 Back
BackChemical Kinetics: Rates, Mechanisms, and Factors Affecting Reactions
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the study of the speed (rate) of chemical reactions and the factors that influence these rates. Understanding kinetics is essential for controlling reactions in industrial, biological, and environmental contexts.
Reaction Rate: The rate of a reaction measures how quickly reactants are converted to products.
Importance: Controlling reaction rates is crucial in chemical manufacturing, biological systems, and environmental processes.
Defining Reaction Rate
The reaction rate is typically expressed as the change in concentration of a reactant or product per unit time. For reactants, a negative sign is used to indicate consumption.
Formula:
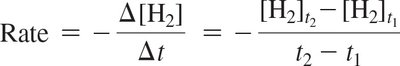
Units: Typically M/s (molarity per second).
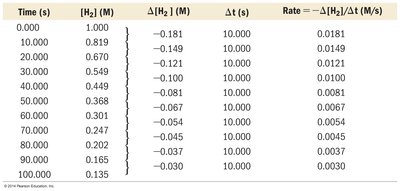
Example: If [H2] decreases from 1.000 M to 0.819 M in 10 seconds, the rate is 0.0181 M/s.
Visualizing Reaction Rates
Reaction rates can be fast or slow, depending on the nature of the reactants and conditions.
Fast vs. Slow Reactions: Fast reactions quickly convert reactants to products, while slow reactions take longer.

Experimental Data and Rate Calculations
Reaction rates are often determined from experimental concentration data over time.
Tabular Data: Shows how [H2] changes over time and how rate is calculated for each interval.
Time (s) | [H2] (M) | Δ[H2] (M) | Δt (s) | Rate (M/s) |
|---|---|---|---|---|
0.000 | 1.000 | -0.181 | 10.000 | 0.0181 |
10.000 | 0.819 | -0.149 | 10.000 | 0.0149 |
20.000 | 0.670 | -0.121 | 10.000 | 0.0121 |
30.000 | 0.549 | -0.100 | 10.000 | 0.0100 |
40.000 | 0.449 | -0.081 | 10.000 | 0.0081 |
50.000 | 0.368 | -0.067 | 10.000 | 0.0067 |
60.000 | 0.301 | -0.054 | 10.000 | 0.0054 |
70.000 | 0.247 | -0.045 | 10.000 | 0.0045 |
80.000 | 0.202 | -0.037 | 10.000 | 0.0037 |
90.000 | 0.165 | -0.030 | 10.000 | 0.0030 |
100.000 | 0.135 |
Average and Instantaneous Rate
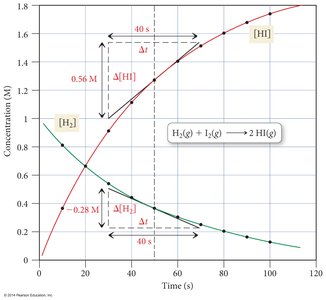
The average rate is calculated over a time interval, while the instantaneous rate is the rate at a specific moment (the slope of the tangent to the concentration vs. time curve).
Example Calculation:
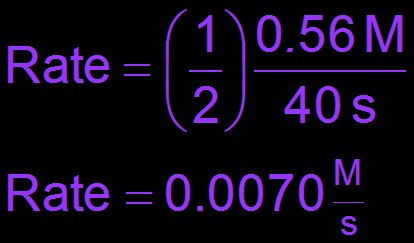
Graphical Representation: Concentration vs. time curves show how rates change as reactants are consumed and products are formed.
Stoichiometry and Reaction Rate
For reactions with different stoichiometric coefficients, the rate is normalized by dividing by the coefficient for each species.
General Rate Expression:
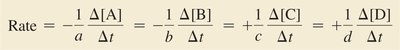
Measuring Reaction Rates
Experimental Techniques
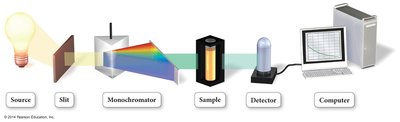
Reaction rates are measured by monitoring the concentration of reactants or products over time using various techniques.
Spectrophotometry: Measures absorbance of light by a sample to determine concentration changes.
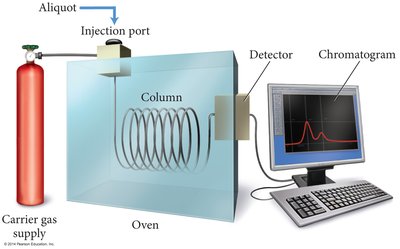
Gas Chromatography: Separates and quantifies volatile components in a mixture.
