
Energy changes: At equilibrium, there is no net change in energy.
Driving force: High reactant concentration drives the reaction toward products; high product concentration drives the reaction toward reactants.
The Equilibrium Expression
The equilibrium expression quantitatively describes the relationship between the concentrations (or partial pressures) of reactants and products at equilibrium for a given reaction.
General form: For the reaction aA + bB ⇌ cC + dD, the equilibrium constant expression is: $K_c = \frac{[C]^c[D]^d}{[A]^a[B]^b}$
For gaseous reactions: The equilibrium constant can be expressed in terms of partial pressures: $K_p = \frac{(P_C)^c(P_D)^d}{(P_A)^a(P_B)^b}$
Stoichiometry: The exponents in the equilibrium expression correspond to the coefficients in the balanced chemical equation.
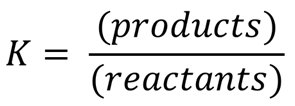
Extent of Reaction: Meaning of K
The value of the equilibrium constant, K, indicates the extent to which a reaction proceeds.
Large K: Equilibrium mixture contains mostly products.
Small K: Equilibrium mixture contains mostly reactants.
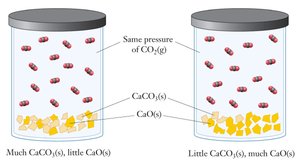
Heterogeneous Equilibrium
Heterogeneous equilibrium involves reactions where reactants and products are in different phases (solid, liquid, gas, aqueous). In such cases, only the concentrations (or partial pressures) of gases and dissolved substances appear in the equilibrium expression; pure solids and liquids are omitted.
Example: For the reaction CaCO3(s) ⇌ CaO(s) + CO2(g), the equilibrium expression is: $K = P_{CO_2}$
Solids and liquids: Their concentrations are constant and do not affect the equilibrium expression.
Manipulating Equilibrium Expressions
Equilibrium expressions can be manipulated mathematically when reactions are reversed, combined, or multiplied by a factor.
Reversing a reaction: $K_{new} = \frac{1}{K_{old}}$
Combining reactions: $K_{new} = K_1 \times K_2 \times ...$
Multiplying a reaction by n: $K_{new} = (K_{old})^n$
Dividing a reaction by n: $K_{new} = \sqrt[n]{K_{old}}$
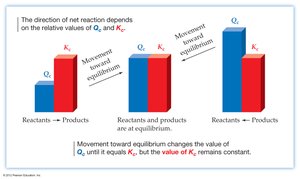
Reaction Quotient (Q) and Predicting Direction
The reaction quotient, Q, is calculated using the same form as the equilibrium constant, but with concentrations or pressures at any point in time (not necessarily at equilibrium).
If Q < K: Reaction proceeds forward (toward products).
If Q > K: Reaction proceeds in reverse (toward reactants).
If Q = K: System is at equilibrium.
Calculating Equilibrium Constants and Concentrations
ICE Tables: Initial, Change, Equilibrium
ICE tables are used to organize and solve equilibrium problems by tracking the initial concentrations, changes during the reaction, and final equilibrium concentrations.
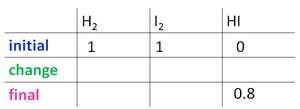
Step 1: Write the balanced equation and set up the ICE table.
Step 2: Express changes in terms of a variable (usually x).
Step 3: Substitute equilibrium values into the equilibrium expression and solve for x.
Step 4: Calculate equilibrium concentrations.

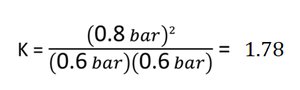
Worked Example: Calculating K
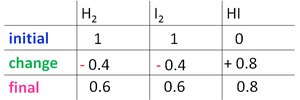
For the reaction H2(g) + I2(g) ⇌ 2 HI(g), starting with 1 atm each of H2 and I2, and finding 0.8 atm HI at equilibrium:
Change: 2x = 0.8 atm, so x = 0.4 atm.
Final pressures: H2 = 0.6 atm, I2 = 0.6 atm, HI = 0.8 atm.
Equilibrium constant: $K = \frac{(0.8)^2}{(0.6)(0.6)} = 1.78$


Calculating Equilibrium Concentrations from K
Given the equilibrium constant and initial concentrations, equilibrium concentrations can be found by setting up an ICE table and solving the resulting equation, often a quadratic.
Example: For PCl5(g) ⇌ PCl3(g) + Cl2(g) with K = 33 at 250°C, and initial concentrations of 0.050 M PCl3 and 0.015 M Cl2:
Quadratic equation: $33x^2 - 3.145x + 0.02475 = 0$
Solution: Use the quadratic formula to find x, then calculate equilibrium concentrations.
Summary Table: Equilibrium Calculations
Given | Find |
|---|---|
Initial concentrations, K | Equilibrium concentrations |
Equilibrium concentrations | K |


Key Takeaways
Chemical equilibrium is a dynamic state where forward and reverse reaction rates are equal.
Equilibrium expressions are based on stoichiometry and only include gases and dissolved substances.
ICE tables are essential tools for solving equilibrium problems.
Manipulating equilibrium expressions allows calculation of new equilibrium constants for modified reactions.
Reaction quotient (Q) helps predict the direction a reaction will proceed to reach equilibrium.
Additional info:
Quadratic equations are often required when solving for equilibrium concentrations.
Practice problems and further reading can be found at ChemWiki ICE Tables.
