 Back
BackChemical Kinetics and Rate Laws: Structured Study Notes
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the study of the rates at which chemical reactions occur and the factors that affect these rates. Understanding reaction rates is essential for predicting how quickly products form and reactants are consumed.
Reaction Rate: The change in concentration of reactants or products per unit time, typically expressed in mol/L·s or M/s.
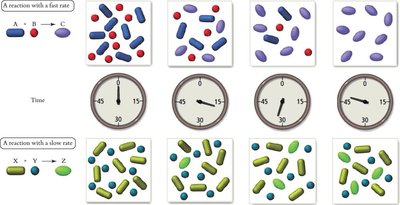
Factors Affecting Rate: Concentration, temperature, presence of catalysts, and surface area.
Example: The decomposition of hydrogen iodide: 2 HI(g) → H2(g) + I2(g)
Defining Reaction Rate
The rate of a chemical reaction can be defined in terms of the change in concentration of reactants or products over time. For a general reaction:
General Formula:
Units: mol/L·s or M/s
Example: For 2 HI(g) → H2(g) + I2(g), the rate of HI disappearance is twice the rate of H2 and I2 formation.
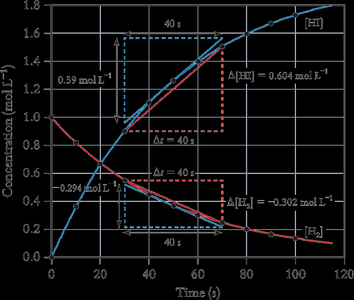
Average and Instantaneous Rate
The average rate is calculated over a finite time interval, while the instantaneous rate is the rate at a specific moment, determined by the slope of the tangent to the concentration vs. time curve.
Average Rate Formula:
Instantaneous Rate Formula:
Initial Rate: The rate at the moment reactants are mixed (t = 0).


Rate Laws and Reaction Order
Rate Law Definition
The rate law expresses the relationship between the reaction rate and the concentrations of reactants. It is determined experimentally and cannot be deduced from the balanced equation (except for elementary steps).
General Rate Law:
k: Rate constant, specific to each reaction and temperature.
n, m: Reaction orders with respect to each reactant.
Overall Order: Sum of exponents (n + m).

Determining Reaction Order
Reaction order is found by analyzing how changes in reactant concentrations affect the rate. Experimental data is used to compare rates at different concentrations.
Example Table:
Exp. | [A] (M) | Initial Rate (mol L−1 s−1) |
|---|---|---|
1 | 0.10 | 0.015 |
2 | 0.20 | 0.030 |
3 | 0.40 | 0.060 |
Order Calculation: If doubling [A] doubles the rate, n = 1 (first order).
Reaction Order for Multiple Reactants
For reactions with multiple reactants, the rate law includes each reactant's concentration raised to its respective order.
General Formula:
Example: For 2 H2 + 2 NO → 2 H2O + N2, rate law might be (first order in H2, second order in NO).
Integrated Rate Laws
Zero Order Reactions
Zero order reactions have a constant rate independent of reactant concentration.
Rate Law:
Integrated Law:
Half-life:
Plot: [A] vs. t is linear.
First Order Reactions
First order reactions depend linearly on the concentration of one reactant.
Rate Law:
Integrated Law:
Half-life:
Plot: ln[A] vs. t is linear.
Second Order Reactions
Second order reactions depend on the square of one reactant or the product of two reactants.
Rate Law:
Integrated Law:
Half-life:
Plot: 1/[A] vs. t is linear.
Temperature and Reaction Rate
Arrhenius Equation
The Arrhenius equation describes how the rate constant (k) depends on temperature and activation energy.
Equation:
A: Frequency factor (number of collisions with correct orientation).
Ea: Activation energy (energy barrier).
R: Gas constant (8.314 J·mol−1·K−1).
T: Temperature in Kelvin.
Linear Form:
Reaction Mechanisms
Elementary Steps and Molecularity
A reaction mechanism is a sequence of elementary steps that describe the pathway from reactants to products. Each step has a molecularity and a corresponding rate law.
Unimolecular: One molecule decomposes (Rate = k[A])
Bimolecular: Two molecules collide (Rate = k[A][B])
Termolecular: Three molecules collide (rare, Rate = k[A][B][C])
Rate-Determining Step (RDS)
The slowest step in a reaction mechanism determines the overall rate law. The mechanism must match the experimentally observed rate law.
Example: For 2 NO + 2 H2 → N2 + 2 H2O, if the rate law is rate = k[NO]2[H2], the mechanism must include a slow step involving two NO and one H2.
Mathematical Operations and Functions in Chemistry
Scientific Notation and Calculator Use
Scientific notation is used to express very large or small numbers in chemistry. Calculators are essential tools for performing these operations.
Scientific Notation: (e.g., )
Calculator Functions: EE or EXP keys are used to enter scientific notation.
Example:





Logarithms and Antilogs
Logarithmic functions are used in chemistry for calculations involving pH, rate laws, and the Arrhenius equation.
Common Logarithm:
Natural Logarithm:
Antilog: The inverse of a logarithm, or
Example: (rounded to 4 significant figures)
Additional info: Logarithms are essential for understanding exponential decay, pH calculations, and the Arrhenius equation in chemical kinetics.