Factors Affecting Reaction Rate
Nature of Reactants
The physical and chemical properties of reactants influence reaction rates.
Small molecules react faster than large ones.
Gases react faster than liquids, which react faster than solids.
Powdered solids react faster than blocks due to greater surface area.
Ions react faster than molecules because no bonds need to be broken.
Temperature
Increasing temperature generally increases reaction rate by providing more kinetic energy to reactant molecules.
Concentration
Higher concentration of reactants increases the frequency of collisions, leading to a faster reaction rate.
Catalysts
Catalysts speed up reactions by providing an alternative pathway with lower activation energy. They are not consumed in the reaction.
Homogeneous Catalysts: Same phase as reactants.
Heterogeneous Catalysts: Different phase from reactants.
The Rate Law
Mathematical Expression of Rate Law
The rate law relates the reaction rate to the concentrations of reactants, each raised to a power (order).
General Form:
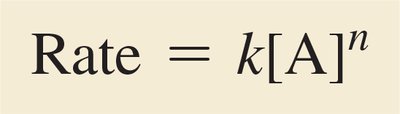
Order: The exponent n is the order with respect to A; the sum of all exponents is the overall order.
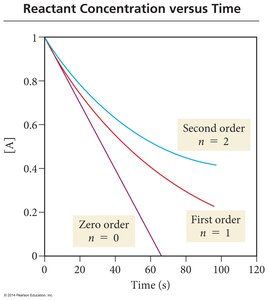
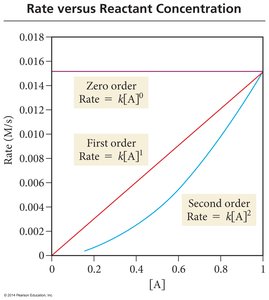
Reaction Order and Graphical Analysis
Different orders of reaction show distinct relationships between concentration and time.
Zero Order: Rate is constant, independent of concentration.
First Order: Rate is directly proportional to concentration.
Second Order: Rate is proportional to the square of concentration.
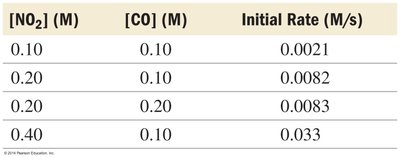
Determining Rate Law from Experimental Data
Initial rate data for different concentrations are used to determine the rate law and rate constant.
[NO2] (M) | [CO] (M) | Initial Rate (M/s) |
|---|---|---|
0.10 | 0.10 | 0.0021 |
0.20 | 0.10 | 0.0082 |
0.20 | 0.20 | 0.0083 |
0.40 | 0.10 | 0.033 |
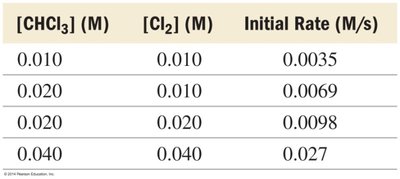
[CHCl3] (M) | [Cl2] (M) | Initial Rate (M/s) |
|---|---|---|
0.010 | 0.010 | 0.0035 |
0.020 | 0.010 | 0.0069 |
0.020 | 0.020 | 0.0098 |
0.040 | 0.040 | 0.027 |
Integrated Rate Laws and Half-Life
Integrated Rate Laws
Integrated rate laws relate concentration to time for zero, first, and second order reactions.
First Order:
Second Order:
Zero Order:
Graphical Determination of Reaction Order
Plotting concentration, ln(concentration), or 1/concentration versus time helps identify the reaction order.
Linear [A] vs. time: Zero order
Linear ln[A] vs. time: First order
Linear 1/[A] vs. time: Second order
Half-Life
The half-life (t1/2) is the time required for the concentration of a reactant to decrease by half.
First Order: (independent of concentration)
Second Order: (depends on initial concentration)
Zero Order: (depends on initial concentration)
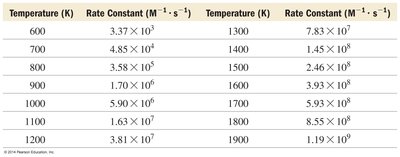
Temperature and Reaction Rate
Arrhenius Equation
The Arrhenius equation describes how the rate constant (k) depends on temperature and activation energy.
