 Back
BackChemical Kinetics: Rates, Rate Laws, and Integrated Rate Equations
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Chemical Kinetics
Chemical kinetics is the study of the rates at which chemical reactions occur and the factors that influence these rates. Understanding kinetics is essential for applications in drug design, industrial processes, and environmental chemistry.
Reaction Rate: The change in concentration of a reactant or product per unit time.
Applications: Pharmacokinetics, industrial synthesis, environmental impact (e.g., slow removal of CFCs).
Defining Reaction Rate
The rate of a chemical reaction is typically expressed as the change in concentration of a reactant or product over a specified time interval.
General Formula:
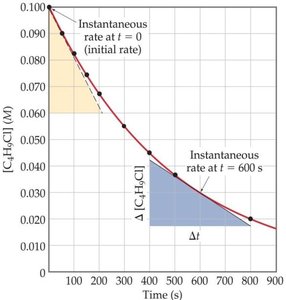
Instantaneous Rate: The rate at a specific moment, determined by the slope of the tangent to the concentration vs. time curve.
Initial Rate: The rate at the very start of the reaction.
Factors Affecting Reaction Rates
Several factors influence how quickly a reaction proceeds:
Concentration of Reactants: Higher concentrations generally increase the rate.
Temperature: Raising temperature typically increases reaction rate.
Physical State: Reactants in the same phase react faster than those in different phases.
Catalysts: Catalysts speed up reactions without being consumed.
Measuring Reaction Rates
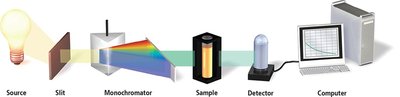
Experimental Determination
Reaction rates are measured by monitoring the concentration of at least one component over time. Spectrophotometry is a common technique, where absorbance is proportional to concentration according to Beer’s Law:
Beer’s Law: A: Absorbance \varepsilon: Molar absorptivity b: Path length c: Concentration (M)
Reaction Rates and Stoichiometry
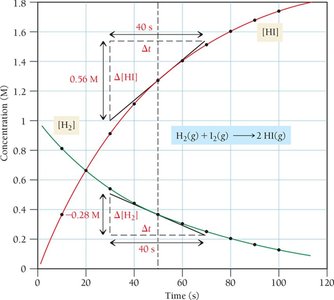
Stoichiometric Relationships
The rate of disappearance of reactants and appearance of products depends on their stoichiometric coefficients. For a general reaction:
General Reaction:
Rate Expression:
Rate Laws
Definition and Formulation
A rate law expresses the relationship between the reaction rate and the concentrations of reactants, each raised to a power (order of reaction). The exponents are determined experimentally and are not necessarily the stoichiometric coefficients.
General Rate Law:
k: Rate constant (depends on reaction and temperature)
Order: The sum of exponents (m + n) gives the overall order.
Method of Initial Rates
The initial rate method involves measuring the rate at the start of the reaction for various reactant concentrations to determine the exponents in the rate law.
Integrated Rate Laws
First Order Reactions
For a first order reaction (A → products), the integrated rate law relates concentration to time:
Integrated Rate Law:
Linear Form:
Plot: A plot of vs. time yields a straight line with slope -k.
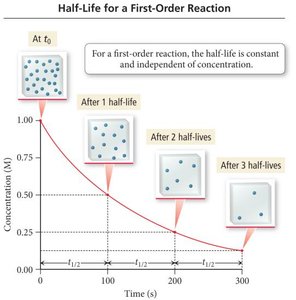
Half-Life for First Order Reactions
The half-life (t1/2) is the time required for the concentration of a reactant to decrease by half. For first order reactions:
Half-Life Formula:
Characteristic: Half-life is independent of initial concentration.
Second Order Reactions
For a second order reaction (A → products):
Integrated Rate Law:
Plot: A plot of vs. time yields a straight line with slope +k.
Half-Life Formula:
Characteristic: Half-life increases as concentration decreases.
Zeroth Order Reactions
For a zeroth order reaction (A → products):
Integrated Rate Law:
Plot: A plot of vs. time yields a straight line with slope -k.
Half-Life Formula:
Characteristic: Half-life decreases as concentration decreases.
Summary Table: Integrated Rate Laws
Order | Rate Law | Integrated Rate Law | Half-Life | Linear Plot |
|---|---|---|---|---|
0 | R = k | [A] vs. t | ||
1 | R = k[A] | ln[A] vs. t | ||
2 | R = k[A]^2 | 1/[A] vs. t |
Example Applications
Drug Decomposition: Understanding the rate law helps predict how quickly a drug breaks down in the body.
Industrial Synthesis: Optimizing reaction conditions for maximum yield and efficiency.
Environmental Chemistry: Predicting the persistence of pollutants in the atmosphere.
