 Back
BackChemical Kinetics: Reaction Rates and Factors Affecting Rate
Study Guide - Smart Notes

Chemical Kinetics
Introduction to Reaction Rates
Chemical kinetics is the study of the speed at which chemical reactions occur and the factors that influence these rates. Understanding reaction rates is essential for controlling chemical processes in laboratory and industrial settings.
Reaction rate is a measure of how fast reactants are converted to products.
Rates can be expressed as the change in concentration of reactants or products per unit time.
Defining Rate
The concept of rate is fundamental in chemistry and other sciences. It describes how much a quantity changes over a given period.
For example, speed is defined as the change in distance over time.
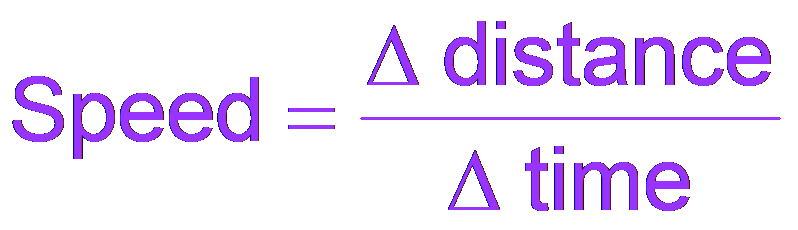
Defining Reaction Rate
The rate of a chemical reaction is typically measured by the change in concentration of a reactant or product over time.
For reactants, the rate is negative because their concentration decreases.
For products, the rate is positive as their concentration increases.
Visualizing Reaction Rates
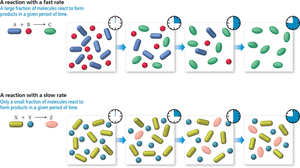
A reaction with a fast rate converts a large fraction of reactants to products quickly.
A reaction with a slow rate converts only a small fraction of reactants to products in the same time period.
Measuring Reaction Rates
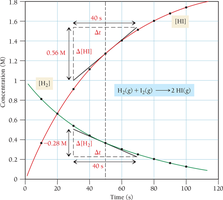
Reactant and Product Concentrations as a Function of Time
Reaction rates are often monitored by measuring concentrations of reactants and products at various times.
Graphs of concentration versus time help visualize how rates change during a reaction.
Average Rate
The average rate is calculated over a specific time interval and is a linear approximation of the actual rate.
The larger the time interval, the more the average rate deviates from the instantaneous rate.
Instantaneous Rate
The instantaneous rate is the rate at a specific moment and is determined by the slope of the tangent to the concentration vs. time curve.
Mathematically, it is the first derivative of concentration with respect to time.
Reaction Rate and Stoichiometry
For reactions with different stoichiometric coefficients, the rate of change for each substance is adjusted by dividing by its coefficient.
Example: For the reaction , the rate expressions for each species are related by their coefficients.
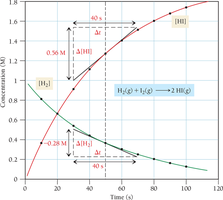
Expressing Reaction Rates
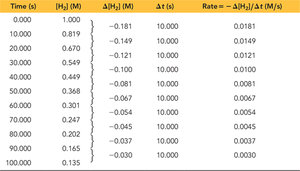
Calculating Average Rate
To calculate the average rate, use the formula: Example calculation for I-: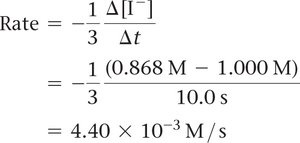
Relating Rate to Other Species
For other reactants or products, use their stoichiometric coefficients to relate their rates of change.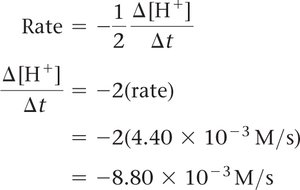
Factors Affecting Reaction Rate
Nature of Reactants
The physical state and chemical nature of reactants influence reaction rates.
Small molecules react faster than large ones.
Gases react faster than liquids, which react faster than solids.
Powdered solids react faster than chunks due to greater surface area.
Ions react faster than molecules because no bonds need to be broken.
Temperature
Increasing temperature generally increases reaction rate.
Rule of thumb: For many reactions, a 10°C increase doubles the rate.
Catalysts
Catalysts speed up reactions without being consumed.
Positive catalysts increase rate; negative catalysts decrease rate.
Homogeneous catalysts are in the same phase as reactants; heterogeneous catalysts are in a different phase.
Reactant Concentration
Higher concentration increases reaction rate by increasing collision frequency.
For gases, concentration depends on partial pressure.
For solutions, concentration is measured in molarity.
