 Back
BackChemical Equilibrium: Principles, Expressions, and Calculations
Study Guide - Smart Notes

Chemical Equilibrium
Introduction to Chemical Equilibrium
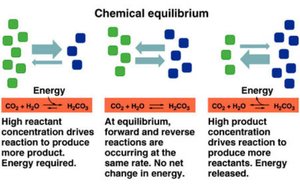
Chemical equilibrium is a fundamental concept in chemistry where the rates of the forward and reverse reactions are equal, resulting in constant concentrations of reactants and products over time. At equilibrium, the system is dynamic, with ongoing reactions in both directions but no net change in the amounts of substances.
Dynamic Equilibrium: Both forward and reverse reactions continue to occur at equal rates.
Constant Concentrations: The concentrations of all reactants and products remain unchanged at equilibrium.
Energy Considerations: No net energy change occurs at equilibrium, though energy may be required or released as the system approaches equilibrium.
Directionality: The system can shift toward products or reactants depending on initial conditions and external changes.
Equilibrium Expressions
Writing and Manipulating Equilibrium Expressions
The equilibrium constant expression quantifies the ratio of product concentrations to reactant concentrations, each raised to the power of their stoichiometric coefficients. This expression is essential for predicting the composition of an equilibrium mixture.
General Form: For a reaction aA + bB ⇌ cC + dD, the equilibrium constant (Kc) is given by:
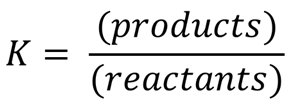
Partial Pressures: For gaseous reactions, the equilibrium constant can also be expressed in terms of partial pressures (Kp):
Magnitude of K: A large K indicates a product-favored equilibrium; a small K indicates a reactant-favored equilibrium.

Extent of Reaction: What Does K Mean?
The value of the equilibrium constant (K) provides insight into the extent of a reaction:
Large K (>>1): The equilibrium mixture contains mostly products.
Small K (<<1): The equilibrium mixture contains mostly reactants.
Relation Between Kp and Kc
Connecting Concentration and Pressure Equilibria
For reactions involving gases, the equilibrium constant can be expressed in terms of concentrations (Kc) or partial pressures (Kp). The relationship between them depends on the change in the number of moles of gas (Δn):
If Δn = 0:
If Δn ≠ 0:
Where: Δn = (moles of gaseous products) – (moles of gaseous reactants), R = 0.0821 L·atm/(mol·K), T = temperature in Kelvin.
Example: For the reaction , Δn = -2.
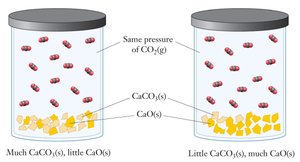
Heterogeneous Equilibria
Equilibria Involving Multiple Phases
In heterogeneous equilibria, pure solids and liquids do not appear in the equilibrium expression. Only aqueous and gaseous species are included.
Solids and Liquids: Their concentrations are constant and thus omitted from the equilibrium expression.
Example: For , the equilibrium expression is .
Manipulations of the Equilibrium Expression
Rules for Modifying Equilibrium Constants
When chemical equations are manipulated, the equilibrium constant must be adjusted accordingly:
Reversing a Reaction:
Multiplying by n:
Dividing by n:
Adding Reactions:
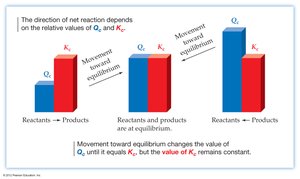
Reaction Quotient (Q) and Predicting Direction
Comparing Q and K
The reaction quotient (Q) is calculated using the same form as the equilibrium expression but with current (not necessarily equilibrium) concentrations. Comparing Q to K predicts the direction the reaction will proceed:
If Q < K: The reaction proceeds forward (toward products).
If Q > K: The reaction proceeds in reverse (toward reactants).
If Q = K: The system is at equilibrium.
Calculating Equilibrium Constants and Concentrations
ICE Tables (Initial, Change, Equilibrium)
ICE tables are a systematic way to organize data and solve equilibrium problems. They track the initial amounts, changes during the reaction, and final equilibrium values for all species involved.
Step 1: Write the balanced equation and set up the ICE table.
Step 2: Express changes in terms of x (the amount reacted or formed).
Step 3: Substitute equilibrium values into the equilibrium expression and solve for x.
Step 4: Calculate equilibrium concentrations or pressures.
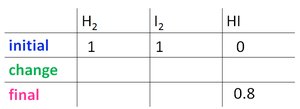

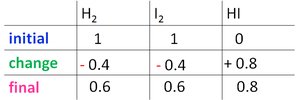
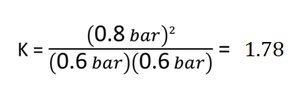
Worked Example: Calculating K
For the reaction , starting with 1 atm each of H2 and I2, and finding 0.8 atm HI at equilibrium:
Set up the ICE table and solve for x.
Calculate K using the equilibrium expression:

Worked Example: Calculating Equilibrium Concentrations
Given initial concentrations and K, use the ICE table and solve the resulting equation (sometimes quadratic) to find equilibrium concentrations.
Example: For , with at 250°C, and initial concentrations of 0.050 M PCl3 and 0.015 M Cl2, set up the ICE table and solve for x.
Use the quadratic formula if necessary.
Summary Table: Key Equilibrium Relationships
Manipulation | Effect on K |
|---|---|
Reverse reaction | |
Multiply equation by n | |
Add equations | |
Divide equation by n |
Practice and Application
Practice setting up and solving ICE tables for a variety of equilibrium problems.
Apply the relationships between Kc and Kp for gaseous systems.
Use Q to predict the direction of reaction shifts.
Additional info: For more practice, refer to online resources such as ChemWiki's ICE table exercises.
