 Back
BackChemical Kinetics: Rates, Mechanisms, and Catalysis
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the study of the speed (rate) of chemical reactions and the factors that affect these rates. Understanding reaction rates is crucial for controlling chemical processes in both laboratory and industrial settings.
Reaction Rate: The speed at which reactants are converted to products.
Importance: Allows chemists to optimize conditions for desired outcomes, such as maximizing product yield or minimizing unwanted side reactions.
Example: Ectothermic animals like lizards slow down in cold environments because their body reactions slow with temperature.
Defining Reaction Rate
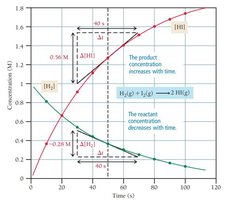
The rate of a reaction is measured as the change in concentration of reactants or products per unit time. Fast reactions convert a large fraction of molecules quickly, while slow reactions convert only a small fraction over the same period.
Mathematical Expression:
Average Rate: Change over a time interval.
Instantaneous Rate: Rate at a specific moment, determined by the slope of a tangent to the concentration vs. time curve.
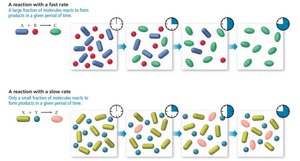
Factors Affecting Reaction Rate
Reactant Concentration
Increasing the concentration of reactants generally increases the reaction rate by raising the frequency of molecular collisions.
Gas Reactions: Rate depends on partial pressure.
Solution Reactions: Rate depends on molarity (solute-to-solution ratio).
Temperature
Higher temperatures increase reaction rates by providing more kinetic energy to molecules, resulting in more collisions with sufficient energy to break bonds.
Activation Energy: Minimum energy required to initiate a reaction.
Arrhenius Relationship: Reaction rate increases exponentially with temperature.
Nature of Reactants
The physical and chemical properties of reactants influence reaction rates.
Small molecules react faster than large ones.
Physical state: Gases > Liquids > Solids.
Surface area: Powdered solids react faster than blocks.
Ions react faster than molecules (no bonds to break).
Structure and Orientation
The orientation of colliding molecules affects whether a reaction occurs. Some reactions require specific alignment for bond formation or breaking.
Measuring Reaction Rate
Continuous Monitoring
For fast reactions, continuous monitoring methods are used to track concentration changes in real time.
Pressure changes: Useful for gas reactions.
Polarimetry: Measures optical rotation changes.
Spectroscopy: Measures absorbance changes at specific wavelengths.
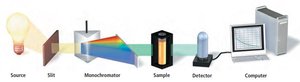
Sampling at Specific Times
For slower reactions, aliquots are taken at intervals and analyzed by titration, gravimetric analysis, or gas chromatography.
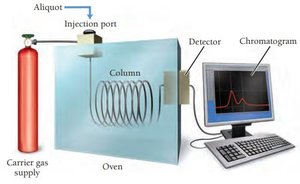
Rate Laws and Reaction Order
Rate Law
The rate law expresses the relationship between reaction rate and reactant concentrations, often determined experimentally.
General Form:
Order: Exponents n and m indicate the order with respect to each reactant; their sum is the overall order.
Rate Constant (k): Proportionality constant specific to the reaction and conditions.
Reaction Orders
Zero Order: Rate is independent of concentration.
First Order: Rate is directly proportional to concentration.
Second Order: Rate is proportional to the square of concentration.
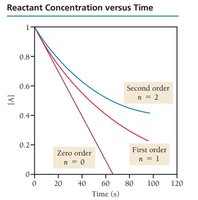
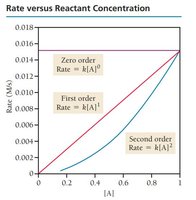
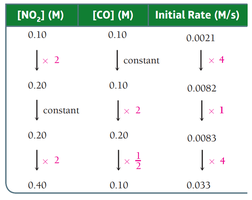
Determining Reaction Order
Reaction order is determined by varying reactant concentrations and observing changes in rate. Data tables help visualize these relationships.
