 Back
BackChemical Kinetics: Rates, Mechanisms, and Catalysis
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the study of the speed (rate) of chemical reactions and the factors that affect these rates. Understanding reaction rates is crucial for controlling chemical processes in both industrial and biological contexts. - Reaction rate: The speed at which reactants are converted to products. - Chemical kinetics: The study of reaction rates and the factors influencing them, such as temperature, concentration, and nature of reactants.
Factors Affecting Reaction Rate
Several factors influence how fast a chemical reaction occurs: - Concentration: Higher concentration of reactants increases the rate. - Temperature: Increasing temperature generally increases the rate; for many reactions, a 10℃ rise doubles the rate. - Nature of reactants: Small molecules, gases, and ions react faster than large molecules, solids, or neutral molecules. - Physical state: Gases react faster than liquids, which react faster than solids; powdered solids are more reactive than blocks.
Defining and Measuring Reaction Rate
Rate Definition
The rate of a reaction is defined as the change in concentration of a reactant or product per unit time. - Instantaneous rate: The rate at a specific moment, given by the slope of the concentration vs. time curve. - Average rate: The rate over a time interval, approximated by the change in concentration divided by the change in time.
Mathematical Expressions for Rate
- For reactants, a negative sign is used to indicate decreasing concentration. 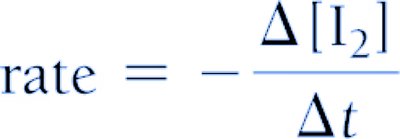
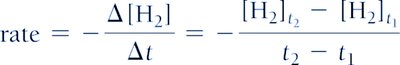
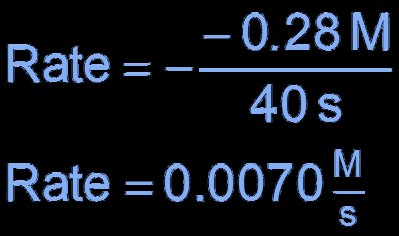
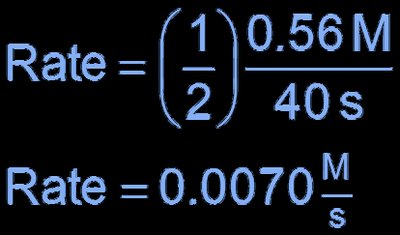
Stoichiometry and Rate
The rate of a reaction is related to the stoichiometry of the balanced equation. For a general reaction: 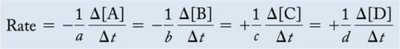
Methods of Measuring Reaction Rate
- Continuous monitoring: Measuring total pressure, polarimetry, or spectrophotometry. - Sampling at intervals: Drawing aliquots for titration, gravimetric analysis, or gas chromatography.
Rate Laws and Reaction Order
Rate Law Determination
The rate law expresses how the rate depends on the concentrations of reactants and catalysts. It must be determined experimentally. - For a reaction: - k: Rate constant (depends on temperature) - n: Order of reaction with respect to A
General Rate Law for Multiple Reactants
- m, n: Reaction orders (small integers) - Overall order:
Reaction Order Examples
- The exponent for each reactant in the rate law is its order. - The sum of exponents is the overall order. Example: Rate law: - Second order with respect to NO - First order with respect to O2 - Third order overall
Integrated Rate Laws
First-Order Reactions
- Rate law: - Integrated rate law: - Half-life: - Unit for k:
Second-Order Reactions
- Rate law: - Integrated rate law: - Half-life: 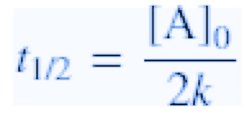 - Unit for k:
- Unit for k:
Zero-Order Reactions
- Rate law: - Integrated rate law: - Unit for k:
Activation Energy and Temperature Effects
Activation Energy and Transition State
- Activation energy (Ea): The minimum energy required to form the activated complex (transition state). - The activated complex has partially broken and formed bonds and is high in energy.
Arrhenius Equation
The Arrhenius equation relates the rate constant to temperature and activation energy: - T: Temperature in Kelvin - R: Gas constant () - Ea: Activation energy (J) - A: Frequency factor
Two-Point Arrhenius Equation
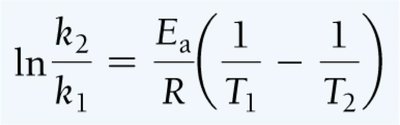
Reaction Mechanisms
Elementary Steps and Intermediates
Most reactions occur via a series of elementary steps. - Elementary step: A single reaction event involving one, two, or three molecules. - Intermediate: A species produced in one step and consumed in another; does not appear in the overall reaction. Example: Steps: 1. 2. HI is an intermediate.
Rate Laws for Elementary Steps
The rate law for an elementary step can be deduced directly from its equation. 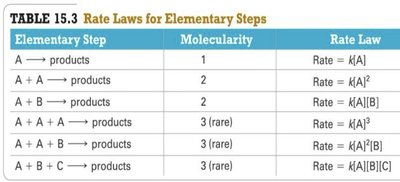
Elementary Step | Molecularity | Rate Law |
|---|---|---|
A → products | 1 | Rate = k[A] |
A + A → products | 2 | Rate = k[A]^2 |
A + B → products | 2 | Rate = k[A][B] |
A + A + A → products | 3 (rare) | Rate = k[A]^3 |
A + A + B → products | 3 (rare) | Rate = k[A]^2[B] |
A + B + C → products | 3 (rare) | Rate = k[A][B][C] |
Rate-Determining Step
- The slowest step in a mechanism determines the overall reaction rate. - The rate law for the rate-determining step matches the experimentally observed rate law.
Validating Mechanisms
To validate a proposed mechanism: - The elementary steps must sum to the overall balanced equation. - The predicted rate law must match the observed rate law.
Catalysis and Enzymes
Catalysts
- Catalyst: A substance that increases reaction rate without being consumed. - Catalysts provide an alternative mechanism with lower activation energy. - Homogeneous catalyst: Same phase as reactants. - Heterogeneous catalyst: Different phase from reactants.
Enzymes: Biological Catalysts
- Enzyme: Protein molecule that catalyzes biological reactions. - Enzymes bind substrate at an active site, orienting it for reaction (lock and key mechanism). 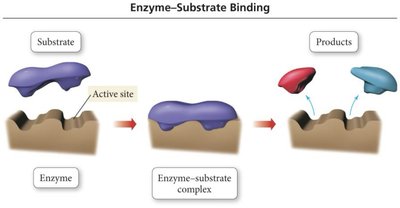
Ozone: Environmental Kinetics
Stratospheric Ozone
- Ozone absorbs UV radiation (220–300 nm), protecting life on Earth. - Ozone also absorbs IR radiation, contributing to the greenhouse effect.
Tropospheric Ozone
- Ground-level ozone forms from reactions between nitrogen oxides and volatile organic compounds. - Natural concentration: ~10 ppb; >70 ppb is unhealthy (EPA). - Harmful effects: Throat/lung irritation, aggravation of asthma/emphysema.
Summary Table: Key Rate Law Equations
Reaction Order | Integrated Rate Law | Half-Life Equation | Unit of k |
|---|---|---|---|
First Order | |||
Second Order | |||
Zero Order |
Additional info: Context and examples were expanded for clarity and completeness.
