Back
BackChemical Kinetics: Rates, Mechanisms, and Catalysis
Study Guide - Smart Notes

Chapter 14: Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the study of the rates at which chemical reactions occur and the factors that affect these rates. Understanding kinetics allows chemists to control reaction speed, improve yields, and optimize conditions for industrial and biological processes.
Reaction rate refers to the change in concentration of reactants or products per unit time.
Kinetics is distinct from thermodynamics, which determines if a reaction is spontaneous, while kinetics determines how fast it occurs.
Applications include controlling industrial reactions, understanding biological processes, and environmental chemistry.
Factors Affecting Reaction Rate
The rate of a chemical reaction depends on several key factors:
Physical state: Gases and liquids react faster than solids due to greater molecular mobility.
Concentration: Higher concentrations increase collision frequency, leading to faster reactions.
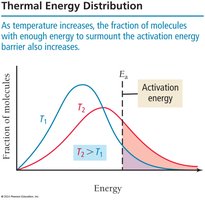
Temperature: Increased temperature raises kinetic energy, resulting in more frequent and energetic collisions.
Catalysts: Catalysts provide alternative pathways with lower activation energy, increasing reaction rate without being consumed.

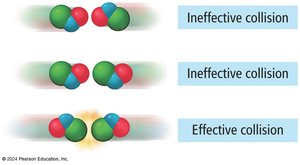
The Collision Model
According to the collision model, molecules must collide to react, and only collisions with sufficient energy and proper orientation result in product formation.
Effective collisions lead to chemical change; ineffective collisions do not.
Factors such as molecular orientation and energy distribution are crucial.
Defining and Measuring Reaction Rate
Reaction rate is quantitatively defined as the change in concentration of a reactant or product over time:
Rate formula: for reactant disappearance, for product appearance.
Units: mol L-1 s-1 (M s-1).
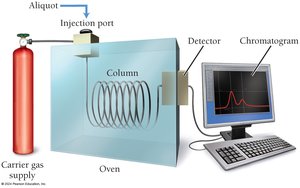
Rates are always positive and can be measured using spectrometry or chromatography.
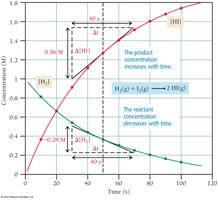
Stoichiometry and Rate
Reaction rates are related to stoichiometry. For example, in the reaction , the rate of appearance of HI is twice the rate of disappearance of H2 or I2.
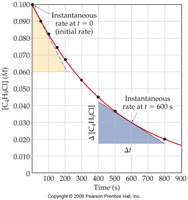
Average and Instantaneous Rates
Average rate is calculated over a time interval, while instantaneous rate is the rate at a specific moment, often determined by the slope of a tangent to a concentration vs. time curve.
The Rate Law
The rate law expresses the relationship between reaction rate and reactant concentrations. It is determined experimentally and has the general form:
k is the rate constant, m and n are reaction orders.
The sum of exponents gives the overall reaction order.
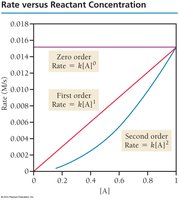
Common Reaction Orders
Reactions are classified by order:
Zero-order: Rate is independent of concentration.
First-order: Rate is proportional to concentration.
Second-order: Rate is proportional to the square of concentration.
Determining the Rate Law
Rate laws are determined by measuring initial rates while varying reactant concentrations. The order with respect to each reactant is found by holding other concentrations constant and observing rate changes.
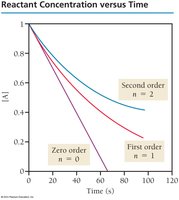
Integrated Rate Laws
Integrated rate laws relate reactant concentration to time and differ for each reaction order:
Zero-order:
First-order:
Second-order:
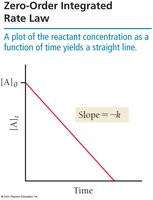

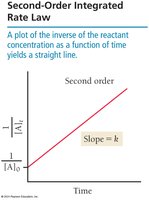
Half-Life of a Reaction
The half-life (t1/2) is the time required for the concentration of a reactant to decrease by half. For first-order reactions, half-life is constant and independent of concentration:
First-order:
Second-order:
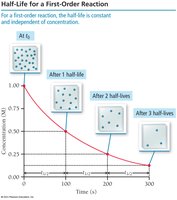
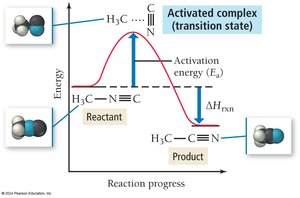
Temperature and Reaction Rate: The Arrhenius Equation
Reaction rates increase with temperature due to higher kinetic energy and more frequent effective collisions. The Arrhenius equation describes this relationship:
Ea is activation energy, R is the gas constant, T is temperature in Kelvin, A is the frequency factor.
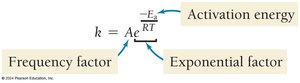
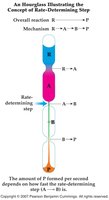
Reaction Mechanisms
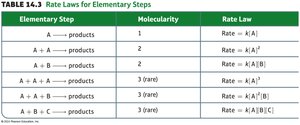
A reaction mechanism is the sequence of elementary steps by which a reaction occurs. The slowest step is the rate-determining step, which controls the overall rate.
Elementary steps can be unimolecular, bimolecular, or termolecular.
Intermediates are formed and consumed during the mechanism but do not appear in the overall reaction.
Catalysis
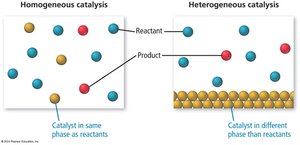
Catalysts increase reaction rates by providing alternative pathways with lower activation energy. They are not consumed in the reaction and can be homogeneous (same phase as reactants) or heterogeneous (different phase).
Homogeneous catalysts participate in the reaction and are regenerated.
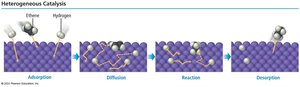
Heterogeneous catalysts provide a surface for reactants to adsorb, react, and desorb as products.
Enzymes and Biological Catalysis
Enzymes are biological catalysts that accelerate reactions in living organisms. They exhibit high specificity and efficiency, often following a lock-and-key model for substrate binding.
Catalase is an enzyme that decomposes hydrogen peroxide in cells, protecting tissues from oxidative damage.
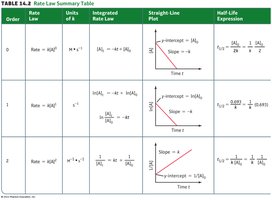
Summary Table: Rate Laws and Half-Life Expressions
The following table summarizes the key features of zero-, first-, and second-order reactions:
Order | Rate Law | Units of k | Integrated Rate Law | Straight-Line Plot | Half-Life Expression |
|---|---|---|---|---|---|
0 | Rate = k[A]0 | mol L-1 s-1 | [A] vs. t | ||
1 | Rate = k[A] | s-1 | ln[A] vs. t | ||
2 | Rate = k[A]2 | L mol-1 s-1 | 1/[A] vs. t |