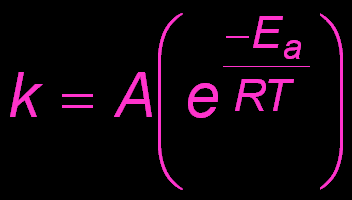
Activation Energy (Ea): Minimum energy required for a reaction to occur.
Frequency Factor (A): Related to the frequency and orientation of collisions.
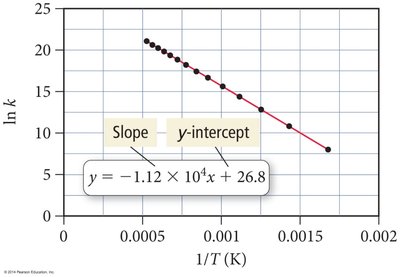
Arrhenius Plots and Activation Energy
Plotting ln(k) versus 1/T yields a straight line whose slope is related to activation energy.
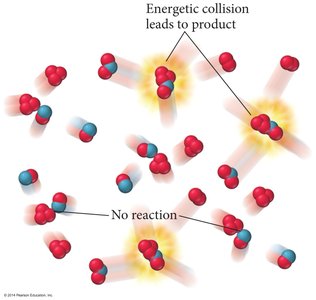
Collision Theory
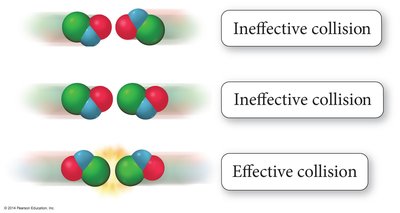
Effective Collisions
For a reaction to occur, molecules must collide with sufficient energy and proper orientation.
Energetic Collisions: Only collisions with enough energy lead to product formation.
Orientation: Reactants must be aligned correctly for bonds to break and form.
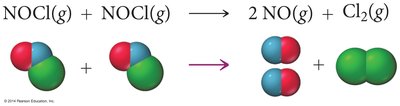
Reaction Mechanisms
Elementary Steps and Molecularity
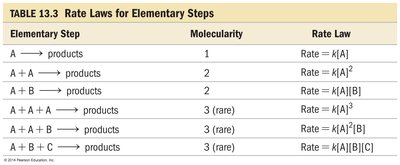
Most reactions occur via a series of elementary steps, each involving one, two, or three reactant particles.
Elementary Step | Molecularity | Rate Law |
|---|---|---|
A → products | 1 | Rate = k[A] |
A + A → products | 2 | Rate = k[A]2 |
A + B → products | 2 | Rate = k[A][B] |
A + A + A → products | 3 (rare) | Rate = k[A]3 |
A + A + B → products | 3 (rare) | Rate = k[A]2[B] |
A + B + C → products | 3 (rare) | Rate = k[A][B][C] |
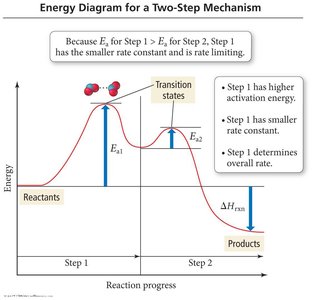
Rate Determining Step
The slowest step in a reaction mechanism determines the overall reaction rate and rate law.
Catalysts and Reaction Pathways
Effect of Catalysts
Catalysts provide an alternative pathway with lower activation energy, increasing the reaction rate.
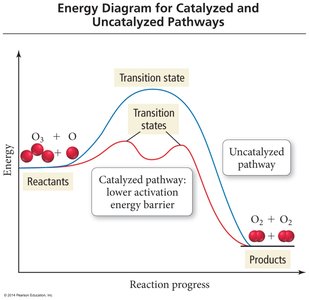
Types of Catalysts
Homogeneous Catalysts: Same phase as reactants (e.g., Cl(g) in O3 destruction).
Heterogeneous Catalysts: Different phase (e.g., solid catalytic converter).
Summary Table: Key Equations and Concepts
Rate Law:
Integrated Rate Laws: First, second, and zero order equations
Arrhenius Equation:
Half-Life: Depends on reaction order
Collision Theory: Effective collisions require sufficient energy and proper orientation
Reaction Mechanisms: Series of elementary steps; rate determined by slowest step
Catalysts: Lower activation energy, increase rate
