 Back
BackChemical Kinetics: Rates, Mechanisms, and Factors Affecting Reactions
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the branch of chemistry that studies the speed (rate) of chemical reactions and the factors that influence these rates. Understanding kinetics is essential for controlling reactions in industrial, biological, and environmental contexts.
Reaction Rate: The rate of a reaction measures how quickly reactants are converted to products.
Importance: Controlling reaction rates is crucial in processes such as drug metabolism, industrial synthesis, and environmental chemistry.
Example: Ectothermic animals like lizards experience slower bodily reactions at lower temperatures, becoming lethargic as a result.

Defining Reaction Rate
Rate as a Change Over Time
The rate of a chemical reaction is analogous to other rates, such as speed, and is defined as the change in a measurable quantity over time.
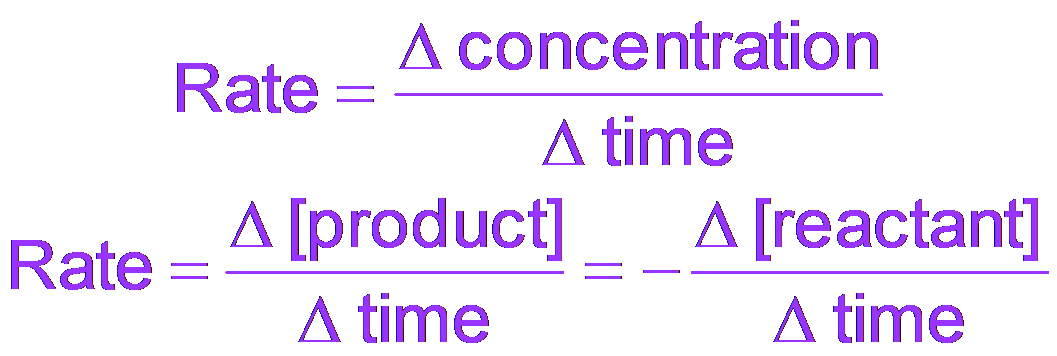
General Rate Formula:
Chemical Rate Formula:
Reactants: Rate is negative for reactants (decreasing concentration).
Products: Rate is positive for products (increasing concentration).
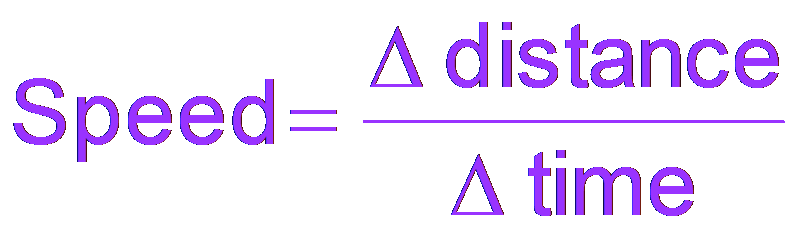
Visualizing Reaction Rates
Fast vs. Slow Reactions
Reactions can proceed at different rates, which can be visualized by tracking the concentration of reactants and products over time.
Fast Reaction: Rapid decrease in reactant concentration and increase in product concentration.
Slow Reaction: Gradual changes in concentrations.



Stoichiometry and Reaction Rate
Relating Rates to Balanced Equations
Reaction rates must account for the stoichiometry of the reaction. The change in concentration for each substance is related to its coefficient in the balanced equation.
Example:
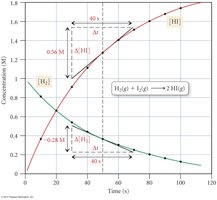
For every 1 mole of H2 used, 2 moles of HI are produced.
Rate is calculated by multiplying the change in concentration by .
Average and Instantaneous Rate
Calculating Reaction Rates
Reaction rates can be measured as average rates over a time interval or as instantaneous rates at a specific moment.
Average Rate:
Instantaneous Rate: Determined by the slope of the tangent to the concentration vs. time curve at a specific point.
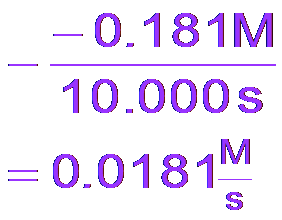
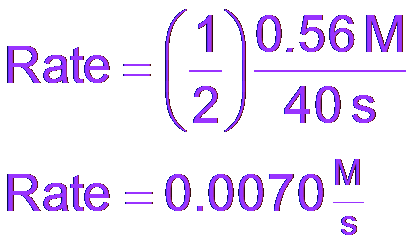
Measuring Reaction Rates
Experimental Methods
Reaction rates are measured by monitoring the concentration of reactants or products over time using various techniques.
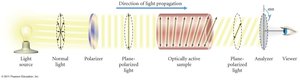
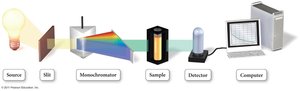
Continuous Monitoring: Used for fast reactions; includes polarimetry, spectrophotometry, and total pressure measurements.
Sampling: Used for slower reactions; involves drawing aliquots and analyzing them by titration, gravimetric analysis, or gas chromatography.
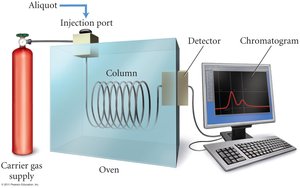
Factors Affecting Reaction Rate
Nature of Reactants, Temperature, Catalysts, and Concentration
Several factors influence how fast a reaction proceeds:
Nature of Reactants: Small molecules, gases, and ions react faster than large molecules, solids, or neutral molecules.
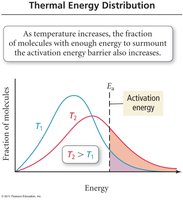
Temperature: Increasing temperature generally increases reaction rate; for many reactions, a 10°C rise doubles the rate.
Catalysts: Substances that speed up (positive catalysts) or slow down (negative catalysts) reactions without being consumed.
Concentration: Higher concentration of reactants increases the frequency of collisions and thus the reaction rate.
The Rate Law
Mathematical Expression of Reaction Rate
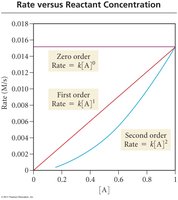
The rate law relates the rate of a reaction to the concentrations of reactants (and sometimes catalysts), each raised to a power called the order.
General Form:
Orders: n and m are determined experimentally and indicate how the rate depends on each reactant.
Rate Constant (k): A proportionality constant unique to each reaction.