Integrated Rate Laws
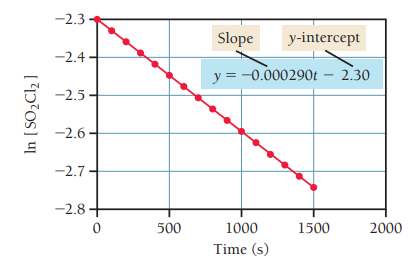
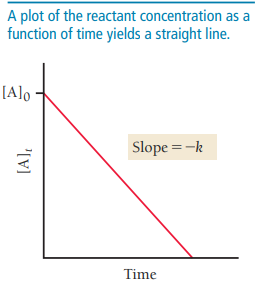
First-Order Integrated Rate Law
For first-order reactions, the integrated rate law relates concentration to time:
Graph of vs. time yields a straight line with slope .
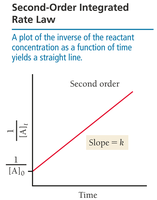
Second-Order Integrated Rate Law
For second-order reactions:
Graph of vs. time yields a straight line with slope .
Zero-Order Integrated Rate Law
For zero-order reactions:
Graph of vs. time yields a straight line with slope .
Half-Life of Reactions
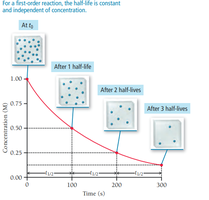
First-Order Half-Life
The half-life () is the time required for the concentration of a reactant to decrease by half. For first-order reactions, half-life is constant and independent of concentration:
Temperature and Reaction Rate
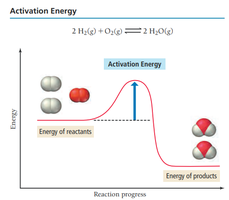
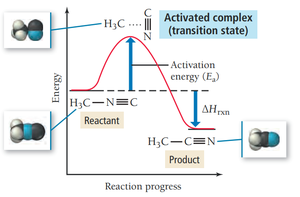
Arrhenius Equation
The Arrhenius equation describes how the rate constant depends on temperature and activation energy:
Activation Energy (): Minimum energy required for a reaction.
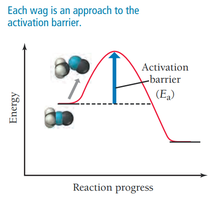
Frequency Factor (A): Number of times reactants approach the activation barrier per unit time.
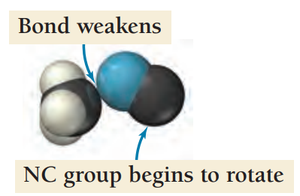
Collision Theory
Effective Collisions
For a reaction to occur, molecules must collide with sufficient energy and proper orientation. Only a fraction of collisions are effective.
Collision Frequency: Number of collisions per second.
Orientation Factor: Probability that molecules are aligned correctly during collision.
Reaction Mechanisms
Elementary Steps and Molecularity
Most reactions occur via a series of elementary steps, each with its own molecularity (number of reactant particles involved).
Unimolecular: One particle.
Bimolecular: Two particles.
Termolecular: Three particles (rare).
Rate-Determining Step
The slowest step in a reaction mechanism determines the overall reaction rate and its rate law.
Catalysis
Catalysts and Reaction Rate
Catalysts increase reaction rates by providing an alternative pathway with lower activation energy. They are not consumed in the reaction.
Homogeneous Catalysts: Same phase as reactants.
Heterogeneous Catalysts: Different phase from reactants.
Enzymes: Biological Catalysts
Enzymes are protein catalysts in biological systems. They bind substrates at active sites, orienting them for reaction and lowering activation energy.
Summary Table: Rate Laws
Order | Rate Law | Integrated Rate Law | Half-Life |
|---|---|---|---|
Zero | |||
First | |||
Second |
