 Back
BackChemical Kinetics and Rate Laws: Mini-Textbook Study Guide
Study Guide - Smart Notes

Chemical Kinetics: The Rate of Chemical Reactions
Introduction to Reaction Rates
Chemical kinetics is the study of the speed at which chemical reactions occur and the factors that affect these rates. Understanding reaction rates is essential for predicting how quickly products form and reactants are consumed.
Reaction Rate: The change in concentration of a reactant or product per unit time, typically expressed in mol/L·s or M/s.
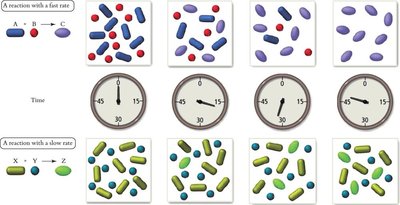
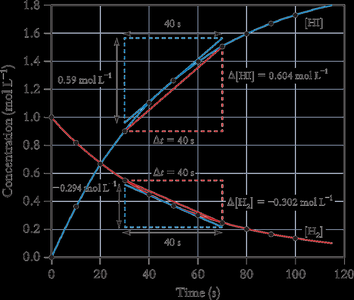
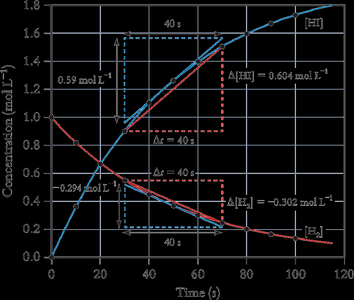
Example: For the reaction 2H2(g) + I2(g) → 2HI(g), the rate can be measured by the change in concentration of H2, I2, or HI over time.
Mathematical Operations in Chemistry
Many chemical calculations require the use of scientific notation, logarithms, and calculators. These mathematical tools are essential for handling large and small numbers, such as Avogadro's number or rate constants.
Scientific Notation: Used to express very large or small numbers, e.g., 6.0221 × 1023.
Calculator Usage: Most scientific calculators have dedicated keys for entering numbers in scientific notation (EE or EXP).
Logarithms: Logarithmic functions (log, ln) are used in rate laws and Arrhenius equations.





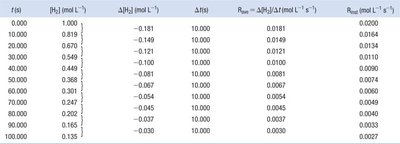
Measuring Reaction Rates
Average and Instantaneous Rates
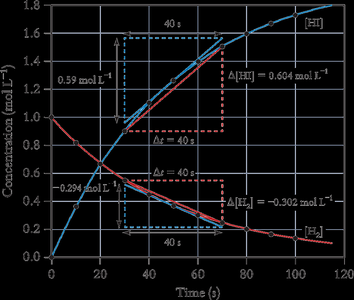
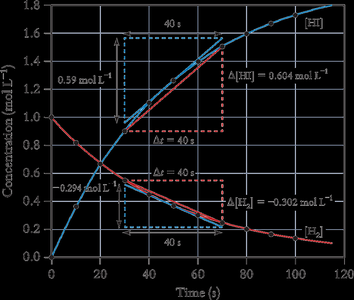
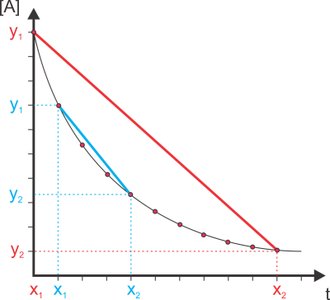
The rate of a reaction can be measured as an average over a time interval or as an instantaneous value at a specific moment.
Average Rate: The slope of the line joining two points on a concentration vs. time curve.
Instantaneous Rate: The slope of the tangent to the curve at a particular time.
Initial Rate: The rate at the moment reactants are mixed (t = 0).












Rate Laws and Reaction Order
Differential Rate Laws
The rate law expresses the relationship between the rate of a reaction and the concentrations of reactants. The form of the rate law must be determined experimentally.
General Form:
Order of Reaction: The exponent (n, m) indicates the order with respect to each reactant.
Rate Constant (k): A proportionality constant specific to the reaction and conditions.







Reaction Order for Multiple Reactants
For reactions involving multiple reactants, the overall order is the sum of the individual orders. The rate law is determined by analyzing experimental data.
Example: For the reaction , the reaction is first order in H2 and second order in NO, overall third order.



Experimental Determination of Rate Laws
Rate laws are determined by varying reactant concentrations and measuring initial rates. The order is deduced by comparing how the rate changes with concentration.
Experiment | [BrO3]− (mol/L) | [Br−] (mol/L) | [H+] (mol/L) | Initial Rate (mol/L·s) |
|---|---|---|---|---|
1 | 0.10 | 0.10 | 0.10 | 8.0 × 10−4 |
2 | 0.20 | 0.10 | 0.10 | 1.6 × 10−3 |
3 | 0.20 | 0.20 | 0.10 | 3.2 × 10−3 |
4 | 0.10 | 0.10 | 0.20 | 3.2 × 10−3 |
Integrated Rate Laws
Zero, First, and Second Order Reactions
Integrated rate laws relate reactant concentration to time and allow calculation of concentrations at any point during the reaction.
Zero Order:
First Order:
Second Order:
Half-life: The time required for the concentration to decrease by half.


Order | Rate Law | Integrated Law | Half-life | Plot |
|---|---|---|---|---|
0 | Rate = k | [A] vs. t | ||
1 | Rate = k[A] | ln[A] vs. t | ||
2 | Rate = k[A]2 | [A]−1 vs. t |
Temperature and Reaction Rates
Arrhenius Equation
The Arrhenius equation describes how the rate constant (k) depends on temperature and activation energy.
Equation:
Activation Energy (Ea): The minimum energy required for a reaction to occur.
Frequency Factor (A): The number of times reactants approach the activation barrier per unit time.
Temperature Effect: As temperature increases, more molecules have sufficient energy to react.
Reaction Mechanisms
Elementary Steps and Rate-Determining Step
Reaction mechanisms describe the sequence of elementary steps that make up the overall reaction. The slowest step determines the rate law.
Molecularity: The number of reactant particles involved in an elementary step (unimolecular, bimolecular, termolecular).
Rate Law for Elementary Steps: Can be written directly from the molecularity.
Rate-Determining Step: The slowest step in the mechanism, which controls the overall rate.
Elementary Step | Molecularity | Rate Law |
|---|---|---|
A → products | 1 | Rate = k[A] |
A + A → products | 2 | Rate = k[A]2 |
A + B → products | 2 | Rate = k[A][B] |
A + A + A → products | 3 | Rate = k[A]3 |
Catalysis and Reaction Mechanisms
Catalysis of Ozone Depletion
Catalysts provide alternative reaction pathways with lower activation energy, increasing the rate of reaction without being consumed.
Example: Chlorine-catalyzed ozone depletion involves a series of steps where Cl acts as a catalyst.
Summary Table: Integrated Rate Laws
Order | Rate Law | Integrated Law | Half-life | Plot |
|---|---|---|---|---|
0 | Rate = k | [A] vs. t | ||
1 | Rate = k[A] | ln[A] vs. t | ||
2 | Rate = k[A]2 | [A]−1 vs. t |
Additional info: This guide covers the essential concepts of chemical kinetics, rate laws, integrated rate laws, and reaction mechanisms, as well as mathematical operations relevant to general chemistry. It is suitable for exam preparation and self-study.