Determining Reaction Order
Experimental Determination
Reaction order is found by varying the concentration of each reactant and observing the effect on the reaction rate.
Zero Order: Rate is independent of reactant concentration.
First Order: Rate is directly proportional to reactant concentration.
Second Order: Rate is proportional to the square of reactant concentration.



Graphical Methods for Rate Law Determination
Using Plots to Identify Reaction Order
Plotting concentration, ln(concentration), or 1/concentration versus time helps determine the order of a reaction.
Zero Order: [A] vs. time is linear.
First Order: ln[A] vs. time is linear.
Second Order: 1/[A] vs. time is linear.
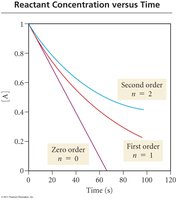
Integrated Rate Laws and Half-Life
Relationship Between Concentration and Time
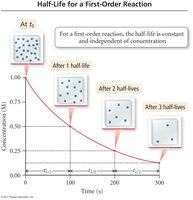
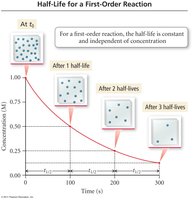
Integrated rate laws describe how reactant concentration changes over time. The half-life is the time required for the concentration to decrease by half.
Zero Order: ;
First Order: ;
Second Order: ;
Temperature and the Arrhenius Equation
Effect of Temperature on Rate Constant
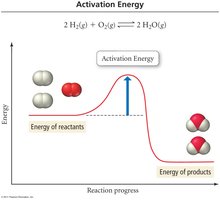
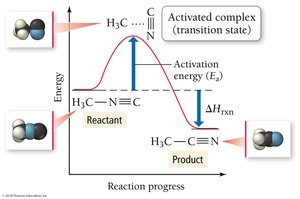
The Arrhenius equation describes how the rate constant (k) depends on temperature and activation energy.
Arrhenius Equation:
Activation Energy (Ea): The minimum energy required for a reaction to occur.
Frequency Factor (A): Represents the frequency of collisions with proper orientation.
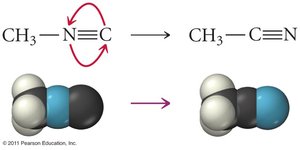
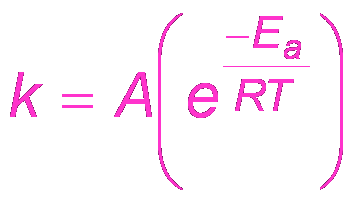
Collision Theory
Requirements for Effective Collisions
For a reaction to occur, molecules must collide with sufficient energy and proper orientation.
Collision Frequency: Number of collisions per second.
Orientation Factor (p): Probability that molecules are oriented correctly during collision.
Effective Collisions: Only collisions meeting both criteria lead to product formation.
Reaction Mechanisms
Elementary Steps and Rate Determining Step
Most reactions occur through a series of elementary steps. The slowest step is the rate-determining step, which controls the overall reaction rate.
Elementary Step: A single molecular event in a mechanism.
Intermediates: Species produced in one step and consumed in another.
Rate Law for Elementary Step: Can be deduced directly from the step's equation.
Catalysts and Enzymes
Role of Catalysts in Reaction Mechanisms
Catalysts provide alternative pathways with lower activation energy, increasing reaction rates without being consumed. Enzymes are biological catalysts that facilitate reactions by binding substrates at active sites.
Homogeneous Catalysts: Same phase as reactants.
Heterogeneous Catalysts: Different phase from reactants.
Enzymes: Protein catalysts in biological systems.
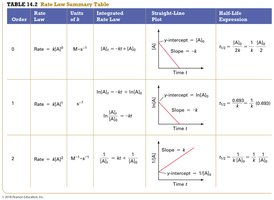
Summary Table: Rate Laws
Comparison of Zero, First, and Second Order Reactions
The following table summarizes the key features of rate laws for different reaction orders:
Order | Rate Law | Integrated Rate Law | Straight-Line Plot | Half-Life Expression |
|---|---|---|---|---|
Zero | Rate = k[A]0 = k | [A] vs. time | ||
First | Rate = k[A] | ln[A] vs. time | ||
Second | Rate = k[A]2 | 1/[A] vs. time |