The Rate Law
Rate Law and Reaction Order
The rate law relates the rate of a reaction to the concentration of reactants.
General form:
Order is the exponent for each reactant; the sum of exponents is the overall order.
Order is determined experimentally, not from stoichiometry.
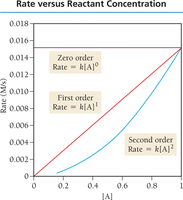
Reaction Order Examples
Zero order: Rate is independent of concentration.
First order: Rate is directly proportional to concentration.
Second order: Rate is proportional to the square of concentration.
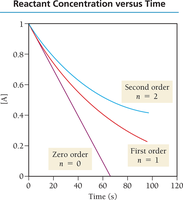
Determining Rate Laws
Experimental Determination
Rate laws are determined by measuring initial rates at different reactant concentrations.
Method of initial rates: Compare how rate changes when one reactant's concentration is varied.
Example: NO2 and CO Reaction
Tabulated data shows how changing concentrations affects rate.
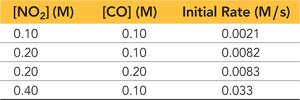
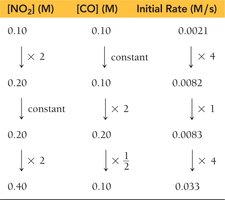
Calculating Reaction Order
Use ratios of rates and concentrations to solve for the order.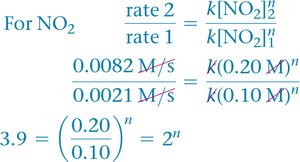
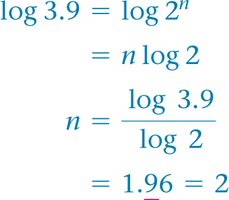
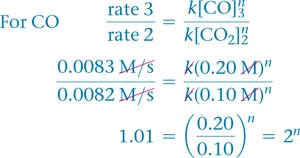
Rate Constant Calculation
Once the rate law is known, solve for the rate constant using any data point.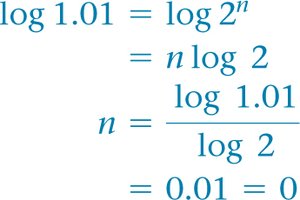
Integrated Rate Laws
Relating Concentration and Time
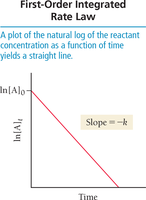
Integrated rate laws relate reactant concentration to time, allowing prediction of concentrations at any point.
First-order:
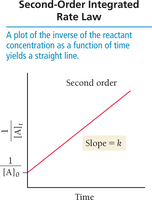
Second-order:
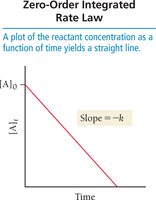
Zero-order:
Example: First-Order Integrated Rate Law
Given and , find at :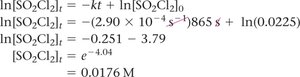
Half-Life
Definition and Calculation
The half-life () is the time required for the concentration of a reactant to decrease by half.
For first-order reactions, half-life is constant and independent of initial concentration:
For zero- and second-order reactions, half-life depends on initial concentration.
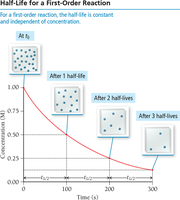
Visualizing Half-Life
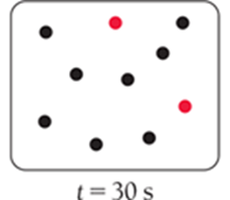
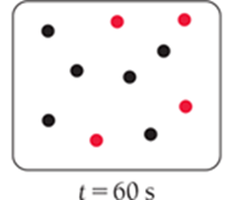
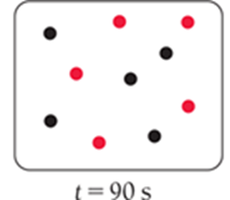
For a first-order reaction, the concentration decreases by half at regular intervals.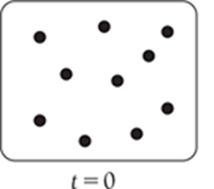
Summary of Kinetics Relationships
Rate law relates rate to concentration.
Integrated rate law relates concentration to time.
Half-life is the time for concentration to halve.
Order and rate law must be determined experimentally.
Graphical Determination of Reaction Order
Plot [A] vs. time: linear for zero order.
Plot ln[A] vs. time: linear for first order.
Plot 1/[A] vs. time: linear for second order.
Practice and Application
Example Problems
Calculate rate constants, half-lives, and concentrations using provided formulas and data.
Interpret graphs and tables to determine reaction order and rate law.
