 Back
BackChemical Equilibrium: Principles, Manipulation, and Kinetic Relationships
Study Guide - Smart Notes

Chapter 15 – Chemical Equilibrium
Introduction to Chemical Equilibrium
Chemical equilibrium occurs when the rates of the forward and reverse reactions in a closed system are equal, resulting in constant concentrations of reactants and products. The position of equilibrium can be described quantitatively by the equilibrium constant, K.
Dynamic Equilibrium: Both forward and reverse reactions continue to occur, but there is no net change in concentrations.
Equilibrium Constant (K): Expresses the ratio of product concentrations to reactant concentrations at equilibrium, each raised to the power of their coefficients in the balanced equation.
Equation for a general reaction:
Equilibrium constant expression:
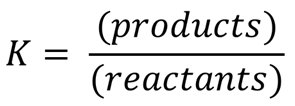
The Equilibrium Expression
The equilibrium constant, K, is a central concept in equilibrium calculations. It is determined by the balanced chemical equation and is temperature-dependent.
K > 1: Products are favored at equilibrium.
K < 1: Reactants are favored at equilibrium.
Chapter 15.2 – Manipulating Reaction Equilibrium
Le Chatelier’s Principle
Le Chatelier’s Principle states that if a system at equilibrium is disturbed by a change in concentration, pressure, or temperature, the system will shift its equilibrium position to counteract the disturbance and reestablish equilibrium.
Disturbance: Any change in concentration, pressure, or temperature.
Response: The system shifts to minimize the effect of the disturbance.
Example: The melting of ice under high pressure is an application of Le Chatelier’s Principle. At high pressure, the melting point of ice decreases because melting reduces volume, relieving the stress of increased pressure.
Additional info: The images above show the molecular structure of solid and liquid water, illustrating the difference in density and hydrogen bonding.
Changes in Concentration
Adding or removing reactants or products disturbs equilibrium. The system shifts to restore equilibrium, but the value of K does not change.
Adding Reactant: Shifts equilibrium toward products.
Removing Product: Also shifts equilibrium toward products.
Adding Product or Removing Reactant: Shifts equilibrium toward reactants.
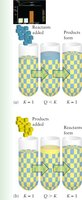
Example: For the reaction , if more NO2 is added, the system shifts to produce more N2O4 until equilibrium is reestablished.
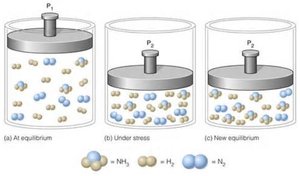
Changes in Pressure and Volume
Changing the pressure or volume affects equilibria involving gases. The system shifts toward the side with fewer moles of gas when pressure is increased (volume decreased), and toward more moles of gas when pressure is decreased (volume increased). For reactions with equal moles of gas on both sides, there is no effect.
Decrease Volume (Increase Pressure): Shifts toward fewer moles of gas.
Increase Volume (Decrease Pressure): Shifts toward more moles of gas.
Changes in Temperature
Temperature changes affect both the position of equilibrium and the value of K. The direction of the shift depends on whether the reaction is exothermic or endothermic.
Exothermic Reaction (releases heat): Increasing temperature shifts equilibrium toward reactants; K decreases.
Endothermic Reaction (absorbs heat): Increasing temperature shifts equilibrium toward products; K increases.
Equations:
Exothermic:
Endothermic:
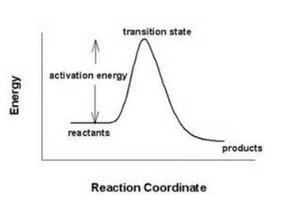
Effect of Catalysts
Catalysts lower the activation energy for both the forward and reverse reactions equally, increasing the rate at which equilibrium is achieved but not affecting the equilibrium position or the value of K.
Link Between Chemical Equilibrium and Chemical Kinetics
Relationship Between Rate Constants and Equilibrium Constant
At equilibrium, the rate of the forward reaction equals the rate of the reverse reaction. The equilibrium constant can be expressed in terms of the rate constants for the forward and reverse reactions:
k: Rate constant for a reaction.
Kc: Equilibrium constant in terms of concentration.
The rate constant is given by the Arrhenius equation:
Ea: Activation energy
R: Universal gas constant (8.3145 J·mol−1·K−1)
T: Temperature in Kelvin
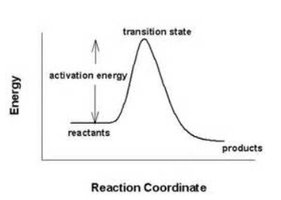
Effect of Temperature on Equilibrium and Kinetics
For exothermic reactions, increasing temperature decreases the equilibrium constant K because the reverse reaction's activation energy decreases more than the forward reaction's. For endothermic reactions, increasing temperature increases K.
Practice Problems and Applications
Sample Problems
Problem 1: For the equilibrium CO(g) + 2 H2(g) ⇌ CH3OH(g), removing CH3OH will shift equilibrium to produce more CH3OH.
Problem 2: For H+(aq) + CH3CO2−(aq) ⇌ CH3CO2H(aq), adding NaOH removes H+, shifting equilibrium to produce more H+ and CH3CO2−.
Problem 3: Pressurizing a reaction vessel with an inert gas at constant volume does not affect the equilibrium position.
Problem 4: For the synthesis of ammonia, heating the reaction will not increase the amount of ammonia because the reaction is exothermic.
Problem 5: Adding a catalyst does not change the equilibrium concentrations of reactants or products.
Problem 6: Increasing the volume favors the side with more moles of gas.
Problem 7: If Kp decreases with increasing temperature, the reaction is exothermic.
Problem 8: The equilibrium constant can be calculated from the ratio of the forward and reverse rate constants.
Problem 9: At equilibrium, the rate constants for the forward and reverse reactions are related by the equilibrium constant.

Summary Table: Effects on Equilibrium
Change | Effect on Equilibrium Position | Effect on K |
|---|---|---|
Add reactant | Shifts toward products | No change |
Remove product | Shifts toward products | No change |
Increase pressure (decrease volume) | Shifts toward fewer moles of gas | May change if Δn ≠ 0 |
Increase temperature (exothermic) | Shifts toward reactants | K decreases |
Increase temperature (endothermic) | Shifts toward products | K increases |
Add catalyst | No effect | No effect |
