 Back
BackChemical Kinetics and Catalysis: Mechanisms, Rate Laws, and Energy Profiles
Study Guide - Smart Notes

Chemical Kinetics and Reaction Mechanisms
Elementary Reactions and Molecularity
Chemical reactions can occur in a single step or through multiple steps. An elementary reaction is a reaction that happens in one step, and its molecularity refers to the number of reactant molecules involved in that step.
Unimolecular: Involves one reactant molecule. Example: Cl2(g) → 2 Cl(g)
Bimolecular: Involves two reactant molecules. Example: NOCl(g) + Cl(g) → NO(g) + Cl2(g)
Termolecular: Involves three reactant molecules. These are rare.
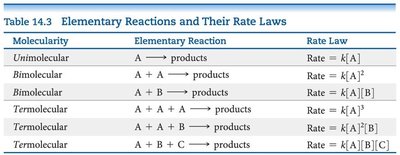
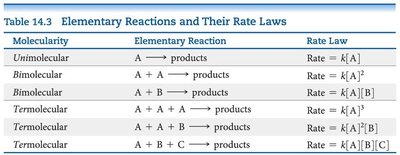
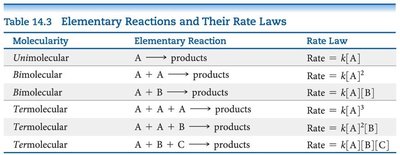
The rate law for an elementary reaction directly follows its stoichiometry.
Molecularity | Elementary Reaction | Rate Law |
|---|---|---|
Unimolecular | A → products | Rate = k[A] |
Bimolecular | A + A → products | Rate = k[A]2 |
Bimolecular | A + B → products | Rate = k[A][B] |
Termolecular | A + A + A → products | Rate = k[A]3 |
Termolecular | A + A + B → products | Rate = k[A]2[B] |
Termolecular | A + B + C → products | Rate = k[A][B][C] |
Reaction Mechanisms
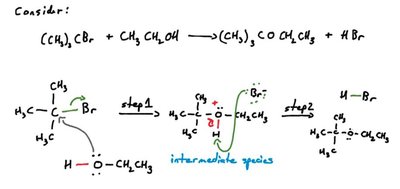
A reaction mechanism is the sequence of elementary steps that make up the overall reaction. Each step may produce or consume intermediates, which are species that appear in some steps but not in the overall reaction equation.
Intermediates: Produced in one step and consumed in another; do not appear in the overall reaction.
Overall Reaction: The sum of all elementary steps.
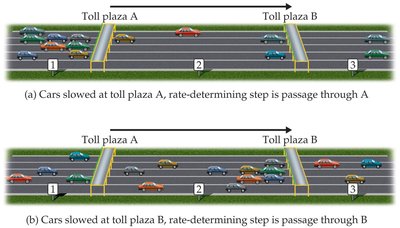
Rate-Determining Step and Multistep Mechanisms
In multistep reactions, the rate-determining step is the slowest step, which controls the overall reaction rate. The observed rate law is often determined by this step.
If the first step is slow, the rate law matches the first step.
If a later step is slow, earlier steps may affect the concentrations of intermediates, leading to more complex rate laws.
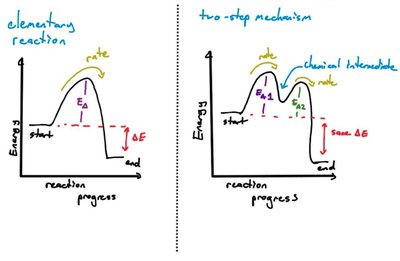
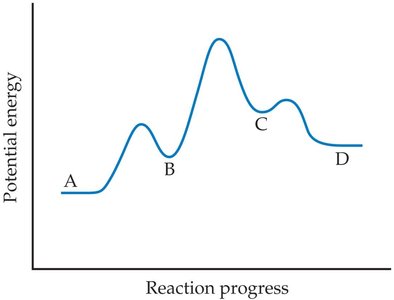
Energy Profiles and Reaction Progress
Potential Energy Diagrams
An energy profile shows the potential energy changes during a reaction. Multistep reactions have multiple maxima (activation energies) and minima (intermediates).
Activation Energy (Ea): The energy barrier for each step.
Intermediates: Correspond to valleys between peaks.
Rate-Determining Step: The step with the highest activation energy.
Catalysis
Definition and Types of Catalysts
A catalyst is a substance that increases the speed of a chemical reaction without being consumed. Catalysts are present at the start and regenerated at the end of the mechanism.
Homogeneous Catalyst: Same phase as reactants (e.g., liquid, gas).
Heterogeneous Catalyst: Different phase from reactants (usually solid).
How Catalysts Work
Catalysts provide an alternative reaction pathway with a lower activation energy, increasing the reaction rate.
Catalysts do not affect the overall thermodynamics (ΔE) of the reaction.
They are consumed in one step and regenerated in another.
Examples of Catalysis
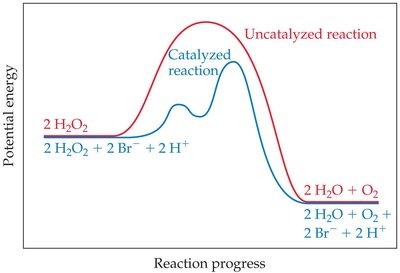
Homogeneous Catalysis: Decomposition of Hydrogen Peroxide
The decomposition of H2O2 can be catalyzed by Br- and H+ ions, which are regenerated at the end.
Overall Reaction: 2 H2O2(aq) → 2 H2O(l) + O2(g)
Intermediates: Br2(aq)
Catalysts: Br-(aq), H+(aq)


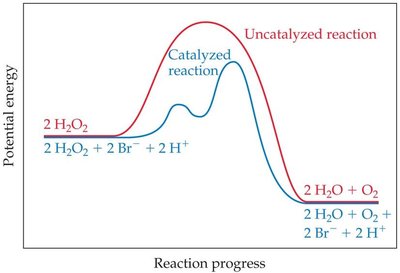
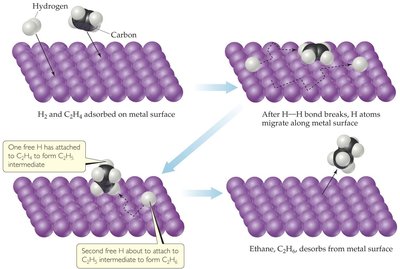
Heterogeneous Catalysis: Hydrogenation of Ethene
Heterogeneous catalysts, such as metals, facilitate reactions on their surfaces. For example, the hydrogenation of ethene to ethane occurs on a metal catalyst.
Reactants adsorb onto the catalyst surface.
Reaction occurs via surface intermediates.
Products desorb from the surface.
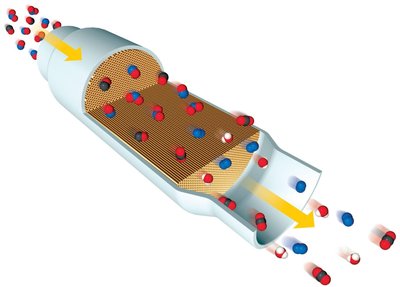
Catalytic Converters
Catalytic converters in automobiles use heterogeneous catalysts to convert harmful gases into less toxic products.
CO(g) or CxHy(g) + O2(g) → CO2(g) + H2O(g)
NO(g) or NO2(g) → N2(g) + O2(g)
Common catalysts: Pt, Pd, Rh
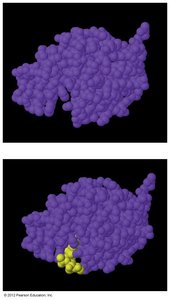
Enzymes: Biological Catalysts
Enzymes are biological catalysts that operate via a "lock and key" mechanism, binding substrates and lowering activation energy by straining bonds.
Highly specific for their substrates.
Examples: Amylase (converts starches to sugars).
Applications and Environmental Chemistry
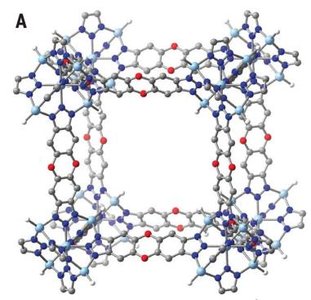
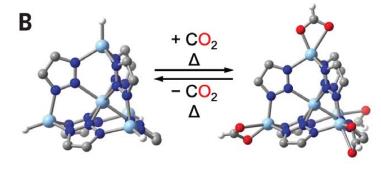
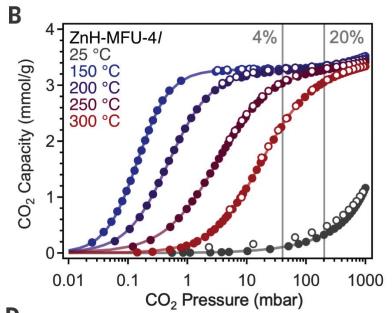
CO2 Capture and Metal-Organic Frameworks (MOFs)
CO2 capture is important for climate change mitigation and fuel generation. MOFs are porous materials designed to bind CO2 efficiently, even at high temperatures.
Challenges: Weak dispersion forces, low-temperature capture, energetic inefficiency.
Research focuses on creating reactive sites within MOFs to bind CO2 strongly.
Catalysis in the Atmosphere: Ozone Depletion
Catalytic mechanisms are involved in atmospheric chemistry, such as the destruction of ozone by chlorofluorocarbons (CFCs).
Ozone protects against UV radiation.
Catalytic cycles involving Cl radicals lead to ozone depletion.
Summary
Elementary reactions and their rate laws are fundamental to understanding reaction kinetics.
Reaction mechanisms consist of sequences of elementary steps, with intermediates and rate-determining steps.
Energy profiles illustrate activation energies and intermediates in multistep reactions.
Catalysts increase reaction rates by lowering activation energy and are crucial in industrial, environmental, and biological processes.